Abstract
Purpose
This study aimed to compare choroidal thickness parameters between patients diagnosed with Crimean–Congo hemorrhagic fever (CCHF) and healthy individuals using spectral domain optical coherence tomography.
Methods
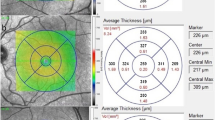
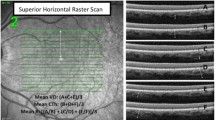
The right eyes of 27 individuals diagnosed with CCHF and 27 healthy subjects were included in this study. CCHF cases were assessed based on a history of tick bites and hospitalization. Choroidal thickness was measured using enhanced depth imaging optical coherence tomography. Measurements of choroidal thickness were taken at 5 different points, including the subfoveal quadrant and the fovea up to 2000 μm with 1000 μm intervals in the temporal and nasal quadrants. Choroidal vascular index (CVI) was calculated as the ratio of luminal area (LA) to total choroidal area (TCA) at the macula's total area and at 1500 μm from the center (CVI and CVI1500).
Results
Choroidal thickness in the CCHF group was found to be thicker in each quadrant compared to the control group, with the subfoveal and nasal quadrants showing significantly greater thickness. TCA, stromal area, and LA were significantly higher in the CCHF group, while no significant difference was observed in CVI and CVI1500.
Conclusion
The findings of this study suggest that CCHF disease may have an impact on the choroidal structure.


Similar content being viewed by others
References
Whitehouse CA (2004) Crimean–congo hemorrhagic fever. Antiviral Res 64(3):145–160. https://doi.org/10.1016/j.antiviral.2004.08.001
Adams MJ, Lefkowitz EJ, King AMQ et al (2017) Changes to taxonomy and the international code of virus classification and nomenclature ratified by the international committee on taxonomy of viruses (2017). Arch Virol 162(8):2505–2538
Leblebicioglu H (2010) Crimean–congo haemorrhagic fever in Eurasia. Int J Antimicrob Agents 36(1):S43–S46
Ergönül Ö (2016) Kırım Kongo kanamalı ateşi tedavisi ve ribavirin kullanımı. Klimik Dergisi 29:2–9
Burt FJ, Swanepoel R, Shieh WJ et al (1997) Immunohistochemical and in situ localization of Crimean–congo hemorrhagic fever (CCHF) virus in human tissues and implications for CCHF pathogenesis. Arch Pathol Lab Med 121:839–846
Schnittler HJ, Feldmann H (2003) Viral hemorrhagic fever-a vascular disease? Thromb Haemost 89:967–972
Agrawal R, Gupta P, Tan KA et al (2016) Choroidal vascularity index as a measure of vascular status of the choroid: measurements in healthy eyes from a population-based study. Sci Rep 6:21090. https://doi.org/10.1038/srep21090
Siam A, Meegan J, Gharbawi K (1980) Rift valley fever ocular manifestations: observations during the 1977 epidemic in Egypt. British J Ophthalmol 64(5):366–374. https://doi.org/10.1136/bjo.64.5.366
Kapoor HK, Bhai S, John M et al (2006) Ocular manifestations of dengue fever in an east Indian epidemic. Can J Ophthalmol 41(6):741–746. https://doi.org/10.3129/i06-069
Engin A, Erdogan H, Ozec AV (2009) Ocular findings in patients with Crimean–congo hemorrhagic fever. Am J Ophthalmol 147(4):634–638. https://doi.org/10.1016/j.ajo.2008.11.014
Bakir M, Ugurlu M, Dokuzoguz B et al (2005) Turkish CCHF study group. Crimean–congo haemorrhagic fever outbreak in middle Anatolia: a multicentre study of clinical features and outcome measures. J Med Microbiol 54:385–389
Ergonul O, Celikbas A, Baykam N et al (2006) Analysis of risk-factors among patients with crimean-congo haemorrhagic fever virus infection: severity criteria revisited. Clin Microbiol Infect 12:551–554
Ergünay K, Polat C, Özkul A (2020) Vector-borne viruses in Turkey: a systematic review and bibliography. Antiviral Res 183:104934
Yalinbas D, Komurluoglu A, Bozali E (2021) Increased retinal vessel tortuosity associated with crimean-congo hemorrhagic fever in children. Pediatr Infect Dis J 40(10):880–884. https://doi.org/10.1097/INF.0000000000003187. (PMID: 34260498)
Yalinbas D, Bozali E, Vural A et al (2022) Purtscher-like retinopathy associated with crimean-congo hemorrhagic fever: a case report. Ocul Immunol Inflamm 30(4):1016–1019. https://doi.org/10.1080/09273948.2020.1841805
Tan KA, Gupta P, Agarwal A et al (2016) State of science: choroidal thickness and systemic health. Surv Ophthalmol 61(5):566–581. https://doi.org/10.1016/j.survophthal.2016.02.007
Konuk ŞG, Kılıç R, Türkyılmaz B et al (2022) Choroidal thickness changes in post-COVID-19 cases. Arq Bras Oftalmol. https://doi.org/10.5935/0004-2749.20230021
Kılıç R, Kurt A, Acer E et al (2017) Choroidal thickness in psoriasis. Int Ophthalmol 37(1):173–177. https://doi.org/10.1007/s10792-016-0252-1
Ishikawa S, Taguchi M, Muraoka T et al (2014) Changes in subfoveal choroidal thickness associated with uveitis activity in patients with Behçet’s disease. Br J Ophthalmol 98(11):1508–1513
Maruko I, Lida T, Sugano Y et al (2011) Subfoveal choroidal thickness after treatment of vogt-koyanagi-harada disease. Retina 31(3):510–517
Agrawal R, Chhablani J, Tan KA (2016) Choroidal vascularity index in central serous chorioretinopathy. Retina 36:1646–1651
Bakthavatsalam M, Ng DS, Lai FH et al (2017) Choroidal structures in polypoidal choroidal vasculopathy, neovascular age-related maculopathy, and healthy eyes determined by binarization of swept source optical coherence tomographic images. Graefes Arch Clin Exp Ophthalmol 255:935–943
Read RW, Zamir E, Rao NA (2002) Neoplastic masquerade syndromes. Surv Ophthalmol 47:81–124
Lopez JS, Chan CC, Burnier M et al (1991) Immunohistochemistry findings in primary intraocular lymphoma. Am J Ophthalmol 112:472–474
Ağın A, Kadayıfçılar S, Sönmez HE et al (2019) Evaluation of choroidal thickness, choroidal vascularity index and peripapillary retinal nerve fiber layer in patients with juvenile systemic lupus erythematosus. Lupus 28(1):44–50. https://doi.org/10.1177/0961203318814196
Funding
No financial support was received for this submission.
Author information
Authors and Affiliations
Contributions
ŞGK and ETY conceived of the presented idea. ŞGK developed the theory and performed the computations. ŞGK and RK verified the analytical methods. All authors discussed the results and contributed to the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Tokat Gaziosmanpaşa University Faculty of Medicine clinical research ethics committee (Date: 02.09.2021/No: 21KAEK-196) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent to publication
All participants agreed to the collection and anonymous publication of the data.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Konuk, Ş.G., Kılıç, R., Yılmaz, E.T. et al. Evaluation of choroidal thickness and choroidal vascularity index in patients with Crimean-Congo hemorrhagic fever. Int Ophthalmol 43, 4163–4169 (2023). https://doi.org/10.1007/s10792-023-02817-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-023-02817-9