Abstract
Purpose
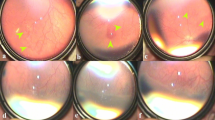
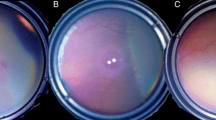
To compare type I retinopathy of prematurity (ROP) with aggressive posterior retinopathy of prematurity (AP-ROP) regarding risk factors, complications and treatment outcomes.
Methods
A prospective cohort approach was applied on premature newborns diagnosed as type I ROP and AP-ROP. An intravitreal injection of bevacizumab (Avastin®) was given to each patient. Demographic features such as gestational age, birth weight, age at the initial injection, involved eye, treatment response, relapses and need to extra interventions were compared.
Results
Seventy-seven patients underwent an initial intravitreal injection of bevacizumab (IVB), and 108 eyes were type I ROP and 30 eyes were AP-ROP. There was no significant difference in birth weight and gestational age between two groups. ROP relapsed in eight eyes of four patients with AP-ROP (26.6%), of which six eyes of three patients received re-injection of bevacizumab, and two eyes of one patient underwent a laser treatment. Recurrence occurred in two eyes of one patient with type I ROP (1.8%), which were treated by laser.
Conclusion
Bilateral eye involvement, relapse and retreatment (IVB, laser and surgical intervention) are more frequent in AP-ROP than type I ROP even when treated with intravitreal bevacizumab injection.

Similar content being viewed by others
References
Martinez-Castellanos MA, Schwartz S, Hernandez-Rojas ML et al (2013) Long-term effect of antiangiogenic therapy for retinopathy of prematurity up to 5 years of follow-up. Retina 33(2):329–338
Terry TL (1944) Retrolental fibroplasia in the premature infant: V. Further studies on fibroplastic overgrowth of the persistent tunica vasculosa lentis. Trans Am Ophthalmol Soc. 42:383–396
Mutlu FM, Sarici SU (2013) Treatment of retinopathy of prematurity: a review of conventional and promising new therapeutic options. Int J Ophthalmol 6(2):228–236
Early Treatment for Retinopathy of Prematurity Cooperative Group (2003) Revised indications for the treatment of retinopathy of prematurity: results of the early treatment for retinopathy of prematurity randomized trial. Arch Ophthalmol (Chicago, Ill 1960) 121(12):1684–1694
Soejima N, Takagi I, Takashima Y (1976) Clinical studies on retinopathy of prematurity, severe and rapidly progressive type. Folia Ophthalmol Jpn 27(1):155–161
Aaberg T, Ben-Sira I, Charles S, Clarkson J, Cohen BZ, Flynn J, Foos R, Garner A, Hirose T, Koerner F, Machemer R (1987) An international classification of retinopathy of prematurity. II. The classification of retinal detachment. The International Committee for the Classification of the Late Stages of Retinopathy of Prematurity. Arch Ophthalmol (Chicago, Ill 1960) 105(7):906–912
International Committee for the Classification of Retinopathy of Prematurity (2005) The international classification of retinopathy of prematurity revisited. Arch Ophthalmol 123(7):991–999. https://doi.org/10.1001/archopht.123.7.991
Gilbert C (2008) Retinopathy of prematurity: a global perspective of the epidemics, population of babies at risk and implications for control. Early Hum Dev 84(2):77–82
Feng J, Qian J, Jiang Y et al (2017) Efficacy of primary intravitreal ranibizumab for retinopathy of prematurity in China. Ophthalmology 124(3):408–409
Wong RK, Hubschman S, Tsui I (2015) Reactivation of retinopathy of prematurity after ranibizumab treatment. Retina. 35(4):675–680
Erol MK, Coban DT, Sari ES et al (2015) Comparison of intravitreal ranibizumab and bevacizumab treatment for retinopathy of prematurity. Arq Bras Oftalmol 78(6):340–343
Khodabande A, Niyousha MR, Roohipoor R (2016) A lower dose of intravitreal bevacizumab effectively treats retinopathy of prematurity. J AAPOS 20:490–492
Darlow BA, Hutchinson JL, Henderson-Smart DJ, Donoghue DA, Simpson JM, Evans NJ (2005) Prenatal risk factors for severe retinopathy of prematurity among very preterm infants of the Australian and New Zealand Neonatal Network. Pediatrics 115(4):990–996
Woo SJ, Park KH, Ahn J et al (2011) A co-twin study of the relative effect of birth weight and gestational age on retinopathy of prematurity. Eye (London) 25(11):1478–1483
Dhaliwal CA, Fleck BW, Wright E, Graham C, McIntosh N (2009) Retinopathy of prematurity in small-for-gestational age infants compared with those of appropriate size for gestational age. Arch Dis Child Fetal Neonatal Ed 94(3):F193–F195
Sanghi G, Dogra MR, Das P, Vinekar A, Gupta A, Dutta S (2009) Aggressive posterior retinopathy of prematurity in Asian Indian babies: spectrum of disease and outcome after laser treatment. Retina 29:1335–1339
Nicoară SD, Ştefănuţ AC, Nascutzy C, Zaharie GC, Toader LE, Drugan TC (2016) Regression rates following the treatment of aggressive posterior retinopathy of prematurity with bevacizumab versus laser: 8-year retrospective analysis. Med Sci Monit 22:1192–1209
Li Xiu-Juan, Yang Xiao-Peng, Sun Shuang, Lyu Xiao-Bei, Jia Heng (2016) Intravitreal ranibizumab for aggressive posterior retinopathy of prematurity. Chin Med J (Engl) 129(23):2879–2881
Chen S-N, Lian I, Hwang Y-C et al (2015) Intravitreal anti-vascular endothelial growth factor treatment for retinopathy of prematurity: comparison between ranibizumab and bevacizumab. Retina 35(4):667–674
Castellanos MAM, Schwartz S, Garcia-Aguirre G, Quiroz-Mercado H (2013) Short-term outcome after intravitreal ranibizumab injections for the treatment of retinopathy of prematurity. Br J Ophthalmol 97(7):816–819
Sukgen EA, Koçluk Y (2017) The vascularization process after intravitreal ranibizumab injections for aggressive posterior retinopathy of prematurity. Arq Bras Oftalmol 80(1):30–34
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no grant and no conflict of interest for all authors.
Ethical approval
This study was conducted based on principles of the Declaration of Helsinki. The study protocol was supervised and approved by the Ethical Committee of Tabriz University of Medical Sciences.
Inform consent
The written inform consent was received from all the participants’ legal parents.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eftekhari Milani, A., Hassanpoor, N., Mousavi Mirkala, M. et al. Intravitreal bevacizumab injection in aggressive posterior retinopathy of prematurity compared with type I retinopathy of prematurity. Int Ophthalmol 40, 477–482 (2020). https://doi.org/10.1007/s10792-019-01208-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-019-01208-3