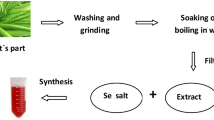
Abstract
Phycoerythrin, a phycobiliprotein recognized for its brilliant crimson hue and essential role in light harvesting during photosynthesis, was extracted from the red alga Porphyridium purpureum. A final concentration of 0.3 mg mL−1, accompanied by an exceptional purity index of 6.05 was achieved. Then, its stability in response to pH fluctuations was evaluated, elucidating its robust resilience to varying pH conditions. To gauge its antioxidant prowess, two established techniques, the Ferric Reducing Antioxidant Power (FRAP) and 2,2'-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) assays, were employed, revealing both the inherent antioxidant capacity and a dose-dependent response. The obtained IC50 values were 22.61 mM for FRAP and 0.159 mg mL−1 for ABTS. After assessing its stability and antioxidant capacity, phycoerythrin encapsulated by an electrospray-assisted technique, employing diverse encapsulating materials. Impressively, each treatment surpassed an encapsulation efficiency threshold of 80%, opening promising avenues for controlled-release systems and targeted applications. The study culminated in an exploration of the protein's antiproliferative potential against colon cancer cells (CaCo2), both in its free and encapsulated states. At the highest tested concentration (0.3 mg mL−1) at 48 h, the non-encapsulated pigment exhibited a cellular viability of 41.0 ± 2.93%, whereas the encapsulated pigment reached a lower level of 27.27 ± 8.29%. These results show that encapsulation significantly enhances the in vitro anticancer activity of this pigment over time, underscoring its potential in biomedical applications.






Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Abdin M, Salama MA, Gawad RMA, Fathi MA, Alnadari F (2021) Two-steps of gelation system enhanced the stability of Syzygium cumini anthocyanins by encapsulation with sodium alginate, maltodextrin, chitosan and gum arabic. J Polym Environ 29:3679–3692
Abdul Mudalip SK, Khatiman MN, Hashim NA, Che Man R, Arshad ZIM (2021) A short review on encapsulation of bioactive compounds using different drying techniques. Mater Today Proc 42:288–296
Adir N, Bar-Zvi S, Harris D (2020) The amazing phycobilisome. Biochim Biophys Acta Bioenerg 1861:148047
Aguilar KC, Tello F, Bierhalz ACK, Romo G, Ma G, Martínez Flores HE, Grosso CRF (2015) Protein adsorption onto alginate-pectin microparticles and films produced by ionic gelation. J Food Eng 154:17–24
ATCC (2023) DMEM: F-12 Medium. https://www.atcc.org/products/30-2006. Accesed 4 Sept 2023
Bao C, Jiang P, Chai J, Jiang Y, Li D, Bao W, Liu B, Liu B, Norde W, Li Y (2019) The delivery of sensitive food bioactive ingredients: Absorption mechanisms, influencing factors, encapsulation techniques and evaluation models. Food Res Int 120:130–140
Basheva D, Moten D, Stoyanov P, Belkinova D, Mladenov R, Teneva I (2018) Content of phycoerythrin, phycocyanin, alophycocyanin and phycoerythrocyanin in some cyanobacterial strains: Applications. Eng Life Sci 18:861–866
Beer S, Eshel A (1985) Determining phycoerythrin and phycocyanin concentrations in aqueous crude extracts of red algae. Mar Freshw Res 36:785
Benavides J, Rito-Palomares M (2004) Bioprocess intensification: A potential aqueous two-phase process for the primary recovery of B-phycoerythrin from Porphyridium cruentum. J Chromatogr B 807:33–38
Bennacef C, Desobry-Banon S, Probst L, Desobry S (2021) Advances on alginate use for spherification to encapsulate biomolecules. Food Hydrocoll 118:106782
Castro-Muñoz R, García-Depraect O (2021) Membrane-based harvesting processes for microalgae and their valuable-related molecules: A review. Membranes 11:585
Chen K, Zhang H (2019) Alginate/pectin aerogel microspheres for controlled release of proanthocyanidins. Int J Biol Macromol 136:936–943
Comunian TA, Monterrey-Quintero ES, Thomazini M, Balieiro JCC, Piccone P, Pittia P, Favaro-Trindade CS (2011) Assessment of production efficiency, physicochemical properties and storage stability of spray-dried chlorophyllide, a natural food colourant, using gum Arabic, maltodextrin and soy protein isolate-based carrier systems. Int J Food Sci Technol 46:1259–1265
Donoso-Quezada J, Guajardo-Flores D, González-Valdez J (2020) Enhanced exosome-mediated delivery of black bean phytochemicals (Phaseolus vulgaris L.) for cancer treatment applications. Biomed Pharmacother 131:110771
Eshaghzadeh H, Shahbazi M, Akhavan SR, Pereira L (2023) Purification and characterization of phycoerythrin from the Caspian Sea red macroalga (Osmundea caspica). J Appl Phycol 35:317–330
Falkeborg MF, Roda-Serrat MC, Burnæs KL, Nielsen ALD (2018) Stabilising phycocyanin by anionic micelles. Food Chem 239:771–780
Fernandes F, Silkina A, Fuentes-Grünewald C, Wood EE, Ndovela VLS, Oatley-Radcliffe DL, Lovitt RW, Llewellyn CA (2020) Valorising nutrient-rich digestate: Dilution, settlement and membrane filtration processing for optimisation as a waste-based media for microalgal cultivation. Waste Manage 118:197–208
Foglio Bonda A, Regis L, Giovannelli L, Segale L (2020) Alginate/maltodextrin and alginate/shellac gum core-shell capsules for the encapsulation of peppermint essential oil. Int J Biol Macromol 162:1293–1302
Gaignard C, Gargouch N, Dubessay P, Delattre C, Pierre G, Laroche C, Fendri I, Abdelkafi S, Michaud P (2019) New horizons in culture and valorization of red microalgae. Biotechnol Adv 37:193–222
Galland-Irmouli AV, Pons L, Luçon M, Villaume C, Mrabet NT, Guéant JL, Fleurence J (2000) One-step purification of R-phycoerythrin from the red macroalga Palmaria palmata using preparative polyacrylamide gel electrophoresis. J Chromatogr B 739:117–123
Gao S, Tan H, Li D (2023) Oridonin suppresses gastric cancer SGC-7901 cell proliferation by targeting the TNF-alpha/androgen receptor/TGF-beta signalling pathway axis. J Cell Mol Med 27:2661–2674
Guo X, Wang Y, Qin Y, Shen P, Peng Q (2020) Structures, properties and application of alginic acid: A review. Int J Biol Macromol 162:618–628
Hariyadi DM, Islam N (2020) Current status of alginate in drug delivery. Adv Pharmacol Pharm Sci 2020:1–16
Hemlata, Afreen S, Fatma T (2018) Extraction, purification and characterization of phycoerythrin from Michrochaete and its biological activities. Biocatal Agric Biotechnol 13:84–89
Hsieh-Lo M, Castillo G, Ochoa-Becerra MA, Mojica L (2019) Phycocyanin and phycoerythrin: Strategies to improve production yield and chemical stability. Algal Res 42:101600
Islan GA, de Verti IP, Marchetti SG, Castro GR (2012) Studies of Ciprofloxacin encapsulation on alginate/pectin matrixes and its relationship with biodisponibility. Appl Biochem Biotechnol 167:1408–1420
Jain D, Bar-Shalom D (2014) Alginate drug delivery systems: Application in context of pharmaceutical and biomedical research. Drug Dev Ind Pharm 40:1576–1584
Kannaujiya VK, Kumar D, Pathak J, Sinha RP (2019) Phycobiliproteins and their commercial significance. In: Mishra AK, Tiwari DN, Rai AN (eds) Cyanobacteria. From Basic Science to Applications. Elsevier, London pp. 207–216
Koo SY, Cha KH, Song D-G, Chung D, Pan C-H (2014) Microencapsulation of peppermint oil in an alginate-pectin matrix using a coaxial electrospray system. Int J Food Sci Technol 49:733–739
Korkmaz O, Girgin B, Sunna Ç, Yavaşer R, Karagözler AA (2016) Production and investigation of controlled drug release properties of tamoxifen loaded alginate-gum arabic microbeads. J Turk Chem Soc Sect A Chem 3:47-58
Li J, Jiang C, Lang X, Kong M, Cheng X, Liu Y, Feng C, Chen X (2016) Multilayer sodium alginate beads with porous core containing chitosan based nanoparticles for oral delivery of anticancer drug. Int J Biol Macromol 85:1–8
Li W, Su H-N, Pu Y, Chen J, Liu L-N, Liu Q, Qin S (2019) Phycobiliproteins: Molecular structure, production, applications, and prospects. Biotechnol Adv 37:340–353
Liu L-N, Chen X-L, Zhang X-Y, Zhang Y-Z, Zhou B-C (2005) One-step chromatography method for efficient separation and purification of R-phycoerythrin from Polysiphonia urceolata. J Biotechnol 116:91–100
López-Alarcón C, Denicola A (2013) Evaluating the antioxidant capacity of natural products: A review on chemical and cellular-based assays. Anal Chim Acta 763:1–10
Lu X, Nan F, Feng J, Lv J, Liu Q, Liu X, Xie S (2020) Effects of different environmental factors on the growth and bioactive substance accumulation of Porphyridium purpureum. Int J Environ Res Public Health 17:2221
Ma J, Hu J, Sha X, Meng D, Yang R (2022) Phycobiliproteins, the pigment-protein complex form of natural food colorants and bioactive ingredients. Crit Rev Food Sci Nutr 4:1–19
Ma Y, Thurecht KJ, Coombes AGA (2021) Development of enteric-coated, biphasic chitosan/HPMC microcapsules for colon-targeted delivery of anticancer drug-loaded nanoparticles. Int J Pharm 607:121026
Mirza UA, Chait BT (1997) Do proteins denature during droplet evolution in electrospray ionization? Int J Mass Spectrom Ion Processes 162:173–181
Morais AÍS, Vieira EG, Afewerki S, Sousa RB, Honorio LMC, Cambrussi ANCO, Santos JA, Bezerra RDS, Furtini JAO, Silva-Filho EC, Webster TJ, Lobo AO (2020) Fabrication of polymeric microparticles by electrospray: The impact of experimental parameters. J Funct Biomater 11:4
Moreira A, Lawson D, Onyekuru L, Dziemidowicz K, Angkawinitwong U, Costa PF, Radacsi N, Williams GR (2021) Protein encapsulation by electrospinning and electrospraying. J Control Release 329:1172–1197
Munier M, Jubeau S, Wijaya A, Morançais M, Dumay J, Marchal L, Jaouen P, Fleurence J (2014) Physicochemical factors affecting the stability of two pigments: R-phycoerythrin of Grateloupia turuturu and B-phycoerythrin of Porphyridium cruentum. Food Chem 150:400–407
Munier M, Morançais M, Dumay J, Jaouen P, Fleurence J (2015) One-step purification of R-phycoerythrin from the red edible seaweed Grateloupia turuturu. J Chromatogr B 992:23–29
Mysliwa-Kurdziel, B., & Solymosi, K. (2017). Phycobilins and phycobiliproteins used in food industry and medicine. Mini-Rev Med Chem 17:1173-1193
Pagels F, Guedes AC, Amaro HM, Kijjoa A, Vasconcelos V (2019) Phycobiliproteins from cyanobacteria: Chemistry and biotechnological applications. Biotechnol Adv 37:422–443
Patel SN, Sonani RR, Jakharia K, Bhastana B, Patel HM, Chaubey MG, Singh NK, Madamwar D (2018) Antioxidant activity and associated structural attributes of Halomicronema phycoerythrin. Int J Biol Macromol 111:359–369
Puzorjov A, McCormick AJ (2020) Phycobiliproteins from extreme environments and their potential applications. J Exp Bot 71:3827–3842
Ramu Ganesan A, Kannan M, Karthick Rajan D, Pillay AA, Shanmugam M, Sathishkumar P, Johansen J, Tiwari BK (2022) Phycoerythrin: A pink pigment from red sources (rhodophyta) for a greener biorefining approach to food applications. Crit Rev Food Sci Nutr 1:1–19
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 26:1231–1237
Reenu G, John J (2023) Phycoerythrin as a potential natural colourant: A mini review. Int J Food Sci Technol 58:513–519
Rossano R, Ungaro N, D’Ambrosio A, Liuzzi GM, Riccio P (2003) Extracting and purifying R-phycoerythrin from Mediterranean red algae Corallina elongata Ellis & Solander. J Biotechnol 101:289–293
Rumpf J, Burger R, Schulze M (2023) Statistical evaluation of DPPH, ABTS, FRAP, and Folin-Ciocalteu assays to assess the antioxidant capacity of lignins. Int J Biol Macromol 233:123470
Sabet S, Rashidinejad A, Melton LD, Zujovic Z, Akbarinejad A, Nieuwoudt M, Seal CK, McGillivray DJ (2021) The interactions between the two negatively charged polysaccharides: Gum Arabic and alginate. Food Hydrocoll 112:106343
Sarkarat R, Mohamadnia S, Tavakoli O (2023) Recent advances in non-conventional techniques for extraction of phycobiliproteins and carotenoids from microalgae. Braz J Chem Eng 40:321–342
Soukoulis C, Bohn T (2018) A comprehensive overview on the micro- and nano-technological encapsulation advances for enhancing the chemical stability and bioavailability of carotenoids. Crit Rev Food Sci Nutr 58:1–36
Sui S-F (2021) Structure of phycobilisomes. Annu Rev Biophys 50:53–72
Sun L, Wang S, Gong X, Zhao M, Fu X, Wang L (2009) Isolation, purification and characteristics of R-phycoerythrin from a marine macroalga Heterosiphonia japonica. Protein Expr Purif 64:146–154
Tan H, Gao S, Zhuang Y, Dong Y, Guan W, Zhang K, Xu J, Cui J (2016) R-Phycoerythrin induces SGC-7901 apoptosis by arresting cell cycle at S phase. Mar Drugs 14:166
Tian Y, Liu Y, Zhang L, Hua Q, Liu L, Wang B, Tang J (2020) Preparation and characterization of gelatin-sodium alginate/paraffin phase change microcapsules. Colloids Surf A 586:124216
Tong WY, Ahmad Rafiee AR, Leong CR, Tan W-N, Dailin DJ, Almarhoon ZM, Shelkh M, Nawaz A, Chuah LF (2023) Development of sodium alginate-pectin biodegradable active food packaging film containing cinnamic acid. Chemosphere 336:139212
Ulagesan S, Nam T-J, Choi Y-H (2021) Extraction and purification of R-phycoerythrin alpha subunit from the marine red algae Pyropia yezoensis and Its biological activities. Molecules 26:6479
UTEX (2023) F/2 medium. https://utex.org/products/f-2-medium?variant=31722216915034. Accessed 7 Jul 2023
Xie J, Jiang J, Davoodi P, Srinivasan MP, Wang C-H (2015) Electrohydrodynamic atomization: A two-decade effort to produce and process micro-/nanoparticulate materials. Chem Eng Sci 125:32–57
Ying X, Gao J, Lu J, Ma C, Lv J, Adhikari B, Wang B (2021) Preparation and drying of water-in-oil-in-water (W/O/W) double emulsion to encapsulate soy peptides. Food Res Int 141:110148
Zhao P, Wang X, Niu J, He L, Gu W, Xie X, Wu M, Wang G (2020) Agar extraction and purification of R-phycoerythrin from Gracilaria tenuistipitata, and subsequent wastewater treatment by Ulva prolifera. Algal Res 47:101862
Acknowledgements
The authors would like to thank the School of Engineering and Science and the FEMSA-Biotechnology Center at Tecnológico de Monterrey for their support through the Molecular and Systems Bioengineering Research Group. Javier Donoso-Quezada and Arisbe Silva-Nuñez thank the National Council for the Humanities, Sciences and Technologies of Mexico (CONAHCyT) for the financial support through scholarships numbers 995384 and 888365, respectively.
Funding
No external funding was received to perform this research work.
Author information
Authors and Affiliations
Contributions
Conceptualization: José González-Valdez; Methodology: José González-Valdez, Arisbe Silva-Núñez; Formal analysis and investigation: Arisbe Silva-Núñez, Javier Donoso-Quezada; Writing—original draft preparation: Arisbe Silva-Núñez; Writing-review and editing: Arisbe Silva-Núñez, Javier Donoso-Quezada, José González-Valdez; Funding acquisition: José González-Valdez; Supervision: José González-Valdez.
All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflicts of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Silva-Núñez, A., Donoso-Quezada, J. & González-Valdez, J. Enhanced antiproliferative activity of phycoerythrin through microencapsulation. J Appl Phycol 36, 205–215 (2024). https://doi.org/10.1007/s10811-023-03138-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-023-03138-1