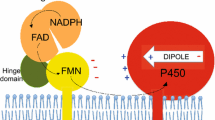
The influence of the amino-acid substitution Arg486Cys on the conformational stability of recombinant cytochrome P450 7B1 (CYP7B1, oxysterol 7α-hydroxylase) was studied. The single base change was shown to decrease the free energy of the transition of the heme-protein from its native state to a denatured one, which pointed to a lower thermodynamic stability for the mutant form of the enzyme. This could be the cause of the metabolic disruption of neurosteroids and, as a consequence, the appearance of neurodegenerative diseases.
Similar content being viewed by others
References
F. P. Guengerich, Chem. Res. Toxicol., 21, No. 1, 70–83 (2008).
D. W. Russell, Ann. Rev. Biochem., 72, 137–174 (2003).
A. R. Stiles, J. G. McDonald, D. R. Bauman, and D. W. Russell, J. Biol. Chem., 284, No. 42, 28485–28489 (2009).
K. A. Rose, G. Stapleton, K. Dott, M. P. Kieny, R. Best, M. Schwarz, D. W. Russell, L. Bjorkhem, J. Seckl, and R. Lathe, Proc. Natl. Acad. Sci. USA, 94, No. 10, 4925–4930 (1997).
K. D. Setchell, M. Schwarz, N. C. O’Connell, E. G. Lund, D. L. Davis, R. Lathe, H. R. Thompson, W. R. Tyson, R. J. Sokol, and D. W. Russell, J. Clin. Invest., 102, No. 9, 1690–1703 (1998).
R. Schule, E. Brandt, K. N. Karle, M. Tsaousidou, S. Klebe, S. Klimpe, M. Auer-Grumbach, A. H. Crosby, C. A. Hubner, L. Schols, T. Deufel, and C. Beetz, Neurogenetics, 10, No. 2, 97–104 (2009).
J. L. Yau, S. Rasmuson, R. Andrew, M. Graham, J. Noble, T. Olsson, E. Fuchs, R. Lathe, and J. R. Seckl, Neuroscience, 121, No. 2, 307–314 (2003).
C. Goizet, A. Boukhris, A. Durr, C. Beetz, J. Truchetto, C. Tesson, M. Tsaousidou, S. Forlani, L. Guyant-Marechal, B. Fontaine, J. Guimaraes, B. Isidor, O. Chazouilleres, D. Wendum, D. Grid, F. Chevy, P. F. Chinnery, P. Coutinho, J. P. Azulay, I. Feki, F. Mochel, C. Wolf, C. Mhiri, A. Crosby, A. Brice, and G. Stevanin, Brain, 132, No. 6, 1589–1600 (2009).
M. K. Tsaousidou, K. Ouahchi, T. T. Warner, Y. Yang, M. A. Simpson, N. G. Laing, P. A. Wilkinson, R. E. Madrid, H. Patel, F. Hentati, M. A. Patton, A. Hentati, P. J. Lamont, T. Siddique, and A. H. Crosby, Am. J. Hum. Genet., 82, No. 2, 510–515 (2008).
A. V. Yantsevich, Y. V. Dichenko, F. Mackenzie, D. V. Mukha, A. V. Baranovsky, A. A. Gilep, S. A. Usanov, and N. V. Strushkevich, FEBS J., 281, No. 6, 1700–1713 (2014).
M. P. Eftink, Biokhimiya, 63, No. 3, 276–284 (1998).
A. K. Bhuyan, Biopolymers, 93, No. 2, 186–199 (2010).
U. K. Laemmli, Nature, 227, No. 5259, 680–685 (1970).
G. Weber and F. J. W. Teale, Discuss. Faraday Soc., 27, 134–141 (1959).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Prikladnoi Spektroskopii, Vol. 82, No. 1, pp. 96–102, January–February, 2015.
Rights and permissions
About this article
Cite this article
Dichenko, Y.V., Yantsevich, A.V. & Usanov, S.A. Structural and Functional Characteristics of Oxysterol 7α-Hydroxylase with Amino-Acid Substitution R486C and Their Relation to the Appearance of Neurodegenerative Diseases. J Appl Spectrosc 82, 91–97 (2015). https://doi.org/10.1007/s10812-015-0069-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10812-015-0069-0