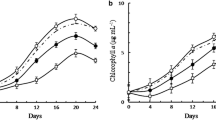
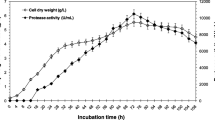
Abstract
This paper describes the characterization of proteases in Microcystis aeruginosa PCC7806 cells being inhibited by a metabolite produced by another Microcystis strain, Microcystis Ku1. With casein and oligopeptide substrates and specific inhibitors we detected activity similar to bacterial serine endoproteases. Substrate SDS-polyacrylamide gel electrophoresis revealed the presence of nine bands of proteases (ca. 35∼125 kDa). The cyanobacterial enzymes were insensitive to endogenous trypsin-inhibitory metabolites. Microcystis Ku1 produced a metabolite, tentatively characterized as microviridin, inhibiting both cyanobacterial proteases and trypsin at an estimated IC50 of, respectively, 2.2 and 9.0 μg mL−1. On activity gels, inhibitors specific to animal trypsin and elastase and the putative microviridin led to an inactivation of the proteases associated with the 88 and 110 kDa bands. We hypothesize that in Microcystis populations there is a “cross-talk” between the inhibitors and the proteases, and only the colonies of identical chemotypes can possibly aggregate to form blooms.






Similar content being viewed by others
References
Agrawal MK, Bagchi D, Bagchi SN (2001) Acute inhibition of protease and suppression of growth in zooplankter, Moina macrocopa, by Microcystis blooms collected in Central India. Hydrobiologia 464:37–44
Agrawal MK, Zittt A, Bagchi D, Weckesser J, Bagchi SN, von Elert E (2005) Characterization of proteases in gut of Daphnia magna and their inhibition by Microcystis aeruginosa PCC7806. Environ Toxicol 20:314–322
Babica P, Bláha L, Maršálek B (2006) Exploring the natural role of microcystins- a review of effects on photoautotrophic organisms. J Phycol 42:9–20
Bharadwaj M, Bharadwaj D, Hati RN (1996) Characterization of a membrane protease from rat submaxillary-gland mitochondria that possesses thrombin-like activity. Biochem J 313:193–200
Bister B, Keller S, Baumann HI, Nicholson G, Weist S, Jung G, Süssmuth R, Jüttner F (2004) Cyanopeptolin 963A, a chymotrypsin inhibitor of Microcystis PCC 7806. J Nat Prod 67:1755–1757
Blom JF, Bister B, Bischoff D, Nicholoson G, Jung G, Süssmuth RD, Jüttner F (2003) Oscillapeptin J, a new grazer toxin of the freshwater cyanobacterium Planktothrix rubescens. J Nat Prod 66:431–434
Czarnecki O, Henning M, Lippert I, Welker M (2006) Identification of peptide metabolites of Microcystis (cyanobacteria) that inhibit-trypsin like activity in planktonic herbivorous Daphnia (Cladocera). Environ Microbiol 8:77–87
Dagnino D, de Abreu Meireles D, de Aquino Almeida JC (2006) Growth of nutrient-replete Microcystis PCC 7806 cultures is inhibited by an extracellular signal produced by chlorotic cultures. Environ Microbiol 8:30–36
Díaz M, Moyano FJ, García-Carreño FL, Alarcon FJ, Sarasquete MC (1997) Substrate-SDS-PAGE determination of protease activity through larval development in sea bream. Aquacultural Int 5:461–471
Dittmann E, Erhard M, Kaebernick M, Scheler C, Neilan BA, von Döhren H, Börner T (2001) Altered expression of two light-dependent genes in microcystin lacking mutant of Microcystis aeruginosa PCC 7806. Microbiology 147:3113–3119
Erlänger BF, Kokowski N, Cohen W (1961) The preparation and properties of two new chromogenic substrates of trypsin. Arch Biochem Biophys 95:271–278
Fastner J, Erhard M, von Döhren H (2001) Determination of oligopeptide diversity with a natural population of Microcystis spp. (cyanobacteria) by typing single colonies by matrix-assisted laser desorption ionization-time of flight mass spectrometry. Appl Environ Microbiol 67:5069–5076
Harada K, Nakano T, Makoto M (2004) Comprehensive analysis system using liquid chromatography-mass spectrophotometry for the biosynthetic study of peptides produced by cyanobacteria. J Chromatogr A 1033:107–113
Huesgen PF, Schuhmann H, Adamska I (2005) The family of Deg proteases in cyanobacteria and chloroplasts of higher plants. Physiol Plant 123:413–420
Jakobi C, Rinehart KL, Neuber R, Mez K, Weckesser J (1996) Cyanopeptolin SS, a disulphated depsipeptide from a water bloom: structural elucidation and biological activities. Phycologia 35:111–116
MacKintosh C, Beattie KA, Klumpp S, Cohen P, Codd GA (1990) Cyanobacterial microcystin-LR is a potent and specific inhibitor of protein phosphatases 1 and 2A from both mammals and higher plants. FEBS Lett 264:187–192
Martin C, Oberer L, Ino T, König WA, Busch M, Weckesser J (1993) Cyanopeptolins, new depsipeptides from the cyanobacterium Microcystis Ku1 PCC 7806. J Antibiot 46:1550–1556
Martin-Luna B, Sevilla E, Hernandez JA, Bes MT, Fillat MF, Teleato ML (2006) Fur from Microcystis aeruginosa binds in vitro promoter regions of the microcystin biosynthesis gene cluster. Phytochemistry 67:876–881
Martinez J, Azam F (1993) Aminopeptidase activity in marine Chroococcoid cyanobacteria. Appl Environ Microbiol 59:3701–3707
Matern U, Oberer L, Falchetto RA, Erhard M, König WA, Herdman M, Weckesser J (2001) Scyptolin A and B, cyclic depsipeptides from axenic cultures of Scytonema hofmanni PCC 7110. Phytochemistry 58:1087–1095
Michaud D (1998) Gel electrophoresis of proteolytic enzymes. Anal Chim Acta 392:173–185
Nanni B, Balestreri E, Dainese E, Cozzani I, Felicioli R (2001) Characterization of a specific phycocyanin-hydrolysing protease from Spirulina platensis. Microbiol Res 156:259–266
Peréz-Lloréns JL, Benítez E, Vergara JJ, Berges JA (2003) Characterization of proteolytic enzyme activities in macroalgae. Eur J Phycol 38:55–64
Radau G, Stürzebecher J (2002) Cyanopeptide analogues: new lead structures for the design and synthesis of new thrombin inhibitors. Pharmazie 57:729–732
Rao MB, Tanksale AM, Ghatge MS, Deshpandey VV (1998) Molecular and biotechnological aspects of microbial proteases. Microbiol Mol Biol Rev 62:597–635
Richter R, Hejazi M, Kraft R, Ziegler K, Lockau W (1999) Cyanophycinase, a peptidase degrading the cyanobacterial reserve material multi-L-arginyl-poly-L-aspartic acid (cyanophycin). Eur J Biochem 263:163–169
Rohrlack T, Christoffersen K, Hansen PE, Zhang W, Czaenecki O, Henning M, Fastner J, Erhard M, Neilan BA, Kabernick M (2003) Isolation, characterization and quantitative analysis of microviridin J, a new Microcystis metabolite toxic to Daphnia. J Chem Ecol 29:1757–1770
Rouhiainen L, Paulin L, Suomalainen S, Hyytiäinen H, Buikema W, Haselkorn R, Sivonen K (2000) Genes encoding synthetases of cyclic depsipeptides, anabaenopeptilides, in Anabaena strain 90. Mol Microbiol 37:156–167
Schagger J, von Jagow V (1987) Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of protein in the range from 1 to 100 kDa. Anal Biochem 166:368–379
Schatz D, Keren Y, Vardi A, Sukenik A, Carmeli S, Börner T, Dittmann E, Kaplan A (2007) Towards clarification of biological role of microcystin, a family of cyanobacterial toxins. Environ Microbiol 9:965–970
Schwarz WH, Zverlov VV (2006) Protease inhibitors in bacteria: an emerging concept for the regulation of bacterial protein complex? Mol Microbiol DOI 10.1111/j. 1365–2958.2006.05181.x
Schwert GW, Takenaka Y (1955) A spectrophotometric determination of trypsin and chymotrypsin. Biochim Biophys Acta 16:570–575
Shi L, Carmichael WW, Kennelly PJ (1999) Cyanobacterial PPP family protein phosphatases possess multifunctional capabilities and are resistant to microcystin-LR. J Biol Chem 274:10039–10046
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provencano MD, Fujimoto EK, Goecke NM, Oslon BJ, Klen DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150:368–379
Welker M, von Döhren H (2006) Cyanobacterial peptides- nature’s own combinatorial biosynthesis. FEMS Microbiol Rev 30:530–563
Welker M, Brunke M, Preussel K, Lippert I, von Döhren H (2004) Diversity and distribution of Microcystis (cyanobacteria) oligopeptide chemotypes from natural community studied by single-colony mass spectrometry. Microbiology 150:1785–1796
Yamaki H, Sitachitta N, Sano T, Kaya K (2005) Two new chymotrypsin inhibitors isolated from the cyanobacterium Microcystis aeruginosa NIES-88. J Nat Prod 68:14–18
Zuther E, Schubert H, Hagemann M (1998) Mutation of a gene encoding a putative glycoprotease leads to a reduced salt tolerance, altered pigmentation, and cyanophycin accumulation in the cyanobacterium Synechocystis sp. strain PCC 6803. J Bacteriol 180:1715–1722
Acknowledgements
The authors would like to thank the Head of the Department of Biological Sciences, Rani Durgavati University, Jabalpur for the use of laboratory facilities, Dr. K.P. Madhusudanan, SAIF, Central Drug Research Institute, Lucknow (India) for helping with spectral analyses, and Prof. J. Weckesser, Institut für Biologie, Mikrobiologie, Albert-Ludwigs Universität, Freiburg (Germany), for providing dried Microcystis aeruginosa PCC7806 cells, The financial support from the Department of Biotechnology, Govt. of India, New Delhi (project no. BT/PR3717/BCE/08/259/2003) is also gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghosh, S.K., Bagchi, D. & Bagchi, S.N. Proteolytic activity in Microcystis aeruginosa PCC7806 is inhibited by a trypsin-inhibitory cyanobacterial peptide with a partial structure of microviridin. J Appl Phycol 20, 1045–1052 (2008). https://doi.org/10.1007/s10811-007-9304-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-007-9304-2