Abstract
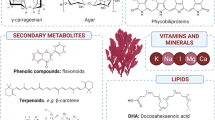
Gelidium corneum (Hudson) J.V.Lamouroux is known for its economic value and wide-scale commercial exploitation across diverse geographical regions, including the Northeast Atlantic Ocean, Mediterranean Sea, Indian Ocean, and Pacific Ocean. Until today, this economic exploitation is mainly focused on the extraction of agar, agarose production, and subsequent commercialization. Nevertheless, this red algae presents a wide range of other high-value products, such as polyphenolic compounds, phycobiliproteins, carotenoids, and mycosporine-like amino acids (MAAs), which have the potential to improve the economic viability of a biorefinery approach. Moreover, these compounds exhibit health-promoting properties and offer various commercial and industrial applications. Thus, the present review provides the state of the art of the full valorization of the red algae G. corneum. First, a description of the chemical composition of G. corneum, namely macromolecules and secondary metabolites, is done to identify the compounds that will maximize the value of this algal biomass. Then, the health benefits and biological activities of G. corneum extracts are reported, and finally, a biorefinery perspective is discussed. This review may assist industries and researchers in further developing innovative and efficient processes for utilizing G. corneum biomass to produce food, feed, and bio-based products.




Similar content being viewed by others
References
Abdala Díaz RT, Casas Arrojo V, Arrojo Agudo MA, Cárdenas C, Dobretsov S, Figueroa FL (2019) Immunomodulatory and antioxidant activities of sulfated polysaccharides from Laminaria ochroleuca, Porphyra umbilicalis, and Gelidium corneum. Mar Biotechnol 21:577–587
Alehosseini A, del Pulgar E-MG, Gómez-Mascaraque LG, Martínez-Sanz M, Fabra MJ, Sanz Y, Sarabi-Jamab M, Ghorani B, Lopez-Rubio A (2018) Unpurified Gelidium-extracted carbohydrate-rich fractions improve probiotic protection during storage. LWT 96:694–703
Álvarez-Viñas M, Flórez-Fernández N, Torres MD, Domínguez H (2019) Successful approaches for a red seaweed biorefinery. Mar Drugs 17:620
Amamou S, Sambusiti C, Monlau F, Dubreucq E, Barakat A (2018) Mechano-enzymatic deconstruction with a new enzymatic cocktail to enhance enzymatic hydrolysis and bioethanol fermentation of two macroalgae species. Molecules 23:174
Baghel RS (2023) Developments in seaweed biorefinery research: A comprehensive review. Chem Eng J 454:140177
Balina K, Romagnoli F, Blumberga D (2017) Seaweed biorefinery concept for sustainable use of marine resources. Energy Procedia 128:504–511
Baptista SL, Romaní A, Oliveira C, Ferreira S, Rocha CMR, Domingues L (2021) Galactose to tagatose isomerization by the l-arabinose isomerase from Bacillus subtilis: A biorefinery approach for Gelidium sesquipedale valorisation. LWT 151:112199
Bondar M, Pedro F, Oliveira MC, da Fonseca MMR, Cesário MT (2022) Red algae industrial residues as a sustainable carbon platform for the co-production of poly-3-hydroxybutyrate and gluconic acid by Halomonas boliviensis. Front Bioeng Biotechnol 10:934432
Boujaber N, Oumaskour K, Hassou N, Lakhdar F, Assobhei O, Etahiri S (2016) Antimicrobial effect of two marine algae Gelidium sesquipedale and Laminaria ochroleuca collected from the coast of El Jadida-Morocco. J Bio Innov 5:16–23
Boujaber N, Oumaskour K, Lakhdar F, Assobhei O, Etahiri S (2017) Potential targets for anti-inflammatory and anticancer activities of marine algae Gelidium sesquipedale and Laminaria ochroleuca. Int J Adv Res 5:2302–2309
Carmona R, Santos R (2006) Is there an ecophysiological explanation for the gametophyte–tetrasporophyte ratio in Gelidium sesquipedale (Rhodophyta)? J Phycol 42:259–269
Carpena M, Caleja C, Pereira E, Pereira C, Ćirić A, Soković M, Soria-Lopez A, Fraga-Corral M, Simal-Gandara J, Ferreira ICFR, Barros L, Prieto MA (2021) Red seaweeds as a source of nutrients and bioactive compounds: Optimization of the extraction. Chemosensors 9:132
Castejón N, Parailloux M, Izdebska A, Lobinski R, Fernandes SCM (2021) Valorization of the red algae Gelidium sesquipedale by extracting a broad spectrum of minor compounds using green approaches. Mar Drugs 19:574
Cavaco M, Duarte A, Freitas MV, Afonso C, Bernardino S, Pereira L, Martins M, Mouga T (2021) Seasonal nutritional profile of Gelidium corneum (Rhodophyta, Gelidiaceae) from the center of Portugal. Foods 10:2394
Chen YW, Lee HV, Juan JC, Phang S-M (2016) Production of new cellulose nanomaterial from red algae marine biomass Gelidium elegans. Carbohydr Polym 151:1210–1219
Cosenza VA, Navarro DA, Ponce NMA, Stortz CA (2017) Seaweed polysaccharides: Structure and applications. In: Goyanes SN, D’Accorso NB (eds) Industrial applications of renewable biomass products: Past, present and future. Springer, Cham, pp 75–116
de la Coba F, Aguilera J, Figueroa FL, de Gálvez MV, Herrera E (2009) Antioxidant activity of mycosporine-like amino acids isolated from three red macroalgae and one marine lichen. J Appl Phycol 21:161–169
de Oliveira JPV, Bruni GP, Fabra MJ, da Rosa ZE, López-Rubio A, Martínez-Sanz M (2019) Development of food packaging bioactive aerogels through the valorization of Gelidium sesquipedale seaweed. Food Hydrocoll 89:337–350
Eggert A, Karsten U (2010) Low molecular weight carbohydrates in red algae–an ecophysiological and biochemical perspective. In: Seckbach J, Chapman D (eds) Red algae in the genomic age. Springer, Dordrecht, pp 443–456
El Wahidi M, El Amraoui B, El Amraoui M, Bamhaoud T (2015) Screening of antimicrobial activity of macroalgae extracts from the Moroccan Atlantic coast. Ann Pharm Franç 73:190–196
Errati H, Bencheqroun SK, Aboutayeb R, Abail Z, Lebbar S, Dari K, Hilali L (2022) Assessment of the red seaweed Gelidium sesquipedale by-products as an organic fertilizer and soil amendment. Sustainability 14:14217
FAO Fishery and Aquaculture Statistics (2023) Global capture production 1950–2020 (FishStatJ). https://www.fao.org/fishery/en/statistics/software/fishstatj. Accessed 07 July 2023.
Faraj A, Lebbar T, Debry G, Najim L (1987) Protein and amino acids analysis during alimentary-agar extraction from Gelidium sesquipedale. Hydrobiologia 151:513–522
Fernandes SCM, Alonso-Varona A, Palomares T, Zubillaga V, Labidi J, Bulone V (2015) Exploiting mycosporines as natural molecular sunscreens for the fabrication of UV-absorbing green materials. ACS Appl Mat Interf 7:16558–16564
Fernandes SCM, Freire CSR, Silvestre AJD, Neto CP, Gandini A, Desbriéres J, Blanc S, Ferreira RAS, Carlos LD (2009) A study of the distribution of chitosan onto and within a paper sheet using a fluorescent chitosan derivative. Carbohydr Polym 78:760–766
Fernandes SCM, Freire CSR, Silvestre AJD, Pascoal Neto C, Gandini A, Berglund LA, Salmén L (2010) Transparent chitosan films reinforced with a high content of nanofibrillated cellulose. Carbohydr Polym 81:394–401
Fleurence J, Morançais M, Dumay J (2018) 9 - Seaweed proteins. In: Yada RY (ed) Proteins in food processing, 2nd edn. Woodhead Publishing, Chichester, pp 245–262
Freitas MV, Pacheco D, Cotas J, Mouga T, Afonso C, Pereira L (2022) Red seaweed pigments from a biotechnological perspective. Phycology 2:1–29
Generalić Mekinić I, Šimat V, Rathod NB, Hamed I, Čagalj M (2023) Algal carotenoids: Chemistry, sources, and application. Foods 12:2768
Givernaud T, Sqali N, Barbaroux O, Orbi A, Semmaoui Y, Rezzoum NE, Mouradi A, Kaas R (2005) Mapping and biomass estimation for a harvested population of Gelidium sesquipedale (Rhodophyta, Gelidiales) along the Atlantic coast of Morocco. Phycologia 44:66–71
Gomes-Dias JS, Pereira SG, Teixeira JA, Rocha CMR (2022) Hydrothermal treatments – A quick and efficient alternative for agar extraction from Gelidium sesquipedale. Food Hydrocoll 132:107898
Gomes-Dias JS, Romaní A, Teixeira JA, Rocha CMR (2020) Valorization of seaweed carbohydrates: Autohydrolysis as a selective and sustainable pretreatment. ACS Sust Chem Eng 8:17143–17153
Gómez Barrio LP, Cabral EM, Zhao M, Álvarez García C, Senthamaraikannan R, Padamati RB, Tiwari U, Curtin JF, Tiwari BK (2022) Comparison study of an optimized ultrasound-based method versus an optimized conventional method for agar extraction, and protein co-extraction, from Gelidium sesquipedale. Foods 11(6):805. https://doi.org/10.3390/foods11060805
Gomez LP, Alvarez C, Zhao M, Tiwari U, Curtin J, Garcia-Vaquero M, Tiwari BK (2020) Innovative processing strategies and technologies to obtain hydrocolloids from macroalgae for food applications. Carbohydr Polym 248:116784
Grina F, Ullah Z, Kaplaner E, Moujahid A, Eddoha R, Nasser B, Terzioğlu P, Yilmaz MA, Ertaş A, Öztürk M, Essamadi A (2020) In vitro enzyme inhibitory properties, antioxidant activities, and phytochemical fingerprints of five Moroccan seaweeds. S Afr J Bot 128:152–160
Grozdanić N, Stanojković TP, Kljajić Z, Etahiri S, Assobhei O, Konić-Ristić A, Srdić-Rajić T, Kardum NĐ, Backović S (2012) In Vitro evaluation of antioxidant and antitumoral activities of marine algae gelidium sesquipedale and fucus spiralis. European Journal of Cancer Elsevier Sci Ltd, Oxford 48:S26–S26. https://doi.org/10.1016/S0959-8049(12)70809-2
Guerrero P, Etxabide A, Leceta I, Peñalba M, de la Caba K (2014) Extraction of agar from Gelidium sesquipedale (Rhodophyta) and surface characterization of agar based films. Carbohydr Polym 99:491–498
Guiry, MD, Guiry GM (2017) AlgaeBase. World-wide electronic publication, National University of Ireland, Galway (2017) https://www.algaebase.org. Accessed 07 July 2023.
Jung KA, Lim SR, Kim Y, Park JM (2013) Potentials of macroalgae as feedstocks for biorefinery. Bioresour Technol 135:182–190
Kannaujiya VK, Kumar D, Pathak J, Sinha RP (2019) Chapter 10 - phycobiliproteins and their commercial significance. In: Mishra AK, Tiwari DN DN, Rai AN (eds) Cyanobacteria. Academic Press, London, pp 207–216. https://doi.org/10.1016/B978-0-12-814667-5.00010-6
Karsten U, Sawal T, Dieter H, Bischof K, Lopez Figueroa F, Flores-Moya A, Wiencke C (1998) An inventory of UV-absorbing mycosporine-like amino acids in macroalgae from polar to warm-temperate regions. Bot Mar 41:443–453
Koçi R, Dupuy F, Lebbar S, Gloaguen V, Faugeron Girard C (2022) A new promising plant defense stimulator derived from a by-product of agar extraction from Gelidium sesquipedale. Horticulturae 8:958
Kovaleski G, Kholany M, Dias LMS, Correia SFH, Ferreira RAS, Coutinho JAP, Ventura SPM (2022) Extraction and purification of phycobiliproteins from algae and their applications. Front Chem 10:1065355
Kumari P, Bijo AJ, Mantri VA, Reddy CRK, Jha B (2013) Fatty acid profiling of tropical marine macroalgae: An analysis from chemotaxonomic and nutritional perspectives. Phytochemistry 86:44–56
Lebbar S, Fanuel M, Le Gall S, Falourd X, Ropartz D, Bressollier P, Gloaguen V, Faugeron-Girard C (2018) Agar extraction by-products from Gelidium sesquipedale as a source of glycerol-galactosides. Molecules 23:3364
Lee WK, Lim YY, Leow AT, Namasivayam P, Ong Abdullah J, Ho CL (2017) Biosynthesis of agar in red seaweeds: A review. Carbohydr Polym 164:23–30
Li W, Su HN, Pu Y, Chen J, Liu LN, Liu Q, Qin S (2019) Phycobiliproteins: Molecular structure, production, applications, and prospects. Biotech Adv 37:340–353
Li Y, Zhao M, Gomez LP, Senthamaraikannan R, Padamati RB, O’Donnell CP, Tiwari BK (2021) Investigation of enzyme-assisted methods combined with ultrasonication under a controlled alkali pretreatment for agar extraction from Gelidium sesquipedale. Food Hydrocoll 120:106905
Martinez-Garcia M, van der Maarel MJEC (2016) Floridoside production by the red microalga Galdieria sulphuraria under different conditions of growth and osmotic stress. AMB Express 6:71
Martínez-Sanz M, Cebrián-Lloret V, Mazarro-Ruiz J, López-Rubio A (2020) Improved performance of less purified cellulosic films obtained from agar waste biomass. Carbohydr Polym 233:115887
Martínez-Sanz M, Gomez-Barrio LP, Zhao M, Tiwari B, Knutsen SH, Ballance S, Zobel HK, Nilsson AE, Krewer C, Östergren K, López-Rubio A (2021) Alternative protocols for the production of more sustainable agar-based extracts from Gelidium sesquipedale. Algal Res 55:102254
Martínez-Sanz M, Gómez-Mascaraque LG, Ballester AR, Martínez-Abad A, Brodkorb A, López-Rubio A (2019) Production of unpurified agar-based extracts from red seaweed Gelidium sesquipedale by means of simplified extraction protocols. Algal Res 38:101420
Martins NCT, Freire CSR, Pinto RJB, Fernandes SCM, Pascoal Neto C, Silvestre AJD, Causio J, Baldi G, Sadocco P, Trindade T (2012) Electrostatic assembly of Ag nanoparticles onto nanofibrillated cellulose for antibacterial paper products. Cellulose 19:1425–1436
Matias M, Pinteus S, Martins A, Silva J, Alves C, Mouga T, Gaspar H, Pedrosa R (2022) Gelidiales are not just agar-revealing the antimicrobial potential of Gelidium corneum for skin disorders. Antibiotics 11:481
Matos J, Gomes A, Cardoso C, Afonso C, Campos AM, Gomes R, Falé P, Delgado I, Coelho I, Castanheira I, Bandarra NM (2020) Commercial red seaweed in Portugal (Gelidium sesquipedale and Pterocladiella capillacea, Florideophyceae): Going beyond a single-purpose product approach by valorizing bioactivity. Thalassas 36:213–224
McReynolds C, Adrien A, Silvestre de Ferron A, Boussetta N, Grimi N, Pecastaing L, Fernandes SCM (2023) Extraction of mycosporine-like amino acids and proteins from the agarophyte Gelidium corneum using pulsed power techniques. Foods 12:1473
Meinita MDN, Harwanto D, Amron HMA, Jeong G-T, Moon IS, Choi J-S (2023) A concise review of the potential utilization based on bioactivity and pharmacological properties of the genus Gelidium (Gelidiales, Rhodophyta). J Appl Phycol 35:1499–1523
Metidji H, Dob T, Toumi ML, Krimat S, Ksouri A, Nouasri A (2015) In vitro screening of secondary metabolites and evaluation of antioxidant, antimicrobial and cytotoxic properties of Gelidium sesquipedale Thuret et Bornet red seaweed from Algeria. J Mater Environ Sci 6:3184–3196
Mittal R, Tavanandi HA, Mantri VA, Raghavarao K (2017) Ultrasound assisted methods for enhanced extraction of phycobiliproteins from marine macro-algae, Gelidium pusillum (Rhodophyta). Ultrason Sonochem 38:92–103
Mouga T, Fernandes IB (2022) The red seaweed Giant Gelidium (Gelidium corneum) for new bio-based materials in a circular economy framework. Earth 3:788–813
Mouradi-Givernaud A, Amina Hassani L, Givernaud T, Lemoine Y, Benharbet O (1999) Biology and agar composition of Gelidium sesquipedale harvested along the Atlantic coast of Morocco. Hydrobiologia 398:391–395
Nil S, Ali-Mehidi S, Zellal A, Abi-Ayad S (2016) Effects of season on the yield and quality of agar from Gelidium sesquipedale (Rhodophyta) from Mostaganem, Algeria. Afr J Biotech 15:350–355
Oumaskour K, Boujaber N, Etahiri S, Assobhei O (2013) Anti-inflammatory and antimicrobial activities of twenty-three marine red algae from the coast of Sidi Bouzid (El Jadida-Morocco). Int J Pharm 5:145–149
Pagels F, Guedes AC, Amaro HM, Kijjoa A, Vasconcelos V (2019) Phycobiliproteins from cyanobacteria: Chemistry and biotechnological applications. Biotechnol Adv 37:422–443
Parailloux M, Godin S, Fernandes SCM, Lobinski R (2020) Untargeted analysis for mycosporines and mycosporine-like amino acids by hydrophilic interaction liquid chromatography (HILIC)—electrospray orbitrap MS2/MS3. Antioxidants 9:1185
Pereira-Pacheco F, Robledo D, Rodríguez-Carvajal L, Freile-Pelegrín Y (2007) Optimization of native agar extraction from Hydropuntia cornea from Yucatán, México. Bioresour Technol 98:1278–1284
Pereira L (2016) Edible Seaweeds of the World. CRC Press, Boca Raton
Pérez MJ, Falqué E, Domínguez H (2016) Antimicrobial action of compounds from marine seaweed. Mar Drugs 14:52
Quintano E, Ganzedo U, Díez I, Figueroa FL, Gorostiaga JM (2013) Solar radiation (PAR and UVA) and water temperature in relation to biochemical performance of Gelidium corneum (Gelidiales, Rhodophyta) in subtidal bottoms off the Basque coast. J Sea Res 83:47–55
Rajkumar R, Yaakob Z, Takriff MS (2013) Potential of the micro and macro algae for biofuel production: a brief review. BioResources 9:1606–1633
Rhimou B, Hassane R, José M, Nathalie B (2010) The antibacterial potential of the seaweeds (Rhodophyceae) of the Strait of Gibraltar and the Mediterranean coast of Morocco. Afr J Biotechnol 9:6365–6372
Ryu B, Li Y-X, Kang K-H, Kim S-K, Kim DG (2015) Floridoside from Laurencia undulata promotes osteogenic differentiation in murine bone marrow mesenchymal cells. J Funct Foods 19:505–511
Samri N, Hsaine L, Elkafhi S, Khlifi S, Etahiri S (2020) Antimicrobial properties of seven brown algae harvested from the coast of Sidi Bouzid (El Jadida-Morocco). Int J Pharm Pharmaceut Sci 12:57-62
Santos R, Melo RA (2018) Global shortage of technical agars: back to basics (resource management). J Appl Phycol 30:2463–2473
Silva J, Santos R (2003) Comparative ecophysiology of Gelidium sesquipedale (Rhodophyta). Erect fronds and prostrate system. In: Chapman ARO, Anderson RJ, Vreeland VJ, Davison IR (eds) Proceedings of the 17th International Seaweed Symposium. Oxford University Press, Oxford, pp 417–424. https://www.researchgate.net/profile/Joao-Silva-254/publication/286044357_Comparative_ecophysiology_of_Gelidium_sesquipedale_Rhodophyta_erect_fronds_and_prostrate_system/links/56659b8e08ae4931cd62447d/Comparativeecophysiology-of-Gelidium-sesquipedale-Rhodophyta-erect-fronds-and-prostrate-system.pdf
Sukwong P, Sunwoo IY, Nguyen TH, Jeong G-T, Kim S-K (2019) R-phycoerythrin, R-phycocyanin and ABE production from Gelidium amansii by Clostridium acetobutylicum. Process Biochem 81:139–147
Torres MD, Flórez-Fernández N, Domínguez H (2019) Integral utilization of red seaweed for bioactive production. Mar Drugs 17(6):314. https://doi.org/10.3390/md17060314
Trigueros E, Sanz M, Filipigh A, Beltrán S, Riaño P (2021a) Enzymatic hydrolysis of the industrial solid residue of red seaweed after agar extraction: Extracts characterization and modelling. Food Bioprod Process 126:356–366
Trigueros E, Sanz MT, Alonso-Riaño P, Beltrán S, Ramos C, Melgosa R (2021b) Recovery of the protein fraction with high antioxidant activity from red seaweed industrial solid residue after agar extraction by subcritical water treatment. J Appl Phycol 33:1181–1194
Tůma S, Izaguirre JK, Bondar M, Marques MM, Fernandes P, da Fonseca MMR, Cesário MT (2020) Upgrading end-of-line residues of the red seaweed Gelidium sesquipedale to polyhydroxyalkanoates using Halomonas boliviensis. Biotech Rep 27:e00491
Usov AI (2011) Polysaccharides of the red algae. Adv Carbohydr Chem Biochem 65:115–217
Vo TS, Ngo DH, Kim SK (2012) Potential targets for anti-inflammatory and anti-allergic activities of marine algae: an overview. Inflamm Allergy Drug Targets 11:90–101
Xu P, Tan H, Jin W, Li Y, Santhoshkumar C, Li P, Liu W (2018) Antioxidative and antimicrobial activities of intertidal seaweeds and possible effects of abiotic factors on these bioactivities. J Oceanol Limnol 36:2243–2256
Yang S, Chen M-F, Ryu B, Chen J, Xiao Z, Hong P, Sun S, Wang D, Qian Z-J, Zhou C (2020) The protective effect of the polysaccharide precursor, D-isofloridoside, from Laurencia undulata on alcohol-induced hepatotoxicity in HepG2 cells. Molecules 25:1024
Yun EJ, Kim HT, Cho KM, Yu S, Kim S, Choi I-G, Kim KH (2016) Pretreatment and saccharification of red macroalgae to produce fermentable sugars. Bioresour Technol 199:311–318
Acknowledgements
The authors would like to thank the Local Fishery Committee, the CIDPMEM 64-40, for their support.
Funding
This work was carried under the framework of E2S UPPA Partnership Chair MANTA (Marine Materials) funded by the ‘Investissements d’Avenir’ French program managed by ANR, grant number #ANR-16-IDEX-0002; by the Région Nouvelle-Aquitaine and by the Communautéd’Agglomération du Pays Basque (France); and by the FLAG “Côte Basque-Sud Landes” through the community-led local development (CLDD: ref number #PFEA621219CR0720024) program funded by the European Maritime and Fisheries Fund (EMFF).
Author information
Authors and Affiliations
Contributions
Conceptualization: Natalia Castejón and Susana C. M. Fernandes; Writing—original draft preparation: Natalia Castejón; Writing—review and editing: Natalia Castejón, Amandine Adrien, Lea Spitzer and Susana C. M. Fernandes; Funding acquisition: Susana C. M. Fernandes.
All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; nor in the writing of the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Castejón, N., Adrien, A., Spitzer, L. et al. Unlocking the full potential of the red seaweed Gelidium corneum: Beyond its use as an agar source. J Appl Phycol 36, 291–311 (2024). https://doi.org/10.1007/s10811-023-03115-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-023-03115-8