Abstract
Purpose
Prenatal diagnosis with ultrasound findings compatible with skeletal dysplasia due to FGFR3 mutations over a 9 year period in pregnancies and abortuses.
Methods
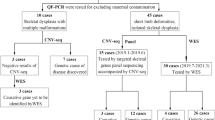
54 samples were studied. Aneuploidy studies were carried out on all samples. By sequencing analysis, we determined mutations for achondroplasia (ACH), hypochondroplasia (HCH), and type I and type II tanathophoric dysplasia (TD).
Results
2 chorionic villi samples had a G380R mutation due to a mother with ACH; 4 amniotic fluid samples with TDs in which the foetuses had micromelia plus hypoplastic thoraces; 5 samples from abortuses with TDs. Neither ACH nor HCH occurred in sporadic cases.
Conclusions
Molecular studies in ongoing pregnancies are indicated in cases with an affected parent, a family history with positive molecular studies (maternal anxiety), and when the US finding demonstrates micromelia with a hypoplastic thorax. A protocol for tissues of abortuses should include an X-ray, pathologic anatomy, and genetic studies.

Similar content being viewed by others
References
Spranger J. Bone dysplasia families. Pathol Immunpathol Res. 1988;7:76–80.
Keegan K, Johnson DE, Williams LT, Hayman MJ. Isolation of an additional member of the fibroblast growth factor receptor family, FGFR3. Proc Acad Sci USA. 1991;88:1095–1099.
Keegan K, Rooke L, Hayman M, Spurr NK. The fibroblast growth factor receptor 3 gene (FGFR3) is assigned to human chromosome 4. Cytogenet Cell Genet. 1993;62:172–175. 12.
Vajo Z, Francomano CA, Wilkin DJ. The molecular and genetic basis of fibroblast growth factor receptor 3 disorders: the achondroplasia family of skeletal dysplasia, Muenke craniosynostosis and Crouzon syndrome with acanthosis nigricans. Endocr Rev. 2000;21:23–39.
Orioli IM, Castila EE, Barbosa-Neto JG. The birth prevalence rates for the skeletal dysplasias. J Med Genet. 1986;23:328–332.
Martínez-Frías ML, Cereijo A, Bermejo E, López M, Sánchez M, Golnalo C. Epidemiological aspects fo Mendelian syndromes in a Spanish population sample: I. autosomal dominant malformation syndromes. Am J Med Genet. 1991;38:622–625.
Tavormina PL, Shiang R, Thompson LM, Zhu YZ, Wilkin DJ, Lachman RS, et al. Thanatophoric dysplasia (types I and II) caused by distint mutations in fibroblast growth factor receptor 3. Nat Genet. 1995;9:321–328.
Tavormina PL, Bellus GA, Webster MK, Bamshad MJ, Fraley AE, McIntosh I, et al. A novel skeletal dysplasia with developmental delay and acanthosis nigricans is caused by a Lys650Met mutation in the fibroblast growth factor receptor 3 gene. Am J Hum Genet. 1999;64:722–731.
Stoll C, Dott B, Roth MP, Alembik Y. Birth prevalence rates of skeletal dysplasias. Clin Genet. 1989;35:88–92.
Rolf C, Nieschlag E. Reproductive functions, fertility and genetic risks of ageing men. Exp Clin Endocrinol Diabetes. 2001;109:68–74.
Wyrobeck AJ, Eskenazi B, Young S, Arnheim N, Tieman-Boege I, Jabs EW, et al. Advancing age has different effects on DNA damage, chromatin integrity, gene mutations, and aneuploidies sperm. Proc Natl Aca Sci USA. 2006;103:9601–9606.
Rousseau F, Bonaventure J, Legeai-Mallet L, Pelet A, Rozet JM, Maroteaux P, et al. Mutations in the gene encoding fibroblast receptor growth factor receptor-3 in achondroplasia. Nature. 1994;371:252–254.
Shiang R, Thompson LM, Zhu YZ, Church DM, Fielder TJ, Bocian M, et al. Mutations in the transmenbrane domain of FGFR3 cause the most common genetic form of dawrfism, achondroplasia. Cell. 1994;78:335–342.
Climent C, Lorda I, Urioste M, Gairi JM, Rodriguez JI, Rubio V. Acondroplasia: estudio molecular de 28 pacientes. Med Clin. 1998;110:492–494.
Bellus GA, McIntosh I, Smith EA, Aylsworth AS, Kaitila I, Horton WA, et al. A recurrent mutation in the tyrosine kinase domain of fibroblast growth factor receptor 3 causes hypochondroplasia. Nat Genet. 1995;10:357–359.
Rousseau F, Bonaventure J, Legeai-Mallet L, Schmidt H, Weissenbach J, Maroteaux P, et al. Clinical and genetic heterogeneity of hypochondroplasia. J Med Genet. 1996;33:749–752.
Parrilla BV, Leeth EA, Kambich MP, Chilis P, MacGregor S. Antenatal detection of skeletal dysplasias. J Ultrasound Med. 2003;22:255–258.
Adinolfi M, Pertl B, Sherlock J. Rapid detection of aneuploidies by microsatellite and the quantitative fluorescent polymerase chain reaction. Prenat Diagn. 1997;17:1299–1311.
Wapner RJ, Sorokin Y, Thom EA, Jonhson F, Dudley DJ, Spong CT, et al. Single versus weekly courses of antenatal corticosteroids: evaluation of safety and efficacy. Am J Obstet Gynecol. 2006;195:633–642.
Trujillo-Tiebas MJ, Riveiro R, Queipo A, Vallespin E, Cantalapiedra D, Lorda-Sánchez I, et al. Gene Symbol: FGFR3. Hum Genet. 2004;115(4):348.
Karadimas C, Sifakis S, Valsamopoulos P, Makatsoris C, Velissariou V, Nasioulas G, et al. Prenatal diagnosis of hypochondroplasia: report of two cases. Am J Med Genet Part A. 2006;140:998–1003.
Gooding HC, Boehm K, Thompson RE, Hadley D, Francomano CA, Bowles BB. Issues sorrounding prenatal testing for achondroplasia. Prenat Diagn. 2002;22:933–940.
Camera G, Dodero D, De Pascale S. Prenatal diagnosis of thanatophoric dysplasia at 24 weeks. Am J Med Genet. 1984;18:39–43.
Elejalde BR, Elejalde MM. Thanatophoric dysplasia: fetal manifestations and prenatal diagnosis. Am J Med Genet. 1985;22:669–683.
Chen Ch-P, Chern S-R, Shih J-Ch, Wayseen W, Yeh L-F, Chang T-Y, et al. Prenatal diagnosis and genetic analysis of type I and type II thanatophoric dysplasia. Prenat Diagn. 2001;21:89–95.
Todros T, Massarenti I, Gaglioti P, Biolcati G, De Felice C. Fetal short femur length in the second trimester and the outcome of pregnancy. BJOG. 2004;111:83–85.
Modaff P, Horton VK, Pauli RM. Errors in the prenatal diagnosis of children with achondroplasia. Prenat Diagn. 1996;16:525–530.
Kataoka S, Sawai H, Yamada H, Kanazawa N, Koyama K, Nishimura G, et al. Radiographic and genetic diagnosis of sporadic hypochondroplasia early in the neonatal period. Prenat Diagn. 2004;24:45–49.
Ramalho C, Matias A, Brandão O, Montenegro N. Critical evaluation of elective termination of pregnancy in a tertiary fetal medicine center during 43 months: correlation of prenatal diagnosis findings and postmortem examination. Prenat Diagn. 2006;26:1084–1088.
Aslan H, Yldirim G, Ongut C, Ceylan Y. Termination of pregnancy for fetal anomaly. Int J Gynaecol Obstet. 2007;99:221–224.
Guillem P, Fabre B, Cans C, Robert-Gnansia E, Jouk PS. Trends in elective termination of pregnancy between 1989 and 2000 in French county (the Isère). Prenat Diagn. 2003;23:877–883.
Vaknin Z, Ben-Ami I, Reish O, Herman A, Maymon R. Fetal abnormalities leading to termination of singleton pregnancy: the 7-year experience of a single medical center. Prenat Diagn. 2006;26:938–943.
Orioli IM, Castilla EE, Scarano G, Mastroiacovo P. Effect of paternal age in achondroplasia, thanatophoric dysplasia, and osteogenesis imperfecta. Am J Med Genet. 1995;50:209–217.
Wilkin DJ, Szabo JK, Cameron R, Henderson S, Bellus GA, Mack ML, et al. Mutation in fibroblast growth-factor receptor 3 in sporadic cases of achondroplasia occur exclusively on the paternally derived chromosome. Am J Hum Genet. 1998;63:711–716.
Acknowledgements
This group is founded by the Centro de Investigaciones Biomédicas en Red de Enfermedades Raras (CIBERER) [ISCIII, Madrid, Spain]. We would like to thank to the Fundación Ramón Areces and INERGEN (FIS PIC03/05; FIS PI05/0263) for its support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule Molecular cytogenetic studies are indicated and complementary to ultrasound in familial cases involving mutations in the FGFR3 gene.
Trujillo-Tiebas and Fenollar-Cortés contributed equally to this work.
Rights and permissions
About this article
Cite this article
Trujillo-Tiebas, M.J., Fenollar-Cortés, M., Lorda-Sánchez, I. et al. Prenatal diagnosis of skeletal dysplasia due to FGFR3 gene mutations: a 9-year experience. J Assist Reprod Genet 26, 455–460 (2009). https://doi.org/10.1007/s10815-009-9339-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-009-9339-1