Abstract
Purpose
Dry eye disease (DED) is a disease with tear film instability because of multiple factors. This study was conducted to explore roles of occludin and MUC5AC in tear film instability in DED rat model.
Methods
A total of 20 SD rats were divided into DED group (n = 10) and normal control (NC) group (n = 10). DED rat model was established by subcutaneously injecting with scopolamine hydrobromide. Clinical examinations, including tear breakup time (tBUT), Schirmer’s test and corneal fluorescein staining, were conducted to determine corneal functions. Transmission electron microscopy was used to measure the ultrastructures of corneal epithelial cells. Western blotting assay was used to identify occludin expression in corneal tissues of DED rats. Real-time PCR (RT-PCR) was performed to verify gene transcription of occludin and MUC5AC. Colocalization between occludin and MUC5AC was identified with confocal fluorescence microscopy.
Results
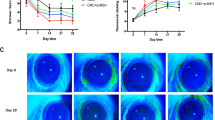
Tear breakup time was significantly shorter, and corneal fluorescein staining score was predominantly higher in DED rats compared to those in normal rats (P < 0.05). Normal rats showed a steady tear secretion throughout the whole experiments, while DED rats showed a dramatic reduction on day 14. DED rats demonstrated ultrastructural damage of Golgi apparatus and endoplasmic reticulum in corneal epithelial cells. Occludin and MUC5AC expressions were significantly downregulated in corneal tissue of DED rats compared with those of normal rats (P < 0.05). Percentage of occludin-MUC5AC-colocalized corneal epithelial cells in DED rats was significantly less compared with those in normal rats (P < 0.01).
Conclusions
Tear film stability was damaged in scopolamine-induced DED rats because of the weakened colocalization between occludin and MUC5AC molecule. This study would provide a potential clue for the pathogenesis and a promising theoretical basis for clinical work of DED.





Similar content being viewed by others
References
Chinese branch of the asian dry eye society, ocular surface and tear film diseases group of ophthalmology committee of cross-straits medicine exchange association, ocular surface and dry eye group of Chinese ophthalmologist association. Expert consensus on dry eye in China: dry eye related to eye surgery (2021) Zhonghua Yan Ke Za Zhi 2021;57:564–572
Craig JP, Nichols KK, Akpek EK et al (2017) TFOS DEWS II definition and classification report. Ocul Surf 15:276–283
Golden MI, Meyer JJ, Patel BC (2021) Dry eye syndrome. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing, Tampa, 2022 Jan
Messmer EM (2015) The pathophysiology, diagnosis, and treatment of dry eye disease. Dtsch Arztebl Int 112:71–81
Lemp MA, Foulks GN (2007) The definition and classification of dry eye disease: report of the definition and classification subcommittee of the international dry eye workshop (2007). Ocul Surf 5:75–92
Fini ME, Bauskar A, Jeong S, Wilson MR (2016) Clusterin in the eye: an old dog with new tricks at the ocular surface. Exp Eye Res 147:57–71
Wu MF, Gao H, Zhao LJ, Chen H, Huang YK (2020) Real dynamic assessment of tear film optical quality for monitoring and early prevention of dry eye. Medicine (Baltimore) 99:e21494
Pflugfelder SC, Stern ME (2020) Biological functions of tear film. Exp Eye Res 197:108115
Shimazaki-Den S, Dogru M, Higa K, Shimazaki J (2013) Symptoms, visual function, and mucin expression of eyes with tear film instability. Cornea 32:1211–1218
Woodward AM, Senchyna M, Argüeso P (2021) Short-term reproducibility of MUC5AC measurement in human tear fluid. Diagnostics (Basel) 11:57
Liu W, Li H, Lu D et al (2010) The tear fluid mucin 5AC change of primary angle-closure glaucoma patients after short-term medications and phacotrabeculectomy. Mol Vis 16:2342–2346
Mantelli F, Argüeso P (2008) Functions of ocular surface mucins in health and disease. Curr Opin Allergy Clin Immunol 8:477–483
Putra I, Rabiee B, Anwar KN et al (2019) Staphylococcus aureus alpha-hemolysin impairs corneal epithelial wound healing and promotes intracellular bacterial invasion. Exp Eye Res 181:263–270
Pu Q, Guo XX, Hu JJ, Li AL, Li GG, Li XY (2022) Nicotinamide mononucleotide increases cell viability and restores tight junctions in high-glucose-treated human corneal epithelial cells via the SIRT1/Nrf2/HO-1 pathway. Biomed Pharmacother 147:112659
Luo X, Guo L, Zhang J et al (2017) Tight junction protein occludin is a porcine epidemic diarrhea virus entry factor. J Virol 91:e00202–e00217
Jiang Y, Song J, Xu Y et al (2021) Piezo1 regulates intestinal epithelial function by affecting the tight junction protein claudin-1 via the ROCK pathway. Life Sci 275:119254
McDermott JR, Bartram RE, Knight PA, Miller HR, Garrod DR, Grencis RK (2003) Mast cells disrupt epithelial barrier function during enteric nematode infection. Proc Natl Acad Sci USA 100:7761–7766
Kim KA, Kim D, Kim JH et al (2020) Autophagy-mediated occludin degradation contributes to blood–brain barrier disruption during ischemia in bEnd. 3 brain endothelial cells and rat ischemic stroke models. Fluids Barriers CNS 17:21
Wang Z, Wade P, Mandell KJ et al (2007) Raf 1 represses expression of the tight junction protein occludin via activation of the zinc-finger transcription factor slug. Oncogene 26:1222–1230
Zhang C, Xie H, Yang Q et al (2019) Erythropoietin protects outer blood-retinal barrier in experimental diabetic retinopathy by up-regulating ZO-1 and occludin. Clin Exp Ophthalmol 47:1182–1197
Lv Y, Chu C, Liu K et al (2021) A combination of CMC and α-MSH inhibited ROS activated NLRP3 inflammasome in hyperosmolarity stressed HCECs and scopolamine-induced dry eye rats. Sci Rep 11:1184
Chu YY, Hua N, Ru YS, Zhao SZ (2017) The protection of hydrogen-rich saline on a rat dry eye model induced by scopolamine hydrobromide. Zhonghua Yan Ke Za Zhi 53:363–372
Ru Y, Huang Y, Liu H et al (2015) α-Melanocyte-stimulating hormone ameliorates ocular surface dysfunctions and lesions in a scopolamine-induced dry eye model via PKA-CREB and MEK-Erk pathways. Sci Rep 5:18619
Sakamoto R, Bennett ES, Henry VA et al (1993) The phenol red thread tear test: a cross-cultural study. Invest Ophthalmol Vis Sci 34:3510–4354
Pauly A, Brignole-Baudouin F, Labbé A, Liang H, Warnet JM, Baudouin C (2007) New tools for the evaluation of toxic ocular surface changes in the rat. Invest Ophthalmol Vis Sci 48:5473–5483
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–212
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(–Delta Delta C(T)) Method. Methods 25:402–408
Eldarov CM, Vangely IM, Vays VB et al (2020) Mitochondria in the nuclei of rat myocardial cells. Cells 9:712
Choi SY, Eom Y, Kim JY, Jang DH, Song JS, Kim HM (2020) Effect of natural extract eye drops in dry eye disease rats. Int J Ophthalmol 13:1023–1030
Surmacz HU, Cotlinski AL, Gehlen ML, Nisihara R, Skare TL (2021) Dry eye and percentage of body fat: a cross-sectional prospective study. Int Ophthalmol 41:1855–1861
Drevets WC, Zarate CA, Furey ML (2013) Antidepressant effects of the muscarinic cholinergic receptor antagonist scopolamine: a review. Biol Psychiatry 73:1156–1163
Lemp MA (2008) Advances in understanding and managing dry eye disease. Am J Ophthalmol 146:350–356
Bandamwar KL, Papas EB, Garrett Q (2014) Fluorescein staining and physiological state of corneal epithelial cells. Cont Lens Anterior Eye 37:213–223
Ramkumar HL, Zhang J, Chan CC (2010) Retinal ultrastructure of murine models of dry age-related macular degeneration (AMD). Prog Retin Eye Res 29:169–190
Ru YX, Zhao SX, Dong SX, Liang HY, Wang Y (2018) Systematic alteration of apoptosis: a review with ultrastructural observations on leukemia cells in vivo. Ultrastruct Pathol 42:1–9
Gipson IK (2004) Distribution of mucins at the ocular surface. Exp Eye Res 78:379–388
Hampel U, Garreis F, Burgemeister F, Eßel N, Paulsen F (2018) Effect of intermittent shear stress on corneal epithelial cells using an in vitro flow culture model. Ocul Surf 16:341–351
Liu JJ, Zhang T, Mi YM (2019) Effects of respiratory syncytial virus infection on epidermal growth factor receptor, tight junction association proteins and mucin in airway epithelial cells. Zhongguo Dang Dai Er Ke Za Zhi 21:294–299
Funding
This study was granted by the National Natural Science Foundation of China (Youth Program) (Grant No. NSFC81400382), Shaanxi Innovation Capability Support Program (Grant No. 2016KJXX-82) and Scientific Research Talent Project of Xi'an Health Commission (Grant No. 2022yb19).
Author information
Authors and Affiliations
Contributions
All authors read and approved final manuscript. XK had the idea for article, drafted the manuscript, performed literature, critically revised manuscript and made the final approval; XNL, HBZ, XPZ and XJZ performed the literature search and data analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This research has been approved by the Ethical Committee of the Xi’an Center for Disease Control and Prevention (Approval No. 2021-001). This study was performed based on the guidance of Declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, K., Liu, Xn., Zhang, Hb. et al. Tear film instability is associated with weakened colocalization between occludin and MUC5AC in scopolamine-induced dry eye disease (DED) rats. Int Ophthalmol 43, 463–473 (2023). https://doi.org/10.1007/s10792-022-02443-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-022-02443-x