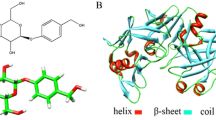
Herein, the interaction between gliclazide (GCZ) and pepsin (PEP) was explored under simulated physiological conditions by multiple spectroscopy methods and molecular docking. The results showed that a new complex of 1:1 was formed between GCZ and PEP, thereby quenching the endogenous fluorescence of PEP. The addition of GCZ changed the conformation of PEP and increased the α-helix content in PEP from 20.16% to 21.13%. Using the binding constant Ka of the reaction between GCZ and PEP and the number of binding sites n, the binding rate formulas of GCZ and PEP were deduced. It was estimated that when the patient takes 40 mg of GCZ, the PEP in the gastric juice will be reduced by 96.69%. That meant taking GCZ will seriously affect the patient's digestive function. The results of molecular docking indicated that the GCZ binding site was located in the active center of PEP. The interaction of the two was driven by electrostatic attraction and hydrogen bonding forces.
Similar content being viewed by others
References
Q. Tang, J. H. Su, H. Y. Cao, L. H. Wang, L. X. Gao, and X. F. Zheng, Spectrosc. Spectr. Anal., 37, No. 11, 3485–3492 (2017).
A. T. Buddanavar and S. T. Nandibewoor. J. Pharmac. Analysis, 7, No. 3, 148–155 (2017).
T. Ahammad, M. Begum, A. F. M. T. Rahman, M. Hasan, S. R. Paul, S. Eamen, M. I. Hussain, M. H. Ali, M. A. Islam, M. M. Rahman, and M. Rashid, Pharmacol. Pharm., 6, No. 3, 125–131 (2015).
Q. J. Zhang, B. S. Liu, G. X. Li, and R. Han, Luminescence, 31, No. 5, 1109–1114 (2016).
Y. R. Wang, Q. Fang, C. H. Guo, and Y. Liu. Spectrosc. Spectr. Anal.,36, No. 10, 3414–3421 (2016).
G. J. Nan, P. Wang, J. Sun, J. H. Lv, M. W. Ding, L. Yang, Y. P. Li, and G. D. Yang, Luminescence, 31, No. 8, 1524–1531 (2016).
Y. Q. Wang and H. M. Zhang, J. Agric. Food Chem., 61, No. 46, 11191–11200 (2013).
R. Shahidha, S. Muthu, M. Raja, R. R. Muhamed, B. Narayana, P. S. Nayak, and B. K. Sarojini, Optik, 140, 1127–1142 (2017).
J. K. Maurya, M. U. Mir, U. K. Singh, N. Maurya, N. Dohare, S. Patel, A. Ali, and R. Patel, Biopolymers, 103, No. 7, 406–415 (2015).
N. Barbero, E. Barni, C. Barolo, P. Quagliotto, G. Viscardi, L. Napione, S. Pavan, and F. Bussolino, Dyes Pigments, 80, No. 3, 307–313 (2009).
M. M. Yin, P. Dong, W. Q. Chen, S. P. Xu, L. Y. Yang, F. L. Jiang, and Y. Liu Langmuir,33, No. 21, 5108–5116 (2017).
J. C. Li, N. Li, Q. H. Wu, Z. Wang, J. J. Ma, C. Wang, and L. J. Zhang. J. Mol. Struct., 833, Nos. 1–3, 184–188 (2007).
L. L. He, X. Wang, B. Liu, J. Wang, Y. G. Sun, E. J. Gao, and S. K. Xu, J. Lumin., 131, No. 2, 285–290 (2011).
Q. J. Zhou, J. F. Xiang, Y. L. Tang, J. P. Liao, C. Y. Yu, H. Zhang, L. Li, Y. Y. Yang, and G. Z. Xu, Colloids Surf. B, 61, No. 1, 75–80 (2008).
W. J. Zhang, X. J. Xiong, F. Wang, L. Li, Y. Zhang, W. P. Xiao, and Y. Liu, Sci. Chin. Chem., 57, No. 12, 1690–1695 (2014).
A. Jahanban-Esfahlan, V. Panahi-Azar, and S. Sajedi. Biopolymers, 103, No. 11, 638–645 (2015).
Y. Q. Wang, X. Y. Wang, J. Wang, Y. M. Zhao, W. J. He, and Z. J. Guo. Inorg. Chem., 50, No. 24, 12661–12668 (2011).
X. Y. Shi, H. Cao, F. L. Ren, and M. Xu. Chem. Biodivers, 4, No. 12, 2780–2790 (2007).
X. J. Guo, X. D. Sun, and S. K. Xu, J. Mol. Struct., 931, Nos. 1–3, 55–59 (2009).
X. R. Pan, P. F. Qin, R. T. Liu, and J. Wang, J. Agric. Food Chem., 59, No. 12, 6650–6656 (2011).
X. Zhao, Y. L. Wang, J. L. Guo, H. R. Han, and M. S. Xie, Acta Sci. Circumstantiae, 25, No. 1, 52–57 (2005).
O. D. Putra, E. Yonemochi, and H. Uekusa. Cryst. Growth. Des., 16, No. 11, 6568–6673 (2016).
J. P. Feng, X. Chen, R. L. Wang, and D. T. Yin, Chin. Med. Eng., 20, No. 3, 3–5 (2012).
X.Y. Gao, Y. C. Tang, W. Q. Rong, X. P. Zhang, W. J. Zhao, and Y. Q. Zi, Am. J. Anal. Chem., 2, No. 2, 250–257 (2011).
F. L. Cui, Q. Z. Zhang, X. J. Yao, H. X. Luo, Y. Yang, L. X. Qin, G. R. Qu, and Y. Lu, Pestic. Biochem. Phys., 90, No. 2, 126–134 (2008).
Y. X. Wu, Y. Qian, H. Cui, X. M. Lai, X. C. Xie, and X. R. Wang, Environ. Toxicol. Chem., 30, No. 12, 2697–2700 (2011).
D. Yuan, Z. L. Shen, R. T. Liu, P. H. Wei, and C. Z. Gao, J. Chin. Chem. Soc., 61, No. 2, 255–262 (2014).
N. Ibrahim, H. Ibrahim, S. Kim, J. P. Nallet, and F. Nepveu. Biomacromolecules, 11, No. 12, 3341–3351 (2010).
M. M. Cui, B. S. Liu, T. T. Li, and S. T. Duan. Spectrosc. Lett., 49, No. 9, 573–581 (2016).
Author information
Authors and Affiliations
Corresponding author
Additional information
Abstract of article is published in Zhurnal Prikladnoi Spektroskopii, Vol. 86, No. 4, p. 671, July–August, 2019.
Rights and permissions
About this article
Cite this article
Ma, LH., Liu, BS., Bian, G. et al. Effect of the Hypoglycemic Agent Gliclazide on the Gastric Digestive Function: Binding Mechanism Between Gliclazide and Pepsin. J Appl Spectrosc 86, 748–755 (2019). https://doi.org/10.1007/s10812-019-00889-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10812-019-00889-z