Abstract
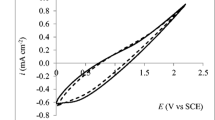
In the present paper, research on the electrochemical bleaching of p-nitrosodimethylaniline (RNO) in different electrolyte systems is presented with special attention to the role of RNO as a selective hydroxyl radical probe compound. At a Ti/Pt90–Ir10 anode, RNO was found to be bleached in 0.050 M sodium sulphate electrolyte due to lattice active oxygen without hydroxyl radicals being intermediately present. In 0.050 M sodium chloride, the bleaching rate was greatly enhanced due to indirect bulk oxidation by active chlorine species, again without the presence of hydroxyl radicals in the oxidation mechanisms. Under galvanostatic electrolysis, a linear relationship was found between the concentration of added chloride to a supporting sodium sulphate electrolyte and the first order rate constant of the bleaching reaction, showing the importance of the indirect bulk chlorine bleaching in chloride electrolyte systems. In this fashion both the chemically bonded active oxygen and the chemical bulk oxidation by active chlorine species proved to be valid bleaching pathways of RNO that according to these findings cannot be regarded as a fully selective hydroxyl radical probe compound. In addition, the difference in the mechanisms of chloride electrolysis at Ti/Pt90–Ir10 and Si–BDD anodes was clearly demonstrated using t-BuOH as hydroxyl radical scavenger.








Similar content being viewed by others
References
Mantzavinos D, Kassinos D, Parsons SA (2009) Water Res 43:3901
Comninellis C, Kapalka A, Malato S, Parsons SA, Poulios L, Mantzavinos D (2008) J Chem Technol Biotechnol 83:769
Comninellis C (1994) Electrochim Acta 39:1857
Kraljic I, Trumbore CN (1965) J Am Chem Soc 87:2547
Baxendale JH, Khan AA (1969) Int J Radiat Phys Chem 1:11
Bors W, Michel C, Saran M (1979) Eur J Biochem 95:621
Holt KB, Forryan C, Compton RG, Foord JS, Marken F (2003) New J Chem 27:698
Martinez-Huitle CA, Quiroz MA, Comninellis C, Ferro S, De Battisti A (2004) Electrochim Acta 50:949
Kapalka A, Foti G, Comninellis C (2008) J Appl Electrochem 38:7
Kunchandy E, Rao MNA (1990) Int J Pharm 58:237
Fitzl M, Suss R, Arnold K, Schiller J (2006) Chem Phys Lipids 140:11
Kaur IP, Geetha T (2006) Mini Rev Med Chem 6:305
Wabner D, Grambow C (1985) J Electroanal Chem 195:95
Feng C, Sugiura N, Shimada S, Maekawa T (2003) J Hazard Mater 103:65
Quiroz M, Reyna S, Sanchez J (2003) J Solid State Electr 7:277
Tanaka F, Feng CP, Sugiura N, Maekawa T (2004) J Environ Sci Health A Tox/hazard Subst Environ Eng 39:773
Bonfatti F, De Battisti A, Ferro S, Lodi G, Osti S (2000) Electrochim Acta 46:305
Holst G (1954) Chem Rev 54:169
Epstein JA, Lewin M (1962) J Polym Sci 58:991
Fukatsu K, Kokot S (2001) Polym Degrad Stab 72:353
Pi Y, Schumacher J, Jekel M (2005) Ozone Sci Eng 27:431
Buxton GV, Greenstock CL, Helman WP, Ross AB (1988) J Phys Chem Ref Data 17:513
Jeong J, Kim C, Yoon J (2009) Water Res 43:895
Canizares P, Martinez F, Diaz M, Garcia-Gomez J, Rodrigo MA (2002) J Electrochem Soc 149:D118
Martinez-Huitle CA, Ferro S (2006) Chem Soc Rev 35:1324
Muff J, Søgaard EG (2010) Water Sci Technol 61:2043
Muff J (2010) Ph.D. Thesis, Esbjerg Institute of Technology, Aalborg University, ISBN: 978-87-90033-71-2
Panizza M, Cerisola G (2009) Chem Rev 109:6541
Kraft A (2007) Int J Electrochem Sci 2:355
Serrano K, Michaud PA, Comninellis C, Savall A (2002) Electrochim Acta 48:431
Simonsen ME, Muff J, Bennedsen LR, Kowalski K, Søgaard EG (2010) J Photochem Photobiol A Chem 216:244
Bonfatti F, Ferro S, Lavezzo F, Malacarne M, Lodi G, De Battisti A (2000) J Electrochem Soc 147:592
Acknowledgments
Financial support from the Danish Ministry of Science, Technology, and Innovation in the form of the Ph.D. study grant is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muff, J., Bennedsen, L.R. & Søgaard, E.G. Study of electrochemical bleaching of p-nitrosodimethylaniline and its role as hydroxyl radical probe compound. J Appl Electrochem 41, 599–607 (2011). https://doi.org/10.1007/s10800-011-0268-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-011-0268-1