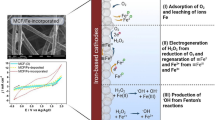
Abstract
Cobalt tetraaminophthalocyanine was anchored covalently on carbon fiber using an easy and moderate one-step deamination method to obtain a supported heterogeneous catalyst (CoPc-CF). Studies were conducted to understand the CoPc-CF electrode’s electrochemical activity, and some typical organic contaminants including dyes, phenols, and carbamazepine could be removed efficiently in this system. This system exhibited a relatively high electrochemical activity over a wide pH range, and provided a nonradical pathway, which was completely different from the traditional electro-Fenton system. The CoPc-CF electrode has a high electrocatalytic activity over a wide reactant concentration range. Repetitive tests showed that CoPc-CF could maintain a high electrocatalytic activity over several cycles. The content of electrogenerated H2O2 during the electrocatalysis process was determined using a photometric method in which N,N-diethyl-phenylenediamine was oxidized by a peroxidase-catalyzed reaction. The possible reaction mechanism was proposed from an electron paramagnetic resonance spin-trap technique. These results show that the CoPc-CF electrode has potential application in wastewater treatment.
Graphical Abstract












Similar content being viewed by others
References
Oturan MA, Peiroten J, Chartrin P, Acher AJ (2000) Complete destruction of ρ-nitrophenol in aqueous medium by electro-Fenton method. Environ Sci Technol 34:3474–3479
Chen M, Peng K, Wang H, Yang Z, Zeng Q, Xu A (2012) High performance of a simple cobalt(II)-monoethanolamine complex for orange II degradation with H2O2 as an oxidation at ambient conditions. Chem Eng J 197:110–115
Panakoulias T, Kalatzis P, Kalderis D, Katsaounis A (2010) Electrochemical degradation of Reactive Red 120 using DSA and BDD anodes. J Appl Electrochem 40:1759–1765
Zhao G, Cui X, Liu M, Li P, Zhang Y, Cao T, Li H, Lei Y, Liu L, Li D (2009) Electrochemical degradation of refractory pollutant using a novel microstructured TiO2 nanotubes/Sb-doped SnO2 electrode. Environ Sci Technol 43:1480–1486
Moreira FC, Garcia-Segura S, Boaventura RA, Brillas E, Vilar VJ (2014) Degradation of the antibiotic trimethoprim by electrochemical advanced oxidation processes using a carbon-PTFE air-diffusion cathode and a boron-doped diamond or platinum anode. Appl Catal B 160–161:492–505
Zhao X, Li A, Mao R, Liu H, Qu J (2014) Electrochemical removal of haloacetic acids in a three dimensional electrochemical reactor with Pd-GAC particles as fixed filler and Pd-modified carbon paper as cathode. Water Res 51:134–143
Sánchez J, Butter B, Rivas BL, Basáez L, Santander P (2015) Electrochemical oxidation and removal of arsenic using water-soluble polymers. J Appl Electrochem 45:151–159
Park AR, Lee YW, Kwak DH, Roh B, Hwang I, Park KW (2014) Enhanced electrocatalytic activity and stability of PdCo@Pt core-shell nanoparticles for oxygen reduction reaction. J Appl Electrochem 44:1219–1223
Ying J, Wang J, Jia J (2009) Improvement of electrochemical wastewater treatment through mass transfer in a seepage carbon nanotube electrode reactor. Environ Sci Technol 43:3796–3802
Vecitis CD, Gao G, Liu H (2011) Electrochemical carbon nanotube filter for adsorption, desorption, and oxidation of aqueous dyes and anions. J Phys Chem C 115:3621–3629
Neto SA, Andrade AR (2009) Electrochemical degradation of glyphosate formulations at DSA® anodes in chloride medium: an AOX formation study. J Appl Electrochem 39:1863–1870
Hsiao YL, Nobe K (1993) Hydroxylation of chlorobenzene and phenol in a packed bed flow reactor with electrogenerated Fenton’s reagent. J Appl Electrochem 23:943–946
Do JS, Yeh WC (1995) In situ degradation of formaldehyde with electrogenerated hypochlorite ion. J Appl Electrochem 25:483–489
Koparal AS, Yavuz Y, Gürel C, Öğütveren UB (2007) Electrochemical degradation and toxicity reduction of C.I. Basic Red 29 solution and textile wastewater by using diamond anode. J Hazard Mater 145:100–108
Panizza M, Cerisola G (2004) Electrochemical oxidation as a final treatment of synthetic tannery wastewater. Environ Sci Technol 38:5470–5475
Comninellis C, Nerini A (1995) Anodic oxidation of phenol in the presence of NaCl for wastewater treatment. J Appl Electrochem 25:23–28
Wu Z, Zhou M (2001) Partial degradation of phenol by advanced electrochemical oxidation process. Environ Sci Technol 35:2698–2703
Samet Y, Agengui L, Abdelhédi R (2010) Electrochemical degradation of chlorpyrifos pesticide in aqueous solutions by anodic oxidation at boron-doped diamond electrodes. Chem Eng J 161:167–172
Miwa DW, Malpass GRP, Machado ASA, Motheo AJ (2006) Electrochemical degradation of carbrayl on oxide electrodes. Water Res 40:3281–3289
Martínez-Huitle CA, Battisti AD, Ferro S, Reyna S, Cerro-López M, Quiro MA (2008) Removal of the pesticide methamidophos from aqueous solutions by electrooxidation using Pb/PbO2, Ti/SnO2, and Si/BDD electrodes. Environ Sci Technol 42:6929–6935
Li F, Zhou Y, Yang W, Chen G, Yang F (2006) Electrochemical degradation of amaranth aqueous solution on ACF. J Hazard Mater 137:1182–1188
Rodgers JD, Jedral W, Bunce NJ (1999) Electrochemical oxidation of chlorinated phenols. Environ Sci Technol 33:1453–1457
Chen A, Nigro S (2003) Influence of a nanoscale gold thin layer on Ti/Sb2O5 electrodes. J Phys Chem B 107:13341–13348
Reis RM, Valim RB, Rocha RS, Lima AS, Castro PS, Bertotti M, Lanza MRV (2014) The use of copper and cobalt phthalocyanines as electrocatalysts for the oxygen reduction reaction in acid medium. Electrochim Acta 139:1–6
Lu W, Chen W, Li N, Xu M, Yao Y (2009) Oxidative removal of 4-nitrophenol using cativated carbon fiber and hydrogen peroxide to enhance reactivity of metallophthalocyanine. Appl Catal B 87:146–151
Chen S, Lu W, Yao Y, Chen H, Chen W (2014) Oxidative desulfurization of dibenzothiophene with molecular oxygen catalyzed by carbon fiber-supported iron phthalocyanine. Reac Kinet Mech Cat 111:535–547
Achar BN, Fohlen GM, Parker JA, Keshavayya J (1987) Synthesis and structural studies of metal (II) 4, 9, 16, 23-phthalocyanine tetraamines. Polyhedron 6:1463–1467
Bader H, Sturzenegger V, Hoigne J (1988) Photometric method for the determination of low concentrations of hydrogen peroxide by the peroxidase catalyzed oxidation of N, N-diethyl-ρ-phenylenediamine (DPD). Water Res 22(9):1109–1115
Li N, Lu W, Pei K, Chen W (2015) Interfacial peroxidase-like catalytic activity of surface-immobilized cobalt phthalocyanine on multiwall carbon nanotubes. RSC Adv 5:9374–9380
Yi F, Chen S, Yuan C (2008) Effect of activated carbon fiber anode structure and electrolysis conditions on electrochemical degradation of dye wastewater. J Hazard Mater 157(1):79–87
Georgi A, Schierz A, Trommler U, Horwitz CP, Collons TJ, Kopinke FD (2007) Humic acid modified Fenton reagent for enhancement of the working pH range. Appl Catal B 72:26–36
Wang C, Hu J, Chou W, Kuo Y (2008) Removal of color from real dyeing wastewater by Electro-Fenton technology using a three-dimensional graphite cathode. J Hazard Mater 152:601–606
Brillas E, Calpe JC, Casado J (2000) Mineralization of 2,4-D by advanced electrochemical oxidation processes. Water Res 34(8):2253–2262
Kang S, Liao C, Chen M (2002) Pre-oxidation and coagulation of textile wastewater by the Fenton process. Chemosphere 46:923–928
Doorslaer XV, Heynderickx PM, Demeestere K, Debevere K, Langenhove HV, Dewulf J (2012) TiO2 mediated heterogeneous photocatalytic degradation of moxifloxacin: Operational variables and scavenger study. Appl Catal B 111–112:150–156
Ramirez JH, Duarte FM, Martins FG, Costa CA, Madeira LM (2009) Modelling of the synthetic dye Orange II degradation using Fenton’s reagent: from batch to continuous reactor operation. Chem Eng J 148(2–3):394–404
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant numbers: 51133006 and 51103133); the Textile Vision Science & Education Fund; the 521 Talent Project of ZSTU; Zhejiang Provincial Natural Science Foundation of China (Grant numbers: LY14E030013 and LY14E030015); and the Public Welfare Technology Application Research Project of Zhejiang Province (Grant number: 2015C33018).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, M., Xia, H., Lu, W. et al. Electrocatalytic degradation of organic contaminants using carbon fiber coupled with cobalt phthalocyanine electrode. J Appl Electrochem 46, 583–592 (2016). https://doi.org/10.1007/s10800-016-0939-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-016-0939-z