Abstract
Purpose
To investigate the clinical efficacy of conbercept 1 + pro re nata (PRN) (i.e., reinjection as needed after one injection) and 3 + PRN (reinjection as needed after 3 months of injection) regimens in choroidal neovascularization secondary to pathological myopia (PM-CNV).
Methods
From 06/2019 to 06/2020, 65 patients (65 eyes) confirmed with PM-CNV were included in this retrospective study. Intravitreal injection of 0.5 mg conbercept was conducted either with the 1 + PRN or 3 + PRN strategy. Patients were followed up for 12 months. The best-corrected visual acuity (BCVA), central retinal thickness (CRT), CNV lesion leakage area, the number of injections, and postoperative adverse reactions were observed.
Results
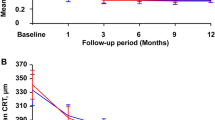
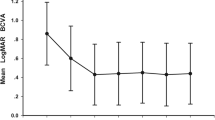
The mean age of the patients was 42.10 ± 4.69 years, and the average diopter was − 11.26 ± 2.97D. The BCVA at month 3 in the 3 + PRN (n = 30) group was lower than in the 1 + PRN (n = 35) group (P < 0.001). The CRT at month 3 in the 3 + PRN group was lower than in the 1 + PRN group (P < 0.001). After 12 months, there were no differences in the BCVA and CRT between the two groups (P > 0.05). The number of injections was less in 1 + PRN than in 3 + PRN (2.14 ± 1.06 vs. 3.37 ± 0.76, P < 0.001) at 12 months. No serious treatment-related ocular complications or serious systemic adverse events were found.
Conclusion
The 1 + PRN and 3 + PRN strategies of intravitreal injection of conbercept are effective in treating PM-CNV. The 1 + PRN regimen required fewer injections, and it might be more suitable for the treatment of PM-CNV.


Similar content being viewed by others
References
Ohno-Matsui K, Kawasaki R, Jonas JB, Cheung CM, Saw SM, Verhoeven VJ, Klaver CC, Moriyama M, Shinohara K, Kawasaki Y, Yamazaki M, Meuer S, Ishibashi T, Yasuda M, Yamashita H, Sugano A, Wang JJ, Mitchell P, Wong TY (2015) International photographic classification and grading system for myopic maculopathy. Am J Ophthalmol 159(5):877–83.e7. https://doi.org/10.1016/j.ajo.2015.01.022
Williams KM, Bertelsen G, Cumberland P, Wolfram C, Verhoeven VJ, Anastasopoulos E, Buitendijk GH, Cougnard-Grégoire A, Creuzot-Garcher C, Erke MG, Hogg R, Höhn R, Hysi P, Khawaja AP, Korobelnik JF, Ried J, Vingerling JR, Bron A, Dartigues JF, Fletcher A, Hofman A, Kuijpers RW, Luben RN, Oxele K, Topouzis F, von Hanno T, Mirshahi A, Foster PJ, van Duijn CM, Pfeiffer N, Delcourt C, Klaver CC, Rahi J, Hammond CJ (2015) Increasing prevalence of myopia in europe and the impact of education. Ophthalmology 122(7):1489–1497. https://doi.org/10.1016/j.ophtha.2015.03.018
Yoshida T, Ohno-Matsui K, Yasuzumi K, Kojima A, Shimada N, Futagami S, Tokoro T, Mochizuki M (2003) Myopic choroidal neovascularization: a 10-year follow-up. Ophthalmology 110(7):1297–1305. https://doi.org/10.1016/s0161-6420(03)00461-5
Ohno-Matsui K, Yoshida T, Futagami S, Yasuzumi K, Shimada N, Kojima A, Tokoro T, Mochizuki M (2003) Patchy atrophy and lacquer cracks predispose to the development of choroidal neovascularisation in pathological myopia. Br J Ophthalmol 87(5):570–573. https://doi.org/10.1136/bjo.87.5.570
Zhu Y, Zhang T, Xu G, Peng L (2016) Anti-vascular endothelial growth factor for choroidal neovascularisation in people with pathological myopia. Cochrane Database Syst Rev 12(12):Cd011160. https://doi.org/10.1002/14651858.CD011160.pub2
Tong JP, Chan WM, Liu DT, Lai TY, Choy KW, Pang CP, Lam DS (2006) Aqueous humor levels of vascular endothelial growth factor and pigment epithelium-derived factor in polypoidal choroidal vasculopathy and choroidal neovascularization. Am J Ophthalmol 141(3):456–462. https://doi.org/10.1016/j.ajo.2005.10.012
Ohno-Matsui K, Ikuno Y, Lai TYY, Gemmy Cheung CM (2018) Diagnosis and treatment guideline for myopic choroidal neovascularization due to pathologic myopia. Prog Retin Eye Res 63:92–106. https://doi.org/10.1016/j.preteyeres.2017.10.005
Kasahara K, Moriyama M, Morohoshi K, Yoshida T, Simada N, Nagaoka N, Yokoi T, Shinohara K, Kaneko Y, Suga M, Ohno-Matsui K (2017) Six-year outcomes of intravitreal bevacizumab for choroidal neovascularization in patients with pathologic myopia. Retina 37(6):1055–1064. https://doi.org/10.1097/iae.0000000000001313
Tufail A, Patel PJ, Sivaprasad S, Amoaku W, Browning AC, Cole M, Gale R, George S, Lotery AJ, Majid M, McKibbin M, Menon G, Yang Y, Andrews C, Brittain C, Osborne A (2013) Ranibizumab for the treatment of choroidal neovascularisation secondary to pathological myopia: interim analysis of the REPAIR study. Eye (Lond) 27(6):709–715. https://doi.org/10.1038/eye.2013.8
Cheung CMG, Arnold JJ, Holz FG, Park KH, Lai TYY, Larsen M, Mitchell P, Ohno-Matsui K, Chen SJ, Wolf S, Wong TY (2017) Myopic choroidal neovascularization: review, guidance, and consensus statement on management. Ophthalmology 124(11):1690–1711. https://doi.org/10.1016/j.ophtha.2017.04.028
Wang E, Chen Y (2013) Intravitreal anti-vascular endothelial growth factor for choroidal neovascularization secondary to pathologic myopia: systematic review and meta-analysis. Retina 33(7):1375–1392. https://doi.org/10.1097/IAE.0b013e31827d260a
Calvo-González C, Reche-Frutos J, Fernández-Vigo JI, Sáenz-Francés F, Fernández-Pérez C, García-Feijóo J (2017) Long-term outcomes of two different initial dosing regimens of intravitreal Ranibizumab used to treat myopic choroidal neovascularization. Ophthalmologica 238(4):196–204. https://doi.org/10.1159/000478030
Kung YH, Wu TT, Huang YH (2014) One-year outcome of two different initial dosing regimens of intravitreal ranibizumab for myopic choroidal neovascularization. Acta Ophthalmol 92(8):e615–e620. https://doi.org/10.1111/aos.12457
Ikuno Y, Ohno-Matsui K, Wong TY, Korobelnik JF, Vitti R, Li T, Stemper B, Asmus F, Zeitz O, Ishibashi T (2015) Intravitreal aflibercept injection in patients with myopic choroidal neovascularization: the MYRROR study. Ophthalmology 122(6):1220–1227. https://doi.org/10.1016/j.ophtha.2015.01.025
Zhang M, Yu D, Yang C, Xia Q, Li W, Liu B, Li H (2009) The pharmacology study of a new recombinant human VEGF receptor-fc fusion protein on experimental choroidal neovascularization. Pharm Res 26(1):204–210. https://doi.org/10.1007/s11095-008-9718-9
Li X, Xu G, Wang Y, Xu X, Liu X, Tang S, Zhang F, Zhang J, Tang L, Wu Q, Luo D, Ke X (2014) Safety and efficacy of conbercept in neovascular age-related macular degeneration: results from a 12-month randomized phase 2 study: AURORA study. Ophthalmology 121(9):1740–1747. https://doi.org/10.1016/j.ophtha.2014.03.026
Liu K, Wang H, He W, Ye J, Song Y, Wang Y, Liu X, Wu Z, Chen S, Fan K, Liu Y, Zhang F, Li Z, Liu L, Zhang J, Zhang X, Ye J, Liang X, Li X, Ke X, Wu Q, Li J, Tao S, Wang X, Rosenfeld P, Heier JS, Kaiser P, Xu X (2021) Intravitreal conbercept for diabetic macular oedema: 2-year results from a randomised controlled trial and open-label extension study. Br J Ophthalmol. https://doi.org/10.1136/bjophthalmol-2020-318690
Yan M, Huang Z, Lian HY, Song YP, Chen X (2019) Conbercept for treatment of choroidal neovascularization secondary to pathologic myopia. Acta Ophthalmol 97(5):e813–e814. https://doi.org/10.1111/aos.13632
Nie X, Wang Y, Yi H, Qiao Y (2021) Intravitreal conbercept for choroidal neovascularisation secondary to pathological myopia in a real-world setting in China: intravitreal conbercept was safe and effective in treating myopic choroidal neovascularization. BMC Ophthalmol 21(1):116. https://doi.org/10.1186/s12886-021-01877-8
Keane PA, Liakopoulos S, Chang KT, Heussen FM, Ongchin SC, Walsh AC, Sadda SR (2008) Comparison of the optical coherence tomographic features of choroidal neovascular membranes in pathological myopia versus age-related macular degeneration, using quantitative subanalysis. Br J Ophthalmol 92(8):1081–1085. https://doi.org/10.1136/bjo.2008.138891
Maloney MH, Payne SR, Herrin J, Sangaralingham LR, Shah ND, Barkmeier AJ (2021) Risk of systemic adverse events after intravitreal bevacizumab, Ranibizumab, and Aflibercept in routine clinical practice. Ophthalmology 128(3):417–424. https://doi.org/10.1016/j.ophtha.2020.07.062
Cho HJ, Yoo SG, Kim HS, Kim JH, Kim CG, Lee TG, Kim JW (2015) Risk factors for geographic atrophy after intravitreal ranibizumab injections for retinal angiomatous proliferation. Am J Ophthalmol 159(2):285–92.e1. https://doi.org/10.1016/j.ajo.2014.10.035
Schütze C, Wedl M, Baumann B, Pircher M, Hitzenberger CK, Schmidt-Erfurth U (2015) Progression of retinal pigment epithelial atrophy in antiangiogenic therapy of neovascular age-related macular degeneration. Am J Ophthalmol 159(6):1100-1114.e1. https://doi.org/10.1016/j.ajo.2015.02.020
Wolf S, Balciuniene VJ, Laganovska G, Menchini U, Ohno-Matsui K, Sharma T, Wong TY, Silva R, Pilz S, Gekkieva M (2014) RADIANCE: a randomized controlled study of ranibizumab in patients with choroidal neovascularization secondary to pathologic myopia. Ophthalmology 121(3):682–92.e2. https://doi.org/10.1016/j.ophtha.2013.10.023
Lu X, Sun X (2015) Profile of conbercept in the treatment of neovascular age-related macular degeneration. Drug Des Devel Ther 9:2311–2320. https://doi.org/10.2147/dddt.S67536
Yao TT, Jin XL, Yang Y, Wang YX, Zhou YL, He FL, Wang ZY (2020) Intraocular pharmacokinetics and safety of subretinal injection compared with intravitreal application of conbercept in vitrectomized rabbit eyes. J Ophthalmol 2020:2674780. https://doi.org/10.1155/2020/2674780
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
None.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
The present study was reviewed and approved by the Institutional Review Board of Jinan 2nd People's Hospital (approval number: [2019] 018) and was conducted in compliance with the principles of the Declaration of Helsinki.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gong, B., Bo, Y., Zhang, P. et al. Efficacy and safety of different conbercept injection regimens in the treatment of choroidal neovascularization in pathological myopia: a retrospective study. Int Ophthalmol 43, 4079–4086 (2023). https://doi.org/10.1007/s10792-023-02825-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-023-02825-9