Abstract
Purpose
We recently described ovarian genotypes and sub-genotypes of the FMR1 gene with distinctly associated ovarian aging patterns, which in infertile women follow a typical X-linked distribution pattern. Whether normally fertile women, however, also demonstrate the same distribution, is unknown.
Methods
We, therefore, investigated ovarian FMR1 genotype and sub-genotype distribution in 182 oocyte donor candidates in comparison to 339 infertile controls. As previously reported, genotype designation was made, based on a normal range of CGG n = 26–34 (median 30), defining women as normal (norm), heterozygous (het) or homozygous (hom). Het and hom genotypes were further subdivided into sub-genotypes, based on whether abnormal alleles were above (high) or below normal range (low).
Results
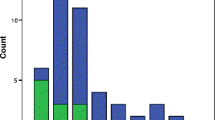
Oocyte donors presented with 47.8% norm, 45.1% het and 7.1% hom genotypes, confirming a typical X-linked distribution pattern. They, however, still subtly differed from infertility patients, especially in het sub-genotypes.
Conclusion
These findings validate recently newly described ovarian genotypes and sub-genotypes, reaffirming their relevance to female fertility/infertility.

Similar content being viewed by others
References
Gleicher N, Weghofer A, Barad DH. Ovarian reserve determinations suggest new function of FMR1 (fragile X gene) in regulating ovarian aeging. Reprod Biomed Online. 2010;20:768–75.
Gleicher N, Weghofer A, Lee IH, Barad DH. FMR1 genotype with autoimmunity-associated polycystic ovary-like phenotype and decreased pregnancy chance. PLoS ONE. 2010;5:e15303.
Gleicher N, Weghofer A, Lee IH, Barad DH. Association of FMR1 genotypes with in vitro fertilization (IVF) outcomes based on ethnicity/race. PLoS ONE. 2011;6:e18781.
Willemsen R, Levenga J, Oostra B. CGG repeat in the FMR1 gene: size matters. Clin Genet. 2011;80:214–25.
Chen LS, Tassone F, Sahota P, Hagerman PJ. The (CGG)n repeat element within the 5’untranslated region of the FMR1 message provides both positive and negative cis effects on in-vivo translation of a downstream reporter. Hum Molec Genetics. 2003;12:307–3074.
Gleicher N, Weghofer A, Oktay K, Barad D. Relevance of triple CGG repeats in the FMR1 gene to ovarian reserve. Reprod Biomed Online. 2009;19:385–90.
Barad DH, Weghofer A, Gleicher N. Age-specific levels of basal follicle-stimulating hormone assessment of ovarian function. Obstet Gynecol. 2007;109:1404–10.
Barad DH, Weghofer A, Kim A. The het-norm/high FMR1 sub-genotype increases oocyte yields at advanced female age. Fertil Steril 2011;Suppl:S119
Funding
This work was supported by the Foundation for Reproductive Medicine and intramural grants from the Center for Human Reproduction.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ roles
N.G. and D.H.B. contributed equally to study design, study conduct and preparation of the manuscript, with N.G, being primarily responsible for writing the manuscript and D.H.B. being primarily responsible for data analysis. A.W. contributed to study design and A.K. contributed to data analysis. All authors reviewed and approved the final manuscript.
Capsule
Egg donor candidates were found to have a similar, but not identical, distribution of ovarian FMR1 genotypes and sub-genotypes, compatible with previously reported functions.
N.G. and D.H.B. are listed as co-inventors on a pending U.S. patent application, which claims diagnostic benefits in infertility from assessing FMR1 genotypes and sub-genotypes.
Here presented material was in part presented in abstract form at the 67th Annual Meeting of the American Society for Reproductive Medicine in Orlando, Florida, October 15–19, 2011.
Rights and permissions
About this article
Cite this article
Gleicher, N., Weghofer, A., Kim, A. et al. Comparison of ovarian FMR1 genotypes and sub-genotypes in oocyte donors and infertile women. J Assist Reprod Genet 29, 529–532 (2012). https://doi.org/10.1007/s10815-012-9745-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-012-9745-7