Abstract
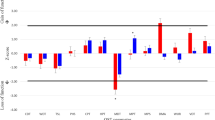
Altered sensory perception has been found in patients with autism spectrum disorders (ASD) and might be related to aberrant sensory perception thresholds. We used the well-established, standardized Quantitative sensory testing (QST) protocol of the German Research Network on Neuropathic Pain to investigate 13 somatosensory parameters including thermal and tactile detection and pain thresholds in 13 ASD adults and 13 matched healthy controls with normal IQ values. There were no group differences between somatosensory detection and pain thresholds. Two ASD patients showed paradoxical heat sensations and another two ASD subjects presented dynamic mechanical allodynia; somatosensory features that were absent in controls. These findings suggest that central mechanisms during complex stimulus integration rather than peripheral dysfunctions probably determine somatosensory alterations in ASD.


Similar content being viewed by others
References
American Psychiatric Association, A. (2000). Diagnostic and statistical manual of mental disorders Dsm-IV-Tr (Text Revision) (4th ed.). Washington, DC: American Psychiatric Press Inc.
APA (2013). Diagnostic and statistical manual of mental disorders (DSM-5(r)).
Baron, R., & Saguer, M. (1995). Mechanical allodynia in postherpetic neuralgia: evidence for central mechanisms depending on nociceptive C-fiber degeneration. Neurology, 45(12 Suppl 8), S63–65.
Baron-Cohen, S., Ashwin, E., Ashwin, C., Tavassoli, T., & Chakrabarti, B. (2009). Talent in autism: hyper-systemizing, hyper-attention to detail and sensory hypersensitivity. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 364(1522), 1377–1383. doi:10.1098/rstb.2008.0337.
Baron-Cohen, S., Hoekstra, R. A., Knickmeyer, R., & Wheelwright, S. (2006). The autism-spectrum quotient (AQ)–adolescent version. Journal of Autism and Developmental Disorders, 36(3), 343–350. doi:10.1007/s10803-006-0073-6.
Baron-Cohen, S., Richler, J., Bisarya, D., Gurunathan, N., & Wheelwright, S. (2003). The systemizing quotient: an investigation of adults with Asperger syndrome or high-functioning autism, and normal sex differences. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 358(1430), 361–374. doi:10.1098/rstb.2002.1206.
Baron-Cohen, S., & Wheelwright, S. (2004). The empathy quotient: an investigation of adults with Asperger syndrome or high functioning autism, and normal sex differences. Journal of Autism and Developmental Disorders, 34(2), 163–175.
Baum, S. H., Stevenson, R. A., & Wallace, M. T. (2015). Behavioral, perceptual, and neural alterations in sensory and multisensory function in autism spectrum disorder. Progress in Neurobiology, 134, 140–160. doi:10.1016/j.pneurobio.2015.09.007.
Beck, A. T., Ward, C. H., Mendelson, M., Mock, J., & Erbaugh, J. (1961). An inventory for measuring depression. Archives of General Psychiatry, 4, 561–571.
Bertone, A., Mottron, L., Jelenic, P., & Faubert, J. (2003). Motion perception in autism: A “complex” issue. Journal of Cognitive Neuroscience, 15(2), 218–225. doi:10.1162/089892903321208150.
Bird, G., Silani, G., Brindley, R., White, S., Frith, U., & Singer, T. (2010). Empathic brain responses in insula are modulated by levels of alexithymia but not autism. Brain: A Journal of Neurology, 133(Pt 5), 1515–1525. doi:10.1093/brain/awq060.
Blakemore, S. J., Tavassoli, T., Calo, S., Thomas, R. M., Catmur, C., Frith, U., et al. (2006). Tactile sensitivity in asperger syndrome. Brain and Cognition, 61(1), 5–13. doi:10.1016/j.bandc.2005.12.013.
Cascio, C., McGlone, F., Folger, S., Tannan, V., Baranek, G., Pelphrey, K. A., et al. (2008). Tactile perception in adults with autism: A multidimensional psychophysical study. Journal of Autism and Developmental Disorders, 38(1), 127–137. doi:10.1007/s10803-007-0370-8.
Cesaroni, L., & Garber, M. (1991). Exploring the experience of autism through firsthand accounts. Journal of Autism and Developmental Disorders, 21(3), 303–313.
Craig, A. D., & Bushnell, M. C. (1994). The thermal grill illusion: Unmasking the burn of cold pain. Science, 265(5169), 252–255.
David, N., Schneider, T. R., Peiker, I., Al-Jawahiri, R., Engel, A. K., & Milne, E. (2016). Variability of cortical oscillation patterns: A possible endophenotype in autism spectrum disorders? Neuroscience and Biobehavioral Reviews, 71, 590–600. doi:10.1016/j.neubiorev.2016.09.031.
Davis, K. D., Pope, G. E., Crawley, A. P., & Mikulis, D. J. (2004). Perceptual illusion of “paradoxical heat” engages the insular cortex. Journal of Neurophysiology, 92(2), 1248–1251. doi:10.1152/jn.00084.2004.
Di Martino, A., Ross, K., Uddin, L. Q., Sklar, A. B., Castellanos, F. X., & Milham, M. P. (2009). Functional brain correlates of social and nonsocial processes in autism spectrum disorders: an activation likelihood estimation meta-analysis. Biological Psychiatry, 65(1), 63–74. doi:10.1016/j.biopsych.2008.09.022.
Duerden, E. G., Taylor, M. J., Lee, M., McGrath, P. A., Davis, K. D., & Roberts, W. (2015). Decreased sensitivity to thermal stimuli in adolescents with autism spectrum disorder: Relation to symptomatology and cognitive ability. The Journal of Pain: Official Journal of the American Pain Society. doi:10.1016/j.jpain.2015.02.001.
Elwin, M., Ek, L., Schroder, A., & Kjellin, L. (2012). Autobiographical accounts of sensing in Asperger syndrome and high-functioning autism. Archives of Psychiatric Nursing, 26(5), 420–429. doi:10.1016/j.apnu.2011.10.003.
Fan, Y. T., Chen, C., Chen, S. C., Decety, J., & Cheng, Y. (2014). Empathic arousal and social understanding in individuals with autism: Evidence from fMRI and ERP measurements. Social Cognitive and Affective Neuroscience, 9(8), 1203–1213. doi:10.1093/scan/nst101.
Gierthmühlen, J., & Baron, R. (2013). Vom Symptom zur Therapie. In R. Baron, W. Koppert, M. Strumpf & A. Willweber-Strumpf (Eds.), Praktische Schmerzmedizin - Interdisziplinäre Diagnostik - Multimodale Therapie (Vol. 3, pp. 67–79). Berlin Heidelberg: Springer-Verlag Berlin Heidelberg.
Grandin, T. (1995). Thinking in pictures; and other reports from my life with autism. New York: Double Day.
Grone, E., Crispin, A., Fleckenstein, J., Irnich, D., Treede, R. D., & Lang, P. M. (2012). Test order of quantitative sensory testing facilitates mechanical hyperalgesia in healthy volunteers. The Journal of Pain: Official Journal of the American Pain Society, 13(1), 73–80. doi:10.1016/j.jpain.2011.10.005.
Guclu, B., Tanidir, C., Mukaddes, N. M., & Unal, F. (2007). Tactile sensitivity of normal and autistic children. Somatosensory and Motor Research, 24(1–2), 21–33. doi:10.1080/08990220601179418.
Hoitsma, E., Reulen, J. P., de Baets, M., Drent, M., Spaans, F., & Faber, C. G. (2004). Small fiber neuropathy: a common and important clinical disorder. Journal of the Neurological Sciences, 227(1), 119–130. doi:10.1016/j.jns.2004.08.012.
Jensen, T. S., & Finnerup, N. B. (2014). Allodynia and hyperalgesia in neuropathic pain: clinical manifestations and mechanisms. Lancet Neurology, 13(9), 924–935. doi:10.1016/S1474-4422(14)70102-4.
Joshi, G., Wozniak, J., Petty, C., Martelon, M. K., Fried, R., Bolfek, A., et al. (2013). Psychiatric comorbidity and functioning in a clinically referred population of adults with autism spectrum disorders: a comparative study. Journal of Autism and Developmental Disorders, 43(6), 1314–1325. doi:10.1007/s10803-012-1679-5.
Kientz, M. A., & Dunn, W. (1997). A comparison of the performance of children with and without autism on the Sensory Profile. The American Journal of Occupational Therapy: Official Publication of the American Occupational Therapy Association, 51(7), 530–537.
Klauenberg, S., Maier, C., Assion, H. J., Hoffmann, A., Krumova, E. K., Magerl, W., et al. (2008). Depression and changed pain perception: Hints for a central disinhibition mechanism. Pain, 140(2), 332–343. doi:10.1016/j.pain.2008.09.003.
Lehrl, S. (2005). Mehrfachwach-Wortschatz-Intelligenztest MWT-B (5th edition ed.). Balingen: Spitta Verlag.
Magerl, W., & Klein, T. (2006). Experimental human models of neuropathic pain. In F. Cervero, & T. S. Jensen (Ed.), Handbook of clinical neurology (Vol. 81 Pain, pp. 503–516). Edinburgh: Elsevier.
Magerl, W., Krumova, E. K., Baron, R., Tolle, T., Treede, R. D., & Maier, C. (2010). Reference data for quantitative sensory testing (QST): Refined stratification for age and a novel method for statistical comparison of group data. Pain, 151(3), 598–605. doi:10.1016/j.pain.2010.07.026.
Marco, E. J., Hinkley, L. B., Hill, S. S., & Nagarajan, S. S. (2011). Sensory processing in autism: a review of neurophysiologic findings. Pediatric Research, 69(5 Pt 2), 48R–54R, doi:10.1203/PDR.0b013e3182130c54.
Milne, E. (2011). Increased intra-participant variability in children with autistic spectrum disorders: evidence from single-trial analysis of evoked EEG. Front Psychol, 2, 51. doi:10.3389/fpsyg.2011.00051.
Minshew, N., & Goldstein, G. (1998). Autism as a disorder of complex information processing. Mental Retardation and Developmental Disabilities Research Reviews, 4(2), 129–136. doi:10.1002/(SICI)1098-2779(1998)4:2<129::AID-MRDD10>3.0.CO;2-X.
Moore, D. J. (2015). Acute pain experience in individuals with autism spectrum disorders: A review. Autism: The International Journal of Research and Practice, 19(4), 387–399. doi:10.1177/1362361314527839.
Mucke, M., Cuhls, H., Radbruch, L., Baron, R., Maier, C., Tolle, T., et al. (2014). [Quantitative sensory testing]. Schmerz, 28(6), 635–646. doi:10.1007/s00482-014-1485-4.
O’Riordan, M., & Passetti, F. (2006). Discrimination in autism within different sensory modalities. Journal of Autism and Developmental Disorders, 36(5), 665–675. doi:10.1007/s10803-006-0106-1.
Puts, N. A., Wodka, E. L., Tommerdahl, M., Mostofsky, S. H., & Edden, R. A. (2014). Impaired tactile processing in children with autism spectrum disorder. Journal of Neurophysiology, 111(9), 1803–1811. doi:10.1152/jn.00890.2013.
Riquelme, I., Hatem, S. M., & Montoya, P. (2016). Abnormal pressure pain, touch sensitivity, proprioception, and manual dexterity in children with autism spectrum disorders. Neural Plasticity, 2016, 1723401. doi:10.1155/2016/1723401.
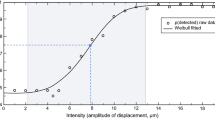
Rolke, R., Andrews, K., Magerl, W., & Treede, R. -D. (2010). Investigator’s Brochure: A standardized battery of quantitative sensory testing according to the protocol of the German Research Network on neuropathic pain (DFNS) - Version 2.1.
Rolke, R., Baron, R., Maier, C., Tolle, T. R., Treede, R. D., Beyer, A., et al. (2006). Quantitative sensory testing in the German research network on neuropathic pain (DFNS): standardized protocol and reference values. Pain, 123(3), 231–243. doi:10.1016/j.pain.2006.01.041.
Sandkuhler, J. (2009). Models and mechanisms of hyperalgesia and allodynia. Physiological Reviews, 89(2), 707–758. doi:10.1152/physrev.00025.2008.
Schneider, G., Pogatzki-Zahn, E., Marziniak, M., Stumpf, A., & Ständer, S. (2015). Cutaneous sensory function is not related to depression and anxiety in patients with chronic pruritus with dysesthetic subqualities. Acta Dermato Venereologica, 95, 289–293.
Schunke, O., Grashorn, W., Kahl, U., Schottle, D., Haggard, P., Munchau, A., et al. (2016). Quantitative sensory testing in adults with tourette syndrome. Parkinsonism & Related Disorders, 24, 132–136. doi:10.1016/j.parkreldis.2016.01.006.
Siegel, D. J., Minshew, N. J., & Goldstein, G. (1996). Wechsler IQ profiles in diagnosis of high-functioning autism. Journal of Autism and Developmental Disorders, 26(4), 389–406.
Silva, L. M., & Schalock, M. (2012). Sense and self-regulation checklist, a measure of comorbid autism symptoms: initial psychometric evidence. The American Journal of Occupational Therapy: Official Publication of the American Occupational Therapy Association, 66(2), 177–186. doi:10.5014/ajot.2012.001578.
Silva, L. M., & Schalock, M. (2013). Prevalence and significance of abnormal tactile responses in young children with autism. North American Journal of Medicine and Science, 6 (No.3), 121–127.
Susser, E., Sprecher, E., & Yarnitsky, D. (1999). Paradoxical heat sensation in healthy subjects: Peripherally conducted by A delta or C fibres? Brain, 122 (Pt 2), 239–246.
Tavassoli, T., Bellesheim, K., Tommerdahl, M., Holden, J. M., Kolevzon, A., & Buxbaum, J. D. (2015). Altered tactile processing in children with autism spectrum disorder. Autism Research. doi:10.1002/aur.1563.
Tonacci, A., Billeci, L., Tartarisco, G., Ruta, L., Muratori, F., Pioggia, G., et al. (2017). [Formula: see text] Olfaction in autism spectrum disorders: A systematic review. Child neuropsychology: A Journal on Normal and Abnormal Development in Childhood and Adolescence, 23(1), 1–25. doi:10.1080/09297049.2015.1081678.
Uddin, L. Q., & Menon, V. (2009). The anterior insula in autism: under-connected and under-examined. Neuroscience and Biobehavioral Reviews, 33(8), 1198–1203. doi:10.1016/j.neubiorev.2009.06.002.
Wahren, L. K., Torebjork, E., & Jorum, E. (1989). Central suppression of cold-induced C fibre pain by myelinated fibre input. Pain, 38(3), 313–319.
Yasuda, Y., Hashimoto, R., Nakae, A., Kang, H., Ohi, K., Yamamori, H., et al. (2016). Sensory cognitive abnormalities of pain in autism spectrum disorder: A case–control study. Ann Gen Psychiatry, 15, 8. doi:10.1186/s12991-016-0095-1.
Acknowledgments
This work was supported by the Else Kröner-Fresenius-Stiftung (Grant 2011_A37; A.M.) and by the EU (FP7-ICT-270212, ERC-2010-AdG-269716, H2020-641321; A.K.E.). We would like to thank all the patients investigated in this study for their support and commitment. Thanks to Jan Vollert (University Medical Center Bochum) and Tina Mainka (University Medical Center Hamburg-Eppendorf) for their advice.
Author Contributions
OF and WG equally participated in the conception and design of the study, performed data acquisition, analysis and interpretation, drafted and reviewed the manuscript. DS, IP, ND, AM and AKE participated in the conception and design of the study, performed data interpretation and manuscript revision for important intellectual content. KF and NW participated in the conception and design of the study, performed data analysis and interpretation as well as manuscript revision for important intellectual content. UB participated in the conception and design of the study, performed data interpretation and manuscript drafting as well as manuscript revision for important intellectual content. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there are no conflicts of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Human and Animal Research Information
Our research involved human participants.
Informed Consent
Informed consent was obtained from all individual participants included in the study. They were told to be free to withdraw from the study at any time.
Additional information
Odette Fründt and Wiebke Grashorn have contributed equally to the work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fründt, O., Grashorn, W., Schöttle, D. et al. Quantitative Sensory Testing in adults with Autism Spectrum Disorders. J Autism Dev Disord 47, 1183–1192 (2017). https://doi.org/10.1007/s10803-017-3041-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10803-017-3041-4