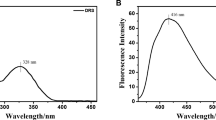
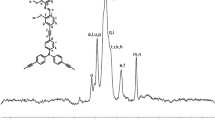
A novel dipodal fluorescent sensor, N1,N3-bis(2-(2,3,4-trihydroxybenzylidene)amino)ethylmalo namide (MEP), suitable for the practical measurement of sodium concentration has been successfully developed and characterized by several spectroscopic techniques. The design of the dipodal scaffold includes a central unit, spacer, and fluorophore moiety as structural key features. The fluorescence sensor MEP adopts a photoinduced electron transfer mechanism and shows excellent selectivity for Na(I) among other biologically and environmentally important metal ions, viz., Na(I), K(I), Al(III), Cr(III), Fe(III), Fe(II), Co(II), Ni(II), Cu(II), and Zn(II) in DMSO by demonstrating a remarkable enhancement in the fluorescence intensity from 345.5 to 705.5 a.u. at λmax = 532.9 nm. The 1:2 binding stoichiometry between the ligand and Na(I) ion was confirmed by Stern–Volmer and Hill plots. The association constant determined for the ligand with the sodium metal ion is found to be very high, 7.7 × 106 M–2, which may be attributed to the trapping of sodium ions into the pseudo cavities of the ligand created by interaction of the ligand and sodium ions. The studies explore potential applications of the ligand for Na(I) ions detection in environmental and industrial applications.
Similar content being viewed by others
References
A. P. de Silva, H. Q. Gunaratne, T. Gunnlaugsson, A. J. Huxley, C. P. McCoy, J. T. Rademacher, and T. E. Rice, Chem. Rev., 97, 1515–1566 (1997).
J. F. Callan, A. P. de Silva, and D. C. Magri, Tetrahedron, 61, 8551–8588 (2005).
А. Coskun, M. Banaszak, R. D. Astumian, J. F. Stoddart, and B. A. Grzybowski, Chem. Soc. Rev., 41, 19–30 (2012).
R. Akbar, M. Baral, and B. K. Kanungo, J. Coord. Chem., 71, No. 1, 135–154 (2018).
M. Baral, A. Gupta, R. Akbar, and B. K. Kanungo, J. Appl. Chem., 2016, 3757418/1–3757418/10 (2016).
M. Baral, A. Gupta, and B. K. Kanungo, Spectrochim. Acta A, 162, 6–15 (2016).
M. Burnier, Sodium in Health and Disease, Informa Healthcare, New York (2008).
The Na+, K+ Pump, Part B: Cellular Aspects, Eds. J. C. Skou, J. G. Norby, A. B. Maunsbach, M. Esmann, and A. R. Liss, Wiley, New York (1998).
E. Murphy and D. A. Eisner, Circ. Res., 104, 292 (2009).
D. M. Bers, W. H. Barry, and S. Despa, Cardiovasc. Res., 57, 897 (2003).
T. R. Harring, N. S. Deal, and D. C. Kuo, Emerg. Med. Clin. North Am., 32, 379–401 (2014).
Z. J. Twardowski, Hemodial. Int., 12, 412–425 (2008).
A. Minta and R. Y. Tsien, J. Biol. Chem., 264, 19449 (1989).
Chemosensors of Ion and Molecule Recognition; NATO ASI Series, Eds. J. P. Desvergne, and A. W. Czarnik, Kluwer Academic, Dordrecht, The Netherlands (1996).
O. S. Wolfbeis, Fiber Optic Chemical Sensors and Biosensors, II, CRC Press, Boca Raton, FL (1991).
P. Gans, A. Sabatini, and A. Vacca, Talanta, 43, 1739–1753 (1996).
P. Gans, A. Sabatini, and A. Vacca, Ann. Chim. (Rome), 89, 45–49 (1999).
S. K. Sahoo, S. E. Muthu, M. Baral, and B. K. Kanungo, Spectrochim. Acta A, 63, 574 (2006).
L. J. Bellamy, The Infra-Red Spectra of Complex Molecules, 3rd ed., Chapman and Hall Ltd., London (1975).
R. M. Silverstein, G. C. Bassler, and T. C. Morrill, Spectrometric Identification of Organic Compounds, 4th edn., Wiley, New York (1981).
Y. K. Tsui, S. Devaraj, and Y. P. Yen, Sens. Actuat. B: Chem., 161, 510–519 (2012).
А. P. de Silva, H. Q. N. Gunaratne, J. L. Habib-Jiwan, C. P. McCoy, T. E. Rice, and J. P. Soumillion, Angew. Chem., Int. Ed. Engl., 34, 1728–1731 (1995).
H. Huarui, M. A. Mortellaro, M. J. P. Leiner, S. T. Young, R. J. Fraatz, and J. K. Tusa, Anal. Chem., 75, 549–555 (2003).
Graham R. C. Hamilton, Suban K. Sahoo, Sukanta Kamila, Narinder Singh, Navneet Kaur, Barry W. Hyland, and John F. Callan, Chem. Soc. Rev., 7, No. 44(13), 4415–4432 (2015).
Y. M. Poronik, G. Clermont, M. Blanchard-Desce, and D. T. Gryko, J. Org. Chem., 78, No. 23, 11721–11732 (2013).
Kundan Tayade, G. Krishna Chaitanya, Jasminder Singh, Narinder Singh, Sopan Ingle, Sanjay Attarde, and Anil Kuwar, J. Lumin., 154, 68–73 (2014).
M. Everett, A. Jolleys, W. Levason, D. Pugh, and G. Reid, Chem. Commun., 50, 5843–5846 (2014).
H. A. Benesi and J. H. Hilderbrand, J. Am. Chem. Soc., 71, 2703–2704 (1949).
G. Kaur and N. Kaur, Sens Actuat. B: Chem., 265, 134–141 (2018).
W. Caetano and M. Tabak, J. Colloid Interface Sci., 225, 69–81 (2000).
Y.-J. Hua, Y. Liua, T.-Q. Sunb, A.-M. Bai, J.-Q. Lu, and Z.-B. Pi, Int. J. Biol. Macromol., 39, 280–285 (2006).
H. W. Jun, L. A. Luzzi, and P. L. Hsu, J. Pharm. Sci., 61, No. 11, 1835–1837 (1972).
X. Mei and C. Wolf, J. Am. Chem. Soc., 126, 14736–14737 (2004).
J. R. Lakowicz, Principles of Fluorescence Spectroscopy, 3rd edn., Springer, Berlin (2006).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Zhurnal Prikladnoi Spektroskopii, Vol. 87, No. 5, pp. 821–831, September–October, 2020.
Rights and permissions
About this article
Cite this article
Dangi, V., Baral, M. & Kanungo, B.K. Dipodal Molecular Device as Fluorescent Sensor for Na(I) Detection. J Appl Spectrosc 87, 893–903 (2020). https://doi.org/10.1007/s10812-020-01086-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10812-020-01086-z