Abstract
Aims
To determine herpes simplex virus (HSV) DNA positivity in corneal scraping samples obtained from patients with microbial keratitis whose findings were not specific for HSV keratitis and to evaluate these particular cases with respect to clinical features and antiviral treatment results.
Methods
Records of patients with microbial keratitis treated in a tertiary eye care hospital within the 3-year period were evaluated retrospectively. Real-time polymerase chain reaction (PCR) was used to identify HSV DNA. Smear slides were evaluated by light microscopy. Patients with typical presentations and histories of HSV keratitis were excluded.
Results
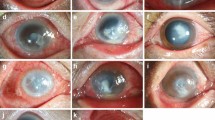
Two hundred and seventy-six eyes of 276 patients were included in the study. HSV-1 DNA was detected in 25 eyes (9%). In these 25 eyes, the initial diagnosis was fungal or bacterial keratitis. The mean symptom duration was 20 ± 14 days (2–60 days). The risk factors were ocular surgery (20%), blepharitis (16%), trauma (8%) and contact lens wear (4%); however, the majority of patients did not have any specific cause for keratitis (52%). Clinical features were variable and not typical for any particular etiology. Culture and microscopic examinations revealed bacteria and/or fungi in 6 patients in addition to herpes infection. Antiviral treatment was successful in 72% of patients.
Conclusion
Herpetic corneal infections can present without typical dendritic or geographic ulcers and may be masked by other infections. Real-time PCR is a useful method for rapid and definitive diagnosis. HSV infection should be considered for microbial keratitis without specific risk factors, with negative culture results and poor response to antimicrobial agents.


Similar content being viewed by others
References
Bradley H, Markowitz LE, Gibson T et al (2014) Seroprevalence of herpes simplex virus types 1 and 2: United States, 1999–2010. J Infect Dis 209:325–333
Whitley RJ, Roizman B (2001) Herpes simplex virus infections. Lancet 357:1513–1518
Tsatsos M, MacGregor C, Athanasiadis I et al (2016) Herpes simplex virus keratitis: an update of the pathogenesis and current treatment with oral and topical antiviral agents. Clin Exp Ophthalmol 44:824–837
Beyer CF, Arens MQ, Hill JM et al (1989) Penetrating keratoplasty in rabbits induces latent HSV-1 reactivation when corticosteroids are used. Curr Eye Res 8:1323–1329
Hudu SA, Alshrari AS, Syahida A et al (2016) Cell culture, technology: enhancing the culture of diagnosing human diseases. J Clin Diagn Res 10:1–5
Hwang KA, Ahn JH, Nam JH (2018) Diagnosis of viral infection using real-time polymerase chain reaction. J Bacteriol Virol 48:1–13
Azher TN, Yin XT, Tajfirouz D et al (2017) Herpes simplex keratitis: challenges in diagnosis and clinical management. Clin Ophthalmol 11:185–191
Subhan S, Jose RJ, Duggirala A et al (2004) Diagnosis of herpes simplex virus-1 keratitis: comparison of Giemsa stain, immunofluorescence assay and polymerase chain reaction. Curr Eye Res 29:209–213
Farhatullah S, Kaza S, Athmanathan S, Garg P et al (2004) Diagnosis of herpes simplex virus-1 keratitis using Giemsa stain, immunofluorescence assay, and polymerase chain reaction assay on corneal scrapings. Br J Ophthalmol 88:142–144
Barrado L, Suarez MJ, Pérez-Blázquez E et al (2014) Could polymerase chain reaction tests on conjunctival swabs be useful to diagnose herpetic keratitis? Enferm Infecc Microbiol Clin 32:28–30
El-Aal AM, El Sayed M, Mohammed E et al (2006) Evaluation of herpes simplex detection in corneal scrapings by three molecular methods. Curr Microbiol 52:379–382
Taravati P, Lam D, Van Gelder RN (2013) Role of molecular diagnostics in ocular microbiology. Curr Ophthalmol Rep 1:1–13
Satpathy G, Behera HS, Sharma A et al (2018) A 20-year experience of ocular herpes virus detection using immunofluorescence and polymerase chain reaction. Clin Exp Optom 101:648–651
Mackay M (2004) Real-time PCR in the microbiology laboratory. Clin Microbiol Infect 10:190–212
Kalezic T, Mazen M, Kuklinski E et al (2018) Herpetic eye disease study: lesson learned. Curr Opin Ophthalmol 29:340–346
Koizumi N, Nishida K, Adachi W et al (1999) Detection of herpes simplex virus DNA in atypical epithelial keratitis using polymerase chain reaction. Br J Ophthalmol 83:957–960
Tei M, Nishida K, Kinoshita S (1996) Polymerase chain reaction of herpes simplex virus in tear fluid from atypical herpetic epithelial keratitis after penetrating keratoplasty. Am J Ophthalmol 122:732–735
Garweg JG, Russ CE, Shellhorn M et al (2003) HSV-1 antigens and DNA in the corneal explant buttons of patients with non-herpetic or clinically atypical herpetic stromal keratitis. Graefe’s Arch Clin Exp Ophthalmol 241:734–739
Kaye S, Choudhary A (2006) Herpes simplex keratitis. Prog Retin Eye Res 25:355–380
Funding
This study was funded by government.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest for this study.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Erdem, E., Harbiyeli, İ.İ., Öztürk, G. et al. Atypical herpes simplex keratitis: frequency, clinical presentations and treatment results. Int Ophthalmol 40, 659–665 (2020). https://doi.org/10.1007/s10792-019-01226-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-019-01226-1