Abstract
Background/objectives
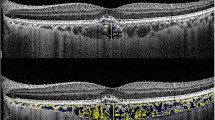
To analyze macular choroidal thickness in patients with pseudoxanthoma elasticum (PXE) by enhanced depth imaging optical coherence tomography (EDI-OCT).
Subjects/methods
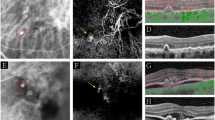
This is a prospective cross-sectional study. Sixty-eight eyes of 34 patients with PXE and 68 normal eyes of 34 controls were included to study the macular area with enhanced depth imaging spectral-domain optical coherence tomography (EDI-OCT). Eyes with PXE were classified in three groups: those without choroidal neovascularization (CNV) or chorioretinal macular atrophy macular (Group 1); those with active CNV (Group 2) and those with macular atrophy secondary to inactive CNV (Group 3).
Results
Mean subfoveal choroidal thickness (CT) was 266.70 ± 46.93 µm in control group, 304.24 ± 65.52 µm in group 1, 198.55 ± 66.33 µm in group 2, and 119.45 ± 63.89 µm in group 3 (p = 0.00). Comparison between PXE subgroups showed that subfoveal CT was significantly decreased in group 2 and 3 compared to group 1 (p < 0.0001 for both groups). The CT in the different quadrants (superior, inferior, temporal and nasal) was significantly thinner in group 3, followed by group 2 and 1 in ascendant order. Group 1 showed significant increased thickness compared to the other groups.
Conclusion
To the best of our knowledge, this is the first report suggesting thicker macular choroid in patients with PXE without active or inactive CNV than in normal eyes. Initial changes in Bruch membrane (MB) and choroid, in addition to the increased oxidative stress, would lead to hyperpermeability of the choroid and alterations of the barrier BM-RPE causing a thick choroid in early stages.



Similar content being viewed by others
References
Bergen AA, Plomp AS, Schuurman EJ, Terry S, Breuning M, Dauwerse H et al (2000) Mutations in ABCC6 cause pseudoxanthoma elasticum. Nat Genet 25:228–231
Le Saux O, Urban Z, Tschuch C, Csiszar K, Bacchelli B, Quaglino D et al (2000) Mutations in a gene encoding an ABC transporter cause pseudoxanthoma elasticum. Nat Genet 25:223–227
Ringpfeil F, Lebwohl MG, Christiano AM, Uitto J (2000) Pseudoxanthoma elasticum: mutations in the MPR6 gene encoding a transembrane ATP-binding cassette (ABC) transporter. Proc Natl Acad Scei USA 97:6001–6006
Struk B, Neldner KH, Rao VS, St Jean P, Lindpaintner K (1997) Mapping of both autosomal recessive and dominant variants of pseudoxanthoma elasticum to chromosome 16p13.1. Hum Mol Genet 6(11):1823–1828
Kool M, van der Linden M, de Haas M, Baas F, Borst P (1999) Expression of human MRP6, a homologue of the multidrug resistance protein gene MRP1, in tissues and cáncer cells. Cáncer Res 59:175–182
Uitto J, Li Q, Jiang Q (2010) Pseudoxanthoma elasticum: molecular genetics and putative pathomechanisms. J Invest Dermatol 130:661–670
Finger RP, Charbel Issa P, Ladewig MS, Götting C, Szliska C, Scholl HP et al (2009) Pseudoxanthoma elasticum: genetics, clinical manifestations and therapeutic approaches. Surv Ophthalmol 54(2):272–285
Gorgels TG, Hu X, Scheffer GL, van der Wal AC, Toonstra J, de Jong PT et al (2005) Disruption of Abcc6 in the mouse: novel insight in the pathogenesis of pseudoxanthoma elasticum. Hum Mol Genet 14:1763–1773
Hu X, Plomp AS, van Soest S, Wijnholds J, de Jong PT, Bergen AA (2003) Pseudoxanthoma elasticum: a clinical, histopathological, and molecular update. Surv Ophthalmol 48(4):424–438
Clarkson JG, Altman RD (1982) Angioid streaks. Surv Ophthalmol 26(5):235–246
Dreyer R, Green WR (1978) The pathology of angioid streaks: a study of twenty-one cases. Trans Penn Acad Ophthalmol Otolaryngol 31(2):158–167
Booij JC, Bass DC, Beisekeeva J, Gorgels TG, Bergen AA (2010) The dynamic nature of Bruch’s membrane. Prog Retin Eye Res 29:1–18
Spaide RF, Koizumi H, Pozonni MC (2008) Enhanced depth imaging spectral-domain optical coherence tomography. Am J Ophthalmol 146(4):496–500
Margolis R, Spaide RF (2009) A pilot study of enhanced depth imaging optical coherence tomography of the choroid in normal eyes. Am J Ophthalmol 147:811–815
Nickla D, Wallman J (2010) The multifunctional choroid. Prog Retin Eye Res 29(2):144–168
Charbel Issa P, Finger RP, Götting C, Hending D, Holz FG, Scholl HP (2009) Centrifugal fundus abnormalities in pseudoxanthoma elasticum. Ophthalmology 117:1406–1414
Gliem M, Fimmers R, Müller PL, Brinkmann CK, Finger RP, Hending D et al (2014) Choroidal changes associated with Bruch membrane pathology in pseudoxanthoma elasticum. Am J Ophthalmol 158:198–207
Ellabban A, Tsujikawa A, Matsumoto A, Ogino K, Hangai M, Ooto S et al (2012) Macular choroidal thickness and volumen in eyes with angioid streaks measured by swept source optical coherence tomography. Am J Ophthalmol 153:1133–1143
Branchini L, Regatieri C, Flores-Moreno I, Baumann B, Fujimoto JG, Duker JS (2012) Reproducibility of choroidal thickness measurements across three spectral domain optical coherence tomography systems. Ophthalmology 119:119–123
Manjunath V, Taha M, Fujimoto JG, Duker JS (2010) Choroidal thickness in normal eyes measured using Cirrus HD optical coherence tomography. Am J Ophthalmol 150(3):325–329
Hirata M, Tsujikawa A, Matsumoto A, Hangai M, Ooto S, Yamashiro K et al (2011) Macular choroidal thickness and volumen in normal subjects measured by swept-source optical coherence tomography. Invest Ophthalmol Vis Sci 52:4971–4978
Tan CS, Ouyang Y, Ruiz H, Sadda SR (2012) Diurnal variation of choroidal thickness in normal, healthy subjects measured by spectral domain optical coherence tomography. Invest Ophthalmol Vis Sci 53(1):261–266
Sizmaz S, Küçükerdönmez C, Pinarci EY, Karalezli A, Canan H, Yilmaz G (2013) The effect of smoking on choroidal thickness measured by optical coherence tomography. Br J Ophthalmol 97(5):601–604
Sim DA, Keane PA, Mehta H, Fung S, Zarranz-Ventura J, Fruttiger M et al (2013) Repeatability and reproducibility of choroidal vessel layer measurements in diabetic retinopathy using enhanced depth optical coherence tomography. Invest Ophthalmol Vis Sci 54(4):2893–2901
Wolf-Schnurrbusch UE, Ceklic L, Brinkmann CK, Iliev ME, Frey M, Rothenbuehler SP et al (2009) Macular thickness measurements in healthy eyes using six different optical coherence tomography instruments. Invest Ophthalmol Vis Sci 50(7):3432–3477
Gass JD (1967) Pathogenesis of disciform detachment of the neuroepithelium. Am J Ophthalmol 63(Supp 1):1–139
Bock J (1938) Zur Klinik and anatomic der gefaBahnlichen Streifen im Augenhintergrund. Z Augenheilk 95(1/2):1–49
Verhoeff FH (1948) Histological finding in a case of angioid streaks. Br J Ophthalmol 32(9):531–544
Wilkelman JE (1948) Angioid streaks. Ophthalmologica 115(2):125
Rothwell R, Meira DM, Oliveira MA, Ribeiro LF, Fonseca SL (2015) Evaluation of choroidal thickness and volume during the third trimester of pregnancy using enhanced depth imaging optical coherence tomography: a pilot study. J Clin Diagn Res 9(8):NC08–NC11
Tittl MK, Spaide RF, Wong D et al (1999) Systemic findings associated with central serous chorioretinopathy. Am J Ophthalmol 128:63–68
Marmor MF, Wolfensberger TJ (1998) The retinal pigment epithelium. Oxford University Press, New York
Park J, Lee S, Son Y (2016) Effects of two different doses of intravitreal bevacizumab on subfoveal choroidal thickness and retinal vessel diameter in branch retinal vein occlusion. Int J Ophthalmol 9(7):999–1005
Tsuiki E, Suzuma K, Ueki R et al (2013) Enhanced depth imaging optical coherence tomography of the choroid in central retinal vein occlusion. Am J Ophthalmol 156(3):543–547
Yamazaki T, Koizumi H, Yamagishi T et al (2012) Subfoveal choroidal thickness after ranibizumab therapy for neovascular age-related macular degeneration: 12-month results. Ophthalmology 119(8):1621–1627
Koizumi H, Kano M, Yamamoto A et al (2015) Short-term changes in choroidal thickness after aflibercept therapy for neovascular age-related macular degeneration. Am J Ophthalmol 159(4):627–633
Ünlü C, Erdogan G, Gunay BO et al (2016) Subfoveal choroidal thickness changes after intravitreal bevacizumab injection for neovascular age-related macular degeneration and diabetic macular edema. Int Ophthalmol. 9:999 (epub ahead of print)
Tolentino MJ, McLeod DS, Taomoto M et al (2002) Pathologic features of vascular endothelial growth factor-induced retinopathy in the nonhuman primate. Am J Ophthalmol 133(3):373–385
Cao XS, Peng XY, You QS et al (2014) Subfoveal choroidal thickness change after intravitreal ranibizumab for idiopathic choroidal neovascularization. Retina 34(8):1554–1559
Booij JC, Baas DC, Beisekeeva J et al (2010) The dynamic nature of Bruch’s membrane. Prog Retin Eye Res 29(1):1–18
Sigler EJ, Randolph JC (2013) Comparison of macular choroidal thickness among patients older than age 65 with early atrophic age-related macular degeneration and normal. Invest Ophthalmol Vis Sci 54(9):6307–6313
Switzer DW Jr, Mendoca LS, Saito M et al (2012) Segregation of ophthalmoscopic characteristics according to choroidal thickness in patients with early age-related macular degeneration. Retina 32(7):1265–1271
Garcia-Fernandez MI, Gheduzzi D, Boraldi F, Paolinelli CD, Sanchez P, Valdivielso P et al (2008) Parameters of oxidative stress are present in the circulation of PXE patients 2008. Biochim Biophys Acta 1782(7–8):474–481
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hidalgo-Díaz, T., Morillo-Sánchez, M.J., Kamal-Salah, R. et al. Macular choroidal thickness in patients with pseudoxanthoma elasticum measured by enhanced-depth imaging spectral-domain optical coherence tomography. Int Ophthalmol 40, 1749–1758 (2020). https://doi.org/10.1007/s10792-020-01343-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-020-01343-2