Abstract
Background
A significant proportion of eyes with polypoidal choroidal vasculopathy (PCV) can be resistant to anti-vascular endothelial growth factor (VEGF) injections. We evaluated the efficacy of a combination of dexamethasone intravitreal implant (DXI) and anti-VEGF therapy in eyes resistant to anti-VEGF monotherapy.
Methods
In this retrospective study, patients with PCV resistant to anti-VEGF injections were additionally injected with a DXI along with an anti-VEGF agent. Best-corrected visual acuity (BCVA), slit-lamp examination, fundus evaluation, and optical coherence tomography (OCT) data were analyzed. Anatomical response on OCT was the primary outcome measure. Change in visual acuity and injection-free interval after DXI were evaluated as secondary outcome measures.
Results
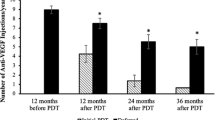
Twelve eyes of 11 patients were included in the study. Mean age of patients at presentation was 64.7 ± 9.5 years (range, 49–78.8 years), and there were seven females (63.6%). Median number of anti-VEGF injections prior to DXI was 4 (interquartile range IQR, 3–7). Median follow-up duration after DXI was 32.2 months (IQR, 6.6–41.6 months). Median logMAR BCVA immediately prior to DXI was 0.41 (IQR, 0.30–0.88) and after injection was 0.40 (IQR, 0.30–1.05), which was not significantly different (p = 0.85). Median Central Retinal Thickness (CRT) after DXI was 305.5 µm (IQR, 249–409 µm), which was significantly (p = 0.003) lesser than pre-injection thickness of 547 µm (IQR, 431–771 µm). Median injection-free interval in these eyes after DXI was 5 months (IQR, 2.8–6.4 months). Kaplan–Meier estimates of first injection after DXI were 27.3% at 3 months, 67.3% at 6 months, and 89.1% at 12 months.
Conclusions
Dexamethasone implant combined with anti-VEGF treatment can prolong the treatment-free interval in eyes with PCV resistant to anti-VEGF injection while maintaining visual acuity.






Similar content being viewed by others
Abbreviations
- BCVA:
-
Best-corrected visual acuity
- CNVM:
-
Choroidal neovascular membrane
- CRT:
-
Central retinal thickness
- DXI:
-
Dexamethasone implant
- FFA:
-
Fundus fluorescein angiography
- ICGA:
-
Indocyanine green angiography
- IOP:
-
Intraocular pressure
- IQR:
-
Inter-quartile range
- ME:
-
Macular edema
- nAMD:
-
Neovascular age-related macular degeneration
- PCV:
-
Polypoidal choroidal vasculopathy
- PED:
-
Pigment epithelial detachment
- OCT:
-
Optical coherence tomography
- VEGF:
-
Vascular endothelial growth factor
References
Koh A, Lee WK, Chen LJ, Chen SJ, Hashad Y, Kim H et al (2012) EVEREST study: efficacy and safety of verteporfin photodynamic therapy in combination with ranibizumab or alone versus ranibizumab monotherapy in patients with symptomatic macular polypoidal choroidal vasculopathy. Retina 32(8):1453–1464
Lee WK, Iida T, Ogura Y, Chen SJ, Wong TY, Mitchell P et al (2018) Efficacy and Safety of Intravitreal Aflibercept for Polypoidal Choroidal Vasculopathy in the PLANET Study: A Randomized Clinical Trial. JAMA Ophthalmol 136(7):786–793
Martin DF, Maguire MG, Fine SL, Ying GS, Jaffe GJ, Grunwald JE et al (2012) Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: two-year results. Ophthalmology 119:1388–1398
Heier JS, Brown DM, Chong V, Korobelnik JF, Kaiser PK, Nguyen QD et al (2012) Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology 119:2537–2548
Lux A, Llacer H, Heussen FM, Joussen AM (2007) Non-responders to bevacizumab (Avastin) therapy of choroidal neovascular lesions. Br J Ophthalmol 91:1318–1322
Krebs I, Glittenberg C, Ansari-Shahrezaei S, Hagen S, Steiner I, Binder S (2013) Non-responders to treatment with antagonists of vascular endothelial growth factor in age-related macular degeneration. Br J Ophthalmol 97:1443–1446
Suzuki M, Nagai N, Izumi-Nagai K, Shinoda H, Koto T, Uchida A et al (2014) Predictive factors for non-response to intravitreal ranibizumab treatment in age-related macular degeneration. Br J Ophthalmol 98:1186–1191
Kokame GT, deCarlo TE, Kaneko KN, Omizo JN, Lian R (2019) Anti-Vascular endothelial growth factor resistance in exudative macular degeneration and polypoidal choroidal vasculopathy. Ophthalmology Retina 3:744–752
Yang S, Zhao J, Sun X (2016) Resistance to anti-VEGF therapy in neovascular age-related macular degeneration: a comprehensive review. Drug Des Dev Ther 10:1857
Agrawal R, Balne PK, Wei X, Bijin VA, Lee B, Ghosh A et al (2019) Cytokine profiling in patients with exudative age-related macular degeneration and polypoidal choroidal vasculopathy. Invest Ophthalmol Vis Sci 60(1):376–382
Kuppermann BD, Goldstein M, Maturi RK, Pollack A, Singer M, Tufail A et al (2015) Dexamethasone intravitreal implant as adjunctive therapy to ranibizumab in neovascular age-related macular degeneration: a multicenter randomized controlled trial. Ophthalmologica 234:40–54
Ranchod TM, Ray SK, Daniels SA, Leong CJ, Ting TD, Verne AZ (2013) LuceDex: a prospective study comparing ranibizumab plus dexamethasone combination therapy versus ranibizumab monotherapy for neovascular age-related macular degeneration. Retina 33(8):1600–1604
Calvo P, Ferreras A, Al Adel F, Wang Y, Brent MH (2015) Dexamethasone intravitreal implant as adjunct therapy for patients with wet age-related macular degeneration with incomplete response to ranibizumab. Br J Ophthalmol 99:723–726
Rezar-Dreindl S, Eibenberger K, Buehl W, Georgopoulos M, Weigert G, Krall C et al (2017) Role of additional dexamethasone for the management of persistent or recurrent neovascular age-related macular degeneration under ranibizumab treatment. Retina 37:962–970
Todorich B, Thanos A, Yonekawa Y, Mane G, Hasbrook M, Thomas BJ et al (2017) Simultaneous dexamethasone intravitreal implant and anti-vegf therapy for neovascular age-related macular degeneration resistant to anti-vegf monotherapy. J Vitreoret Dis 1:65–74
Giancipoli E, Pinna A, Boscia F, Zasa G, Sotgiu G, Dore S et al (2018) Intravitreal dexamethasone in patients with wet age-related macular degeneration resistant to anti-vegf: A prospective pilot study. J Ophthalmol 2018:1–8
Jonas JB, Libondi T, Golubkina L, Spandau UH, Schlichtenbrede F, Rensch F (2010) Combined intravitreal bevacizumab and triamcinolone in exudative age-related macular degeneration. Acta Ophthalmol 88:630–634
Tozer K, Roller AB, Chong LP, Sadda S, Folk JC, Mahajan VB et al (2013) Combination therapy for neovascular age-related macular degeneration refractory to anti-vascular endothelial growth factor agents. Ophthalmology 120:2029–2034
Veritti D, Sarao V, Lanzetta P (2013) Bevacizumab and triamcinolone acetonide for choroidal neovascularization due to age-related macular degeneration unresponsive to antivascular endothelial growth factors. J Ocul Pharmacol Ther 29:437–441
Kauppinen A, Paterno JJ, Blasiak J, Salminen A, Kaarniranta K (2016) Inflammation and its role in age-related macular degeneration. Cell Mol Life Sci 73:1765–1786
Kauppinen A, Niskanen H, Suuronen T, Kinnunen K, Salminen A, Kaarniranta K (2012) Oxidative stress activates NLRP3 inflammasomes in ARPE-19 cells—implications for age-related macular degeneration (AMD). Immunol Lett 147:29–33
Nozaki M, Raisler BJ, Sakurai E, Sarma JV, Barnum SR, Lambris JD et al (2006) Drusen complement components C3a and C5a promote choroidal neovascularization. Proc Natl Acad Sci 103:2328–2333
Machalińska A, Dziedziejko V, Mozolewska-Piotrowska K, Karczewicz D, Wiszniewska B, Machaliński B (2009) Elevated plasma levels of C3a complement compound in the exudative form of age-related macular degeneration. Ophthalmic Res 42:54–59
Oliver A, Ciulla TA (2006) Corticosteroids as antiangiogenic agents. Ophthalmol Clin North Am 19:345–351
Oh H, Takagi H, Takagi C, Suzuma K, Otani A, Ishida K et al (1999) The potential angiogenic role of macrophages in the formation of choroidal neovascular membranes. Invest Ophthalmol Vis Sci 40:1891–1898
Schaal S, Kaplan HJ, Tezel TH (2008) Is there tachyphylaxis to intravitreal anti-vascular endothelial growth factor pharmacotherapy in age-related macular degeneration? Ophthalmology 115:2199–2205
Kwak HW, D’Amico DJ (1992) Evaluation of the retinal toxicity and pharmacokinetics of dexamethasone after intravitreal injection. Arch Ophthalmol 110:259–266
Chang-Lin JE, Attar M, Acheampong AA, Robinson MR, Whitcup SM, Kuppermann BD et al (2011) Pharmacokinetics and pharmacodynamics of a sustained-release dexamethasone intravitreal implant. Invest Ophthalmol Vis Sci 52:80–86
van Wijngaarden P, Coster DJ, Williams KA (2005) Inhibitors of ocular neovascularization: promises and potential problems. JAMA 293:1509–1513
Acknowledgements
Hyderabad Eye Research Foundation and India Alliance DB Wellcome Trust.
Funding
None.
Author information
Authors and Affiliations
Contributions
RN, KM, and AM contributed to data collection, analysis and interpretation, preparation of the manuscript, and provided major revisions to the manuscript. RR, MPS, RSA, and SKP contributed to interpretation and final analysis of manuscript. AM and KM performed statistical analysis. All authors participated in the development and writing of the manuscript and approved the final article for publication.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any competing interests.
Data availability and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on request.
Ethical approval
The study protocol adhered to the tenets of the Declaration of Helsinki and was approved by the local Institutional Review Board (LEC 05–18-089) at L.V. Prasad Eye Institute. All study participants gave a written informed consent before enrollment. Patients were recruited from March 2017 through November 2019.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mallikarjun, K., Narayanan, R., Raman, R. et al. Dexamethasone implant improves anatomic response to anti-VEGF therapy in treatment-resistant polypoidal choroidal vasculopathy. Int Ophthalmol 42, 1263–1272 (2022). https://doi.org/10.1007/s10792-021-02113-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-021-02113-4