Abstract
Purpose
To analyze long-term changes in individual retinal layers (RLs) after intravitreal injections of ranibizumab (IVRs) in patients with neovascular age-related macular degeneration (n-AMD).
Methods
The patients were treated with 0.5-mg IVRs based on an as-needed protocol after the first three monthly doses over a 12-month follow-up period. Patients underwent optical coherence tomography and best-corrected visual acuity (BCVA) evaluation at each visit. The ETDRS grid with central subfield (R1) (r 0.5 mm) and the inner ring (R2) (r 0.5–1.5 mm) was used for calculation of the mean thickness of each RL. Changes in the thickness of segmented RLs within the R1 and R2 of ETDRS circles at months-3, -6, and -12 were compared to baseline.
Results
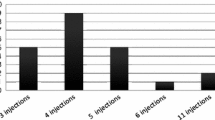
The mean age was 72 ± 7.4 years. The mean number of injections was 9.08 (range 6–11). Mean BCVA improved from 49.7 ± 22.1 to 60.1 ± 19.8 letters. Central macular thickness decreased from 390.25 ± 149.6 to 312.74 ± 118.4 μm. Thicknesses of GCL (from 23.93 ± 13.73 to 19.50 ± 9.50 μm in R1; p 0.001, and from 44.5 ± 12.6 to 39.6 ± 10.6 μm in R2; p 0.005), IPL (from 28.90 ± 14.36 to 22.35 ± 6.23 μm in R1; p 0.001, and from 39.34 ± 8.53 to 35.58 ± 7.93 μm in R2; p 0.004), and total inner RL (ILM to ELM) (from 222.93 ± 93.09 to 180 ± 53 μm in R1; p 0.001, and from 255.06 ± 42.74 to 240.25 ± 40.37 μm in R2; p 0.003) in the central and parafoveal rings decreased statistically at month-12. Decrease in INL was limited to month-6 (from 34.80 ± 15.33 to 27.60 ± 12.59 μm in R1; p 0.001), while decreases in total outer RLs (ELM to RPE) (from 128.32 ± 26.92 to 115.54 ± 43.98 μm in R1; p 0.001, and 103.81 ± 16.73 to 96.38 ± 16.22 μm in R2; p 0.014) and RPE (from 39.12 ± 22.33 to 29.70 ± 22.05 μm in R1; p 0.001, and from 31.27 ± 13.11 to 24.40 ± 9.99 μm in R2; p 0.001) were limited to month-3.
Conclusions
Significant changes were observed in the thickness of the inner RLs after 1-year treatment with IVRs for n-AMD. A significant decrease in RPE thickness confined to the first months disappeared at month-12.
Similar content being viewed by others
References
Holz FG, Pauleikhoff D, Klein R, Bird AC (2004) Pathogenesis of lesions in late age-related macular disease. Am J Ophthalmol 137:504–510
Ferrara N, Adamis AP (2016) Ten years of anti-vascular endothelial growth factor therapy. Nat Rev Drug Discov 15:385–403
Ferrara N, Damico L, Shams N, Lowman H, Kim R (2006) Development of ranibizumab, an anti-vascular endothelial growth factor antigen binding fragment, as therapy for neovascular age-related macular degeneration. Retina 26:859–870
Chong V (2016) Ranibizumab for the treatment of wet AMD: a summary of real-world studies. Eye (Lond) 30:270–286
Nishijima K, Ng YS, Zhong L et al (2007) Vascular endothelial growth factor-A is a survival factor for retinal neurons and a critical neuroprotectant during the adaptive response to ischemic injury. Am J Pathol 171:53–67
Storkebaum E, Lambrechts D, Carmeliet P (2004) VEGF: once regarded as a specific angiogenic factor, now implicated in neuroprotection. BioEssays 26:943–954
Mordenti J, Cuthbertson RA, Ferrara N et al (1999) Comparisons of the intraocular tissue distribution, pharmacokinetics, and safety of 125I-labeled full-length and Fab antibodies in rhesus monkeys following intravitreal administration. Toxicol Pathol 27:536–544
Bakri SJ, McCannel CA, Edwards AO, Moshfeghi DM (2008) Persistent ocular hypertension following intravitreal ranibizumab. Graefes Arch Clin Exp Ophthalmol 246:955–958
Gismondi M, Salati C, Salvetat ML, Zeppieri M, Brusini P (2009) Short-term effect of intravitreal injection of ranibizumab (Lucentis) on intraocular pressure. J Glaucoma 18:658–661
Inan UU, Avci B, Kusbeci T, Kaderli B, Avci R, Temel SG (2007) Preclinical safety evaluation of intravitreal injection of full-length humanized vascular endothelial growth factor antibody in rabbit eyes. Invest Ophthalmol Vis Sci 48:1773–1781
Zayit-Soudry S, Zemel E, Loewenstein A, Perlman I (2010) Safety evaluation of repeated intravitreal injections of bevacizumab and ranibizumab in rabbit eyes. Retina 30:671–681
Nishimura T, Machida S, Harada T, Kurosaka D (2012) Retinal ganglion cell function after repeated intravitreal injections of ranibizumab in patients with age-related macular degeneration. Clin Ophthalmol 6:1073–1082
Ho AC, Busbee BG, Regillo CD et al (2014) Twenty-four-month efficacy and safety of 0.5 mg or 2.0 mg ranibizumab in patients with subfoveal neovascular age-related macular degeneration. Ophthalmology 121:2181–2192
Day S, Acquah K, Mruthyunjaya P, Grossman DS, Lee PP, Sloan FA (2011) Ocular complications after anti-vascular endothelial growth factor therapy in Medicare patients with age-related macular degeneration. Am J Ophthalmol 152:266–272
Kuroda Y, Yamashiro K, Tsujikawa A et al (2016) Retinal pigment epithelial atrophy in neovascular age-related macular degeneration after ranibizumab treatment. Am J Ophthalmol 161:94–103
Loduca AL, Zhang C, Zelkha R, Shahidi M (2010) Thickness mapping of retinal layers by spectral-domain optical coherence tomography. Am J Ophthalmol 150:849–855
Oberwahrenbrock T, Weinhold M, Mikolajczak J et al (2015) Reliability of intra-retinal layer thickness estimates. PLoS ONE 10:e0137316
Martinez-de-la-Casa JM, Ruiz-Calvo A, Saenz-Frances F et al (2012) Retinal nerve fiber layer thickness changes in patients with age-related macular degeneration treated with intravitreal ranibizumab. Invest Ophthalmol Vis Sci 53:6214–6218
Horsley MB, Mandava N, Maycotte MA, Kahook MY (2010) Retinal nerve fiber layer thickness in patients receiving chronic anti-vascular endothelial growth factor therapy. Am J Ophthalmol 150:558–561
Rimayanti U, Kiuchi Y, Yamane K et al (2014) Inner retinal layer comparisons of eyes with exudative age-related macular degeneration and eyes with age-related macular degeneration and glaucoma. Graefes Arch Clin Exp Ophthalmol 252:563–570
Entezari M, Ramezani A, Yaseri M (2014) Changes in retinal nerve fiber layer thickness after two intravitreal bevacizumab injections for wet type age-related macular degeneration. J Ophthalmic Vis Res 9:449–452
Parlak M, Oner FH, Saatci AO (2015) The long-term effect of intravitreal ranibizumab on retinal nerve fiber layer thickness in exudative age-related macular degeneration. Int Ophthalmol 35:473–480
Shin HJ, Kim SN, Chung H, Kim TE, Kim HC (2016) Intravitreal anti-vascular endothelial growth factor therapy and retinal nerve fiber layer loss in eyes with age-related macular degeneration: a meta-analysis. Invest Ophthalmol Vis Sci 57:1798–1806
Zucchiatti I, Parodi MB, Pierro L et al (2015) Macular ganglion cell complex and retinal nerve fiber layer comparison in different stages of age-related macular degeneration. Am J Ophthalmol 160:602–607
Hondur G, Göktaş E, Al-Aswad L, Tezel G (2018) Age-related changes in the peripheral retinal nerve fiber layer thickness. Clin Ophthalmol 12:401–409
Park HY, Kim JH, Park CK (2014) Neuronal cell death in the inner retina and the influence of vascular endothelial growth factor inhibition in a diabetic rat model. Am J Pathol 184:1752–1762
Brar VS, Sharma RK, Murthy RK, Chalam KV (2010) Bevacizumab neutralizes the protective effect of vascular endothelial growth factor on retinal ganglion cells. Mol Vis 16:1848–1853
Beck M, Munk MR, Ebneter A, Wolf S, Zinkernagel MS (2016) Retinal ganglion cell layer change in patients treated with anti-vascular endothelial growth factor for neovascular age-related macular degeneration. Am J Ophthalmol 167:10–17
Hollo G, Naghizadeh F (2015) Influence of a new software version of the RTVue-100 optical coherence tomography on ganglion cell complex segmentation in various forms of age-related macular degeneration. J Glaucoma 24:245–250
Perdicchi A, Peluso G, Iacovello D et al (2015) Ganglion cell complex evaluation in exudative age-related macular degeneration after repeated intravitreal injections of ranibizumab. Biomed Res Int 2015:268796
Kashiwagi K, Iizuka Y, Tanaka Y, Araie M, Suzuki Y, Tsukahara S (2004) Molecular and cellular reactions of retinal ganglion cells and retinal glial cells under centrifugal force loading. Invest Ophthalmol Vis Sci 45:3778–3786
Villegas-Perez MP, Lawrence JM, Vidal-Sanz M, Lavail MM, Lund RD (1998) Ganglion cell loss in RCS rat retina: a result of compression of axons by contracting intraretinal vessels linked to the pigment epithelium. J Comp Neurol 392:58–77
Ramirez JM, Ramirez AI, Salazar JJ, de Hoz R, Trivino A (2001) Changes of astrocytes in retinal ageing and age-related macular degeneration. Exp Eye Res 73:601–615
Savastano MC, Minnella AM, Tamburrino A, Giovinco G, Ventre S, Falsini B (2014) Differential vulnerability of retinal layers to early age-related macular degeneration: evidence by SD-OCT segmentation analysis. Invest Ophthalmol Vis Sci 55:560–566
Huo YJ, Guo Y, Li L, Wang HZ, Wang YX, Thomas R, Wang NL (2018) Age-related changes in and determinants of macular ganglion cell-inner plexiform layer thickness in normal Chinese adults. Clin Exp Ophthalmol 46:400–406
Ebneter A, Wolf S, Abhishek J, Zinkernagel MS (2016) Retinal layer response to ranibizumab during treatment of diabetic macular edema: thinner is not always better. Retina 36:1314–1323
Yu DY, Cringle SJ (2001) Oxygen distribution and consumption within the retina in vascularised and avascular retinas and in animal models of retinal disease. Prog Retin Eye Res 20:175–208
Holfort SK, Klemp K, Kofoed PK, Sander B, Larsen M (2010) Scotopic electrophysiology of the retina during transient hyperglycemia in type 2 diabetes. Invest Ophthalmol Vis Sci 51:2790–2794
Schutze C, Wedl M, Baumann B, Pircher M, Hitzenberger CK, Schmidt-Erfurth U (2015) Progression of retinal pigment epithelial atrophy in antiangiogenic therapy of neovascular age-related macular degeneration. Am J Ophthalmol 159:1100–1114
Funding
This study was supported in part by a Grant from Afyon Kocatepe University Scientific Research Projects Committee with Project No. 14.TUS.12.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in the study involving human participants were in accordance with the ethical standards of the institutional committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Ümit Übeyt Inan, Zeki Baysal, Sibel Inan are formerly at Kocatepe University Medical School Department of Ophthalmology, Afyon, Turkey.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure
1. Demonstration of retinal segmentation in a patient with neovascular age-related macular degeneration at baseline (top) and month-12 after intravitreal ranibizumab treatment (bottom). Segmentation of retinal layers in a representative case. (JPEG 18536 kb)
Supplemental Digital Content 2:
Supplemental Table 1. RNFL and GCL.doc: Baseline Values and Change during follow-up in the thicknesses of RNFL and GCL (DOCX 15 kb)
Supplemental Digital Content 3:
Supplemental Table 2. IPL and INL.doc: Baseline Values and Change during follow-up in the thicknesses of IPL and INL (DOCX 16 kb)
Supplemental Digital Content 4:
Supplemental Table 3. OPL and ONL.doc: Baseline Values and Change during follow-up in the thicknesses of OPL and ONL (DOCX 15 kb)
Supplemental Digital Content 5:
Supplemental Table 4. IRLs and ORL.doc: Baseline Values and Change during follow-up in the thicknesses of IRL and ORL (DOCX 16 kb)
Supplemental Digital Content 6:
Supplemental Table 5. RPE.doc: Baseline Values and Change during follow-up in the thicknesses of RPE (DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Inan, Ü.Ü., Baysal, Z. & Inan, S. Long-term changes in retinal layers in patients undergoing intravitreal ranibizumab for neovascular age-related macular degeneration. Int Ophthalmol 39, 2721–2730 (2019). https://doi.org/10.1007/s10792-019-01116-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10792-019-01116-6