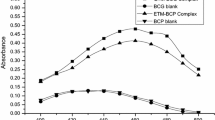
Two simple spectrophotometric methods have been developed to analyze ofloxacin (OFX) in pharmaceuticals. The methods are based on the oxidation of OFX by a measured excess of cerium(IV) sulfate in H2SO4 medium. This was followed by the determination of the unreacted oxidant by reacting it with either p-toluidine (p-TD) and measuring the absorbance at 525 nm (method A) or o-dianisidine (o-DA) and measuring the absorbance at 470 nm (method B). In both methods, the amount of cerium(IV) sulfate reacted corresponds to the amount of OFX. Calibration graphs were linear over the ranges of 0–120 and 0–4 g/ml OFX for methods A and B, respectively. The calculated molar absorptivity (2.34⋅103 and 5.99⋅104), Sandell sensitivity, and limit of quantification for the methods are reported. The intra-day precision (%RSD) and accuracy (%RE) were < 8.0 and ≤ 4.0%, respectively, and the inter-day RSD and RE values were within 5 and 4.0%, respectively. The applicability of the methods was demonstrated by determining OFX in tablets with an accuracy (%RE) of < 3% and precision (%RSD) of ≤2.65%. The accuracy of the methods was further ascertained by recovery experiments via a standard-addition procedure.
Similar content being viewed by others
References
K. Sat, U. Matsura, M. Inone, T. Ueno, Y. Osada, H. Ogawa, M. Mitshuhashi, Antimicrob. Agents Chemother., 22, 548 (1982).
F. A. El-Yazbi, Spectr. Lett., 25, 279–291 (1992).
Q. J. Gong, J. L. Quiao, L. M. Du, C. Dong, Talanta, 53, 359–365 (2000).
A. Le Coguic, R. Bidault, R. Farinotti, A. Dauphin, J. Chromatogr., 434, 320–323 (1988).
O. Okazaki, H. Aoki, H. Hakusui, J. Chromatogr., 563, 313–322 (1991).
G. Carlucci, P. Mazzeo, T. Fantozzi, Anal. Lett., 26, 2193–2201 (1993).
V. M. Shinde, B. S. Desai, N. M. Tendolkar, Indian Drugs, 35, 715–717 (1998).
U. P. Halkar, P. B. Ankalkope, Indian Drugs, 37, 585–588 (2000).
M. S. Ali, M. Ghori, A. Saeed, J. Chromatogr. Sci., 40, 429–433 (2002).
A. J. N. Groenveld, J. R. B. Brouwers, Pharm. World Sci., 8, 79–84 (1986).
R. R. Kalta, R. Sharma, S. C. Chaturvedi, Ind. J. Pharm. Sci., 70, 491–494 (2008).
K. P. Chan, K. O. Chu, W. W-K. Lai, K. W. Choy, C. C. Wang, D. S. C. Lam, C. P. Pang, Anal. Biochem., 353, 30–36 (2006).
T. Ohkubo, M. Kudo, K. Sugawara, Anal. Sci., 7, 741–743 (1991).
M. A. Gracia, C. Solans, A. Calvo, M. Royo, E. Hernandez, R. Rey, M. A. Bregante, Chromatographia, 55, 431–434 (2002).
B. Srividya, R. M. Cardoza, P. D. Amin, Indian Drugs, 40, 41–43 (2003).
S. W. Sun, A. L. Wu, J. Liq. Chromatogr. Relat. Technol., 22, 281–296 (1999).
C. Horstkoetter, G. Blaschke, J. Chromatogr. B., 754, 169–178 (2000).
S. S. Zhang, H. Liu, Y. J. Wu, C. L. Yu, Analyst, 126, 441–445 (2001).
A. Tamer, Anal. Chim. Acta, 231, 129–131 (1990).
M. Tuncel, Z. Atkosar, Pharmazie, 47, 642–643 (1992).
G. Zhou, J. Pan, Anal. Chim. Acta, 307, 49–53 (1995).
M. Rizk, F. Belal, F. Aly, N. El-Enany, Talanta, 46, 83–89 (1998).
J. Wu, H. Zhao, L. Wei, T. Z. Ai, X. Z. Dong, Fenxi Huaxue, 29, 1106 (2001).
F. Paul, J. L. Adcock, Anal. Chim. Acta, 541, 3–12 (2005).
S. Hanwen, L. Liqing, C. Xueyan, Anal. Sci., 22, 1145–1149 (2006).
Y. Rao, Y. Tong, X. R. Zhang, G. Luo, W. Baeyens, J. Fluores., 17, 481–491 (2007).
L. Yi, H. Zhao, S. Chen, L. Jin, D. Zheng, Z. Wu, Talanta, 61, 403–409 (2003).
Y. D. Liang, J. F. Song, X. F. Yang, Anal. Chim. Acta, 510, 21–28 (2004).
B. Li, Z. Zhang, L. Zhao, Ch. Xu, Talanta, 57, 765–771 (2002).
L. da Silveira Ev., E. E. S. Schapoval, J. Pharm. Biomed. Anal., 27, 91–96 (2002).
British Pharmacopoeia. Vol. III, Her Majesty’s Stationery office, London, 1357 (2003).
United States Pharmacopoeia XXII, National Formulary XVII, Rockville, USA Convention, 912 (1990).
European Pharmacopoeia. EDQM, Council of Europe, Strasbourg, France, 5, 2131 (2005).
P. U. Patel, B. N. Suhagia, M. M. Patel, G. C. Patel, G. N. Patel, Indian Pharmacist, 6, 59–61 (2007).
Y. M. Issa, F. M. Abdel-Gawad, M. A. Abou Table, H. M. Hussein, Anal. Lett., 30, 2071–2084 (1997).
C. S. P. Sastry, K. R. Rao, D. Siva Prasad, Talanta, 42, 311–316 (1995).
C. J. Eboka, S. O. Aigbavboa, J. O. Akerele, J. Antimicrob. Chemother., 39, 639–641 (1997).
S. C. Mathur, Y. Kumar, N. Murugesan, Y. K. S. Rathore, P. D. Sethi, Indian Drugs, 29, 376–377 (1992).
M. S. Soledad, M. I. Albero, C. Sanchez-Pedreno, M. S. Abuherba, Eur. J. Pharm. Biopharm., 61, 87–93 (2005).
C. S. P. Sastry, K. R. Rao, D. S. Prasad, Indian Drugs, 32, 172–175 (1995).
K. B. Vinay, H. D. Revanasiddappa, M. R. Divya, N. Rajendraprasad, Ecle. Quim., 34, 65–78 (2009).
J. Bassett, R. C. Denney, G. H. Jeffery, J. Mendham, Vogel’s Textbook of Quantitative Analysis, 4th ed., Longman Group Limited, 598 (1978).
A. K. Batalian, S. M. Polyakova, Zh. Khim., 14, 178 (1963), Abstract No 19G15.
Z. Marczenko, Separation and Spectrophotometric Determination of Elements, Transl. Ed. M. Masson, Ellis Horwood Limited Publ., 222 (1986).
Z. Holzbecher, L. Davis, M. Kral, L. Sucha, and F. Vlacil, Handbook of Organic Reagents in Inorganic Analysis, Ellis Horwood Series in Analytical Chemistry, J. Wiley & Sons, New York, London, Sydney, Toronto, 364 (1976).
Int. Conf. Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use, ICH Harmonised Tripartite Guideline, Validation of Analytical Procedures: Text and Methodology Q2(R 1), Complementary Guideline on Methodology dated 06 November 1996, incorporated in November 2005, London.
J. Inczedy, T. Lengyel, A. M. Ure, IUPAC Compendium of Analytical Nomenclature: Definitive Rules, Blackwell Science Inc., Boston (1998).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Zhurnal Prikladnoi Spektroskopii, Vol. 78, No. 3, pp. 410–418, May–June, 2011.
Rights and permissions
About this article
Cite this article
Ramesh, P.J., Basavaiah, K., Rajendraprasad, N. et al. Spectrophotometric determination of ofloxacin in pharmaceuticals by redox reaction. J Appl Spectrosc 78, 383–391 (2011). https://doi.org/10.1007/s10812-011-9480-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10812-011-9480-3