Abstract
Purpose
To assess the ocular hypotensive effect of 15-keto fluprostenol, the oxidized metabolite of travoprost, on glaucoma patients, through a randomized double-masked placebo-controlled study.
Methods
Twelve patients with ocular normal tension glaucoma (NTG) (intraocular pressure [IOP] < 22 mmHg) were enrolled. In order to ensure patient compliance to treatment, all study subjects were hospitalized. In each patient, the eye to be submitted to the treatments was randomly chosen. After hospital admission (day 1), those patients received for 5 days at 8 P.M. either one drop of 15-keto fluprostenol (35 μg/ml) or one drop of placebo. IOP evaluation was performed within 8 A.M. and 8 P.M. for 6 days. Furthermore, we performed a determination of cardiovascular parameters before and after the treatments.
Results
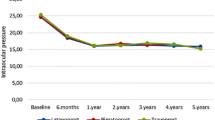
Starting with the first IOP measurement after the first treatment (8 A.M. on day 2), IOP was reduced of about 14% in the eyes treated 15-keto fluprostenol, in comparison with baseline IOP values of 15-keto fluprostenol-treated patients. The IOP reduction in the 15-keto fluprostenol-treated group was significantly compared to placebo group (p < 0.05) starting from day 3 till day 6 of the study. Except for mild hyperemia in one 15-keto fluprostenol-treated eye, no other side effects were observed or reported by the enrolled patients.
Conclusions
The travoprost metabolite 15-keto fluprostenol was effective in decrease IOP and maintained IOP reduction along 5 days of treatment. The 15-keto fluprostenol can be developed as a good candidate for once-a-day NTG patients’ treatment.


Similar content being viewed by others
References
Bucolo C, Platania CB, Reibaldi M, Bonfiglio V, Longo A, Salomone S, Drago F (2015) Controversies in glaucoma: current medical treatment and drug development. Curr Pharm Des 21(32):4673–4681
Coleman AL, Miglior S (2008) Risk factors for glaucoma onset and progression. Surv Ophthalmol 53(Suppl 1):S3–S10
Sommer A, Tielsch JM, Katz J, Quigley HA, Gottsch JD, Javitt J, Singh K (1991) Relationship between intraocular pressure and primary open angle glaucoma among white and black Americans. The Baltimore Eye Survey. Arch Ophthalmol 109:1090–1095
Tielsch JM, Katz J, Singh K, Quigley HA, Gottsch JD, Javitt J, Sommer A (1991) A population based evaluation of glaucoma screening: the Baltimore Eye Survey. Am J Epidemiol 134:1102–1110
The Collaborative Normal-tension Glaucoma Study Group (1998) Comparison of glaucomatous progression between untreated patients with normal-tension glaucoma and patients with therapeutically reduced intraocular pressures. Am J Ophthalmol 126:487–497
The Collaborative Normal-tension Glaucoma Study Group (1998) The effectiveness of intraocular pressure reduction in the treatment of normal tension glaucoma. Am J Ophthalmol 126:498–505
Lee AC, Mosaed S, Weinreb RN, Kripke DF, Liu JH (2007) Effect of laser trabeculoplasty on nocturnal intraocular pressure in medically treated glaucoma patients. Ophthalmology 114:666–670
Nagar M, Luhishi E, Shah N (2009) Intraocular pressure control and fluctuation: the effect of treatment with selective laser trabeculoplasty. Br J Ophthalmol 93:497–501
Heijl A, Leske MC, Hyman L, Yang Z, Bengtsson B, EMGT Group (2011) Intraocular pressure reduction with a fixed treatment protocol in the Early Manifest Glaucoma Trial. Acta Ophthalmol 89:749–754
Zacharia PT, Deppermann SR, Schuman JS (1993) Ocular hypotony after trabeculectomy with mitomycin C. Am J Ophthalmol 116:314–326
The Collaborative Normal-tension Glaucoma Study Group (1998) Comparison of glaucomatous progression between untreated patients with normal-tension glaucoma and patients with therapeutically reduced intraocular pressures. Collaborative Normal-Tension Glaucoma Study Group. Am J Ophthalmol 126(4):487–497
Heijl A, Leske MC, Bengtsson B, Hyman L, Hussein M (2002) Reduction of intraocular pressure and glaucoma progression: results from the Early Manifest Glaucoma Trial. Arch Ophthalmol 120(10):1268–1279
Zheng Y, Wong TY, Mitchell P, Friedman DS, He M, Aung T (2010) Distribution of ocular perfusion pressure and its relationship with open-angle glaucoma: the Singapore Malay eye study. Investig Ophthalmol Vis Sci 51(7):3399–3404
Hollows FC, Graham PA (1966) Intra-ocular pressure, glaucoma, and glaucoma suspects in a defined population. Br J Ophthalmol 50(10):570–586
Bucolo C, Platania CBM, Drago F, Reibaldi M, Bonfiglio V, Avitabile T, Uva M (2018) Novel therapeutics in glaucoma management. Curr Neuropharmacol 16(7):978–992
Bito LZ (1984) Comparison of the ocular hypotensive efficacy of eicosanoids and related compounds. Exp Eye Res 38(2):181–194
Resul B, Stjernschantz J, No K, Liljebris C, Selen G, Astin M, Karlsson M, Bito LZ (1993) Phenyl-substituted prostaglandins: potent and selective antiglaucoma agents. J Med Chem 36(2):243–248
Costagliola C, Del Prete A, Verolino M, Antinozzi P, Fusco R, Parmeggiani F, Mastropasqua L (2002) Effect of 0.005% latanoprost once daily on intraocular pressure in glaucomatous patients not adequately controlled by beta-blockers twice daily: a 3-year follow-up. Experience and incidence of side effects in a prospective study on 76 patients. Graefes Arch Clin Exp Ophthalmol 240(5):379–386
Camras CB, Schumer RA, Marsk A, Lustgarten JS, Serle JB, Stjernschantz J, Bito LZ, Podos SM (1992) Intraocular pressure reduction with PhXA34, a new prostaglandin analogue, in patients with ocular hypertension. Arch Ophthalmol 110(12):1733–1738
Toris CB, Camras CB, Yablonski ME (1993) Effects of PhXA41, a new prostaglandin F2 alpha analog, on aqueous humor dynamics in human eyes. Ophthalmology 100(9):1297–1304
Wang RF, Gagliuso DJ, Mittag TW, Podos SM (2007) Effect of 15-keto latanoprost on intraocular pressure and aqueous humor dynamics in monkey eyes. Investig Ophthalmol Vis Sci 48(9):4143–4147
Parrish RK, Palmberg P, Sheu WP, XLT Study Group (2003) A comparison of latanoprost, bimatoprost, and travoprost in patients with elevated intraocular pressure: a 12-week, randomized, masked-evaluator multicenter study. Am J Ophthalmol 135(5):688–703
Noecker RJ, Earl ML, Mundorf T, Peace J, Williams RD (2003) Bimatoprost 0.03% versus travoprost 0.004% in black Americans with glaucoma or ocular hypertension. Adv Ther 20(2):121–128
Cardascia N, Vetrugno M, Trabucco T, Cantatore F, Sborgia C (2003) Effects of travoprost eye drops on intraocular pressure and pulsatile ocular blood flow: a 180-day, randomized, double-masked comparison with ope-angle glaucoma. Curr Ther Res Clin Exp 64(7):389–400
Racz P, Ruzsonyi MR, Nagy ZT, Bito LZ (1993) Maintained intraocular pressure reduction with once-a-day application of a new prostaglandin F2 alpha analogue (PhXA41). An in-hospital, placebo-controlled study. Arch Ophthalmol 111(5):657–661
Costagliola C, Parmeggiani F, Ciancaglini M, D’Oronzo E, Mastropasqua L, Sebastiani A (2003) Ocular perfusion pressure and visual field indice modifications induced by alpha-agonist compound (clonidine 0.125%, apraclonidine 1.0% and brimonidine 0.2%) topical administration. An acute study on primary open-angle glaucoma patients. Ophthalmologica 217(1):39–44
Bito LZ (1989) A physiological approach to glaucoma management: the use of local hormones and the pharmacokinetics of prostaglandin esters. Prog Clin Biol Res 312:329–347
Bito LZ, Stjernschantz J, Resul B, Miranda OC, Basu S (1993) The ocular effects of prostaglandins and the therapeutic potential of a new PGF2 alpha analog, PhXA41 (latanoprost), for glaucoma management. J Lipid Mediat 6(1–3):535–543
Weinreb RN, Ong T, Scassellati Sforzolini B, Vittitow JL, Singh K, Kaufman PL (2015) A randomised, controlled comparison of latanoprostene bunod and latanoprost 0.005% in the treatment of ocular hypertension and open angle glaucoma: the VOYAGER study. Br J Ophthalmol 99(6):738–745
Sharif NA, Kelly CR, Crider JY, Williams GW, Xu SX (2003) Ocular hypotensive FP prostaglandin (PG) analogs: PG receptor subtype binding affinities and selectivities, and agonist potencies at FP and other PG receptors in cultured cells. J Ocul Pharmacol Ther 19(6):501–515
Abramovitz M, Adam M, Boie Y, Carriere M, Denis D, Godbout C, Lamontagne S, Rochette C, Sawyer N, Tremblay NM, Belley M, Gallant M, Dufresne C, Gareau Y, Ruel R, Juteau H, Labelle M, Ouimet N, Metters KM (2000) The utilization of recombinant prostanoid receptors to determine the affinities and selectivities of prostaglandins and related analogs. Biochem Biophys Acta 1483(2):285–293
Thieme H, Stumpff F, Ottlecz A, Percicot CL, Lambrou GN, Wiederholt M (2001) Mechanisms of action of unoprostone on trabecular meshwork contractility. Investig Ophthalmol Vis Sci 42(13):3193–3201
Cuppoletti J, Malinowska DH, Tewari KP, Chakrabarti J, Ueno R (2007) Cellular and molecular effects of unoprostone as a BK channel activator. Biochem Biophys Acta 1768(5):1083–1092
Thieme H, Steinhausen K, Ottlecz A, Lambrou GN, Strauss O, Wiederholt M, Rosenthal R (2005) Effects of unoprostone and endothelin 1 on L-type channel currents in human trabecular meshwork cells. Ophthalmic Res 37(6):293–300
Shimura M, Yasuda K, Nakzawa T, Kashiwagi K (2006) The effect of unoprostone isopropyl on Ca2+ release-activated Ca2+ currents in cultured monkey trabecular meshwork cells and ciliary muscle cells. J Ocul Pharmacol Ther 22(4):219–226
Hoban K, Peden R, Megaw R, Halpin P, Tatham AJ (2017) 24-hour contact lens sensor monitoring of intraocular pressure-related profiles in normal-tension glaucoma and rates of disease progression. Ophthalmic Res 57(4):208–215
Costagliola C, Parmeggiani F, Virgili G, Lamberti G, Incorvaia C, Perri P, Campa C, Sebastiani A (2008) Circadian changes of intraocular pressure and ocular perfusion pressure after timolol or latanoprost in Caucasians with normal-tension glaucoma. Graefe’s Arch Clin Exp Ophthalmol 246(3):389–396
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Human and animal rights
This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Caruso, C., Pacente, L., Troiano, P. et al. Intraocular pressure reduction with once-a-day application of a new prostaglandin eye drop: a pilot placebo-controlled study in 12 patients. Int Ophthalmol 40, 823–831 (2020). https://doi.org/10.1007/s10792-019-01243-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-019-01243-0