Abstract
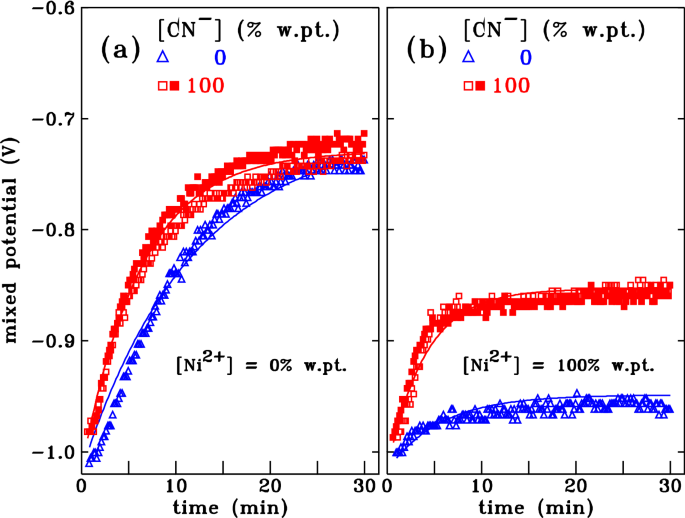
A cyanide-stabilized electroless copper plating process with nickel as a stress-regulating additive was investigated. Small amounts of nickel or cyanide increase the deposition rate, while large amounts of cyanide decrease the deposition rate. The steady-state mixed potential shifts by – 0.23 V when about 0.05 at.% nickel is co-plated with copper. Cyanide by itself does not change the mixed potential. If nickel is also present, cyanide causes an anodic shift by + 0.09 V. Nickel changes the stress during deposition towards tensile, while cyanide changes it towards compressive. Both nickel and cyanide accelerate the transition to steady-state plating conditions.
Graphic Abstract










Similar content being viewed by others
References
Nakaso, K. Yamanoi, T. Okamura, Y. Tsuru (1985) Electroless copper deposition solution, US Patent 4548644
Lu J, Wang M, Deng X, Yan J, Yun J, Jiao S (2019) Evaluation of K3Fe(CN)6 on deposition behavior and structure of electroless copper plating. Electrochemistry 87:214–219
Ghosh S (2019) Electroless copper deposition: a critical review. Thin Solid Films 669:641–658
Bindra P, White JR (1990) Fundamental aspects of electroless copper plating, Ch. 12. In: Mallory GO, Hajdu JB (eds) Electroless plating fundamentals & applications. William Andrew, New York, pp 289–329
Bamberg S, Perry LK, Muir B, Abuzir A, Brüning F, Brüning R (2012) The effect of nickel on the strain evolution in chemical copper films. Thin Solid Films 520:6935–6941
Li L-S, Li X-R, Zhao W-X, Ma Q, Lu X, Wang Z (2013) A Study of low temperature and low stress electroless copper plating bath. Int J Electrochem Sci 8:5191–5202
Sharma T, Landry AE, Leger A, Brown DA, Bernhard T, Zarwell S, Brüning F, Brüning R (2018) Nickel dependence of hydrogen generation, hydrogen co-deposition and film stress in an electroless copper process. Thin Solid Films 666:76–84
Ballesteros JC, Chainet E, Ozil P, Meas Y, Trejo G (2011) Electrodeposition of copper from non-cyanide alkaline solution containing tartrate. Int J Electrochem Sci 6:2632–2651
Kochemirovky VA, Fateev SA, Logunov LS, Tumkin II, Safonov SV (2014) Laser-induced copper deposition with weak reducing agents. Int J Electrochem Sci 9:644–658
Shacham-Diamand Y, Dubin V, Angyal M (1995) Electroless copper deposition for ULSI. Thin Solid Films 262:93–103
Veleva R (1986) Role of potassium ferrocyanide in electroless copper baths. Surf Coat Technol 29:87–93
Schneble FW Jr, Zeblisky RJ, McCormack JF, Williamson JD (1968) Process for stabilizing autocatalytic metal plating solutions, US Patent 3403035
Bittner A, Wanner M, Weil KG (1992) The role of the microstructure of copper deposits during electroless plating in formaldehyde containing alkaline baths. Comparison of fourier transform impedance spectroscopy and surface enhanced Raman spectroscopy. Ber Bunsenges Phys Chem 96:647-655
Bernhard T, Zarwell S, Steinhäuser E, Kempa S, Brüning F (2019) The effect of Cu target pad roughness on the growth mode and void formation in electroless Cu films. In: International Wafer Level Packaging Conference (IWLPC), San Jose, CA, USA, pp 1-7
Nakahara S, Okinaka Y (1983) Microstructure and ductility of electroless copper deposits. Acta Metall 31:713–724
Saubestre EB (1972) Stabilizing electroless copper solutions. Plating 59:563–566
Logan E, Sharma T, Brüning F, Zarwell S, Steinhäuser E, Bernhard T, Chen N, Brüning R (2017) The effect of Ni on the kinetics of electroless Cu film deposition. Thin Solid Films 626:131–139
Brüning R, Sibley A, Sharma T, Brown DA, Demay T, Brüning F, Bernhard T (2014) Stress of electroless copper deposits on insulating and metal substrates. Thin Solid Films 565:136–142
Pötzelberger I, Mardare CC, Uiberlacker LM, Hild S, Mardare AI, Hassel AW (2017) Electrocatalysis on copper-palladium alloys for amperometric formaldehyde sensing. RSC Adv 7:6031–6039
Meerakker JE, Bakker JWG (1990) On the mechanism of electroless plating. Part 3. Electroless copper alloys. J Appl Electrochem 20:85–90
Bernhard T, Bamberg S, Brüning F, Brüning R, Gregoriades LJ, Sharma T, Brown D, Klaus M, Genzel Ch (2013) Analysis of stress/strain in electroless copper films. Int Symp Microelectr 2013:26–30
Underkofler WL, Zucconi TD (1974) Electroless copper plating, US Patent 3844799
Enyo M (1986) Electrooxidation of formaldehyde on Cu + Ni alloy electrodes in alkaline solutions. J Electroanal Chem 201:47–59
Acknowledgements
We thank Mr. Stefan Schweder for carrying out XRF measurements. SEM images were prepared by Mr. James Ehrman.
Funding
This study was funded by Atotech Deutschland GmbH.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Brown, D.A., MacDonald, A.R., McCarron, E.A. et al. Properties of an electroless copper process as a function of nickel and cyanide ion concentrations. J Appl Electrochem 51, 795–802 (2021). https://doi.org/10.1007/s10800-021-01535-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-021-01535-3