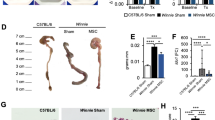
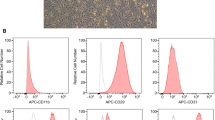
Abstract
Inflammatory bowel diseases (IBDs) are prevalent and debilitating diseases with limited clinical treatment strategies. Mesenchymal stem cell (MSCs) are pluripotent stem cells with self-renewal capability and multiple immunomodulatory effects, which make them a promising therapeutic approach for IBDs. Thus, optimization of MSCs regimes is crucial for their further clinical application. Wogonin, a flavonoid-like compound with extensive immunomodulatory and adjuvant effects, has been investigated as a potential pretreatment for MSCs in IBD treatment. In this study, we employed the DSS-induced acute colitis mouse model to compare the therapeutic effectiveness of MSCs in pretreated with or without wogonin and further explore the underlying mechanism. Compared to untreated MSCs, MSCwogonin (pretreated with wogonin) showed greater effectiveness in the treatment of colitis. Further experiments revealed that wogonin treatment activated the AKT signaling pathway, resulting in higher cellular glycolysis. Inhibition of AKT phosphorylation by perifosine not only decreased glycolysis but impaired the therapeutic efficiency of MSCwogonin. Consistent with these results, qPCR data indicated that wogonin treatment induced the expression of immunomodulatory molecules IL-10, IDO, and AGR1, which were reduced by perifosine. Together, our data demonstrated that wogonin preconditioning strategy further augmented the therapeutic efficacy of MSCs via promoting glycolysis, which should be a promising strategy for optimizing MSCs therapy in IBDs.







Similar content being viewed by others
Data availability
The datasets generated or analyzed for the present study are available from the corresponding author upon reasonable request.
References
Andrzejewska A, Lukomska B, Janowski M (2019) Concise review: mesenchymal stem cells: from roots to boost. Stem Cells (Dayton, Ohio) 37:855–864. https://doi.org/10.1002/stem.3016
Cabalzar-Wondberg D, Turina M, Biedermann L et al (2021) Allogeneic expanded adipose-derived mesenchymal stem cell therapy for perianal fistulas in Crohn’s disease: a case series. Colorectal Dis Off J Assoc Coloproctol Great Br Irel 23:1444–1450. https://doi.org/10.1111/codi.15587
Cheng W, Su J, Hu Y et al (2017) Interleukin-25 primed mesenchymal stem cells achieve better therapeutic effects on dextran sulfate sodium-induced colitis via inhibiting Th17 immune response and inducing T regulatory cell phenotype. Am J Transl Res 9:4149–4160
Danese S, Solitano V, Jairath V et al (2022) The future of drug development for inflammatory bowel disease: the need to ACT (advanced combination treatment). Gut 71:2380–2387. https://doi.org/10.1136/gutjnl-2022-327025
De Souza HS, Fiocchi C (2016) Immunopathogenesis of IBD: current state of the art. Nat Rev Gastroenterol Hepatol 13:13–27. https://doi.org/10.1038/nrgastro.2015.186
Fan H, Zhao G, Liu L et al (2012) Pre-treatment with IL-1beta enhances the efficacy of MSC transplantation in DSS-induced colitis. Cell Mol Immunol 9:81–473. https://doi.org/10.1038/cmi.2012.40
Filipescu IE, Leonardi L, Menchetti L et al (2018) Preventive effects of bovine colostrum supplementation in TNBS-induced colitis in mice. PLoS One 13:e0202929. https://doi.org/10.1371/journal.pone.0202929
Forbes GM, Sturm MJ, Leong RW et al (2014) A phase 2 study of allogeneic mesenchymal stromal cells for luminal Crohn’s disease refractory to biologic therapy. Clin Gastroenterol Hepatol 12:64–71. https://doi.org/10.1016/j.cgh.2013.06.021
García-olmo D, García-arranz M, Herreros D et al (2005) A phase I clinical trial of the treatment of Crohn’s fistula by adipose mesenchymal stem cell transplantation. Dis Colon Rectum 48:1416–1423. https://doi.org/10.1007/s10350-005-0052-6
Gergely M, Prado E, Deepak P (2022) Management of refractory inflammatory bowel disease. Curr Opin Gastroenterol 38:347–357. https://doi.org/10.1097/MOG.0000000000000849
Gills JJ, Dennis PA (2009) Perifosine: update on a novel Akt inhibitor. Curr Oncol Rep 11:102–110. https://doi.org/10.1007/s11912-009-0016-4
Gubser PM, Bantug GR, Razik L et al (2013) Rapid effector function of memory CD8+ T cells requires an immediate-early glycolytic switch. Nat Immunol 14:72–1064. https://doi.org/10.1038/ni.2687
He J, Zhang N, Zhu Y et al (2021) MSC spheroids-loaded collagen hydrogels simultaneously promote neuronal differentiation and suppress inflammatory reaction through PI3K-Akt signaling pathway. Biomaterials 265:120448. https://doi.org/10.1016/j.biomaterials.2020.120448
Hu X, Xu Q, Wan H et al (2020) PI3K-Akt-mTOR/PFKFB3 pathway mediated lung fibroblast aerobic glycolysis and collagen synthesis in lipopolysaccharide-induced pulmonary fibrosis. Lab Investig J Tech Methods Pathol 100:801–811. https://doi.org/10.1038/s41374-020-0404-9
Huldani H, Margiana R, Ahmad F et al (2022) Immunotherapy of inflammatory bowel disease (IBD) through mesenchymal stem cells. Int Immunopharmacol 107:108698. https://doi.org/10.1016/j.intimp.2022.108698
Huynh DL, Ngau TH, Nguyen NH et al (2020) Potential therapeutic and pharmacological effects of Wogonin: an updated review. Mol Biol Rep 47:9779–9789. https://doi.org/10.1007/s11033-020-05972-9
Jones N, Vincent EE, Cronin JG et al (2019) Akt and STAT5 mediate naive human CD4+ T-cell early metabolic response to TCR stimulation. Nat Commun 10:2042. https://doi.org/10.1038/s41467-019-10023-4
Killer MC, Nold P, Henkenius K et al (2017) Immunosuppressive capacity of mesenchymal stem cells correlates with metabolic activity and can be enhanced by valproic acid. Stem Cell Res Ther 8:100. https://doi.org/10.1186/s13287-017-0553-y
Li M, Zhang YX, Zhang Z et al (2015) Endomicroscopy will track injected mesenchymal stem cells in rat colitis models. Inflamm Bowel Dis 21:77–2068. https://doi.org/10.1097/MIB.0000000000000458
Lin H, Li C, Qian S et al (2022) Evaluation of the biological properties of human umbilical cord mesenchymal stem cells in difffferent passages. J Histochem Cytochem 31:341–350. https://doi.org/10.16705/j.cnki.1004-1850.2022.04.003
Liu W, Yu M, Xie D et al (2020) Melatonin-stimulated MSC-derived exosomes improve diabetic wound healing through regulating macrophage M1 and M2 polarization by targeting the PTEN/AKT pathway. Stem Cell Res Ther 11:259. https://doi.org/10.1186/s13287-020-01756-x
Lügering A, Schmidt M, Lügering N et al (2001) Infliximab induces apoptosis in monocytes from patients with chronic active Crohn’s disease by using a caspase-dependent pathway. Gastroenterology 121(5):1145–1157. https://doi.org/10.1053/gast.2001.28702
Manning BD, Toker A (2017) AKT/PKB signaling: navigating the network. Cell 169:381–405. https://doi.org/10.1016/j.cell.2017.04.001
Markov A, Thangavelu L, Aravindhan S et al (2021) Mesenchymal stem/stromal cells as a valuable source for the treatment of immune-mediated disorders. Stem Cell Res Ther 12:192. https://doi.org/10.1186/s13287-021-02265-1
Menk AV, Scharping NE, Moreci RS et al (2018) Early TCR signaling induces rapid aerobic glycolysis enabling distinct acute T cell effector functions. Cell Rep 22:1509–1521. https://doi.org/10.1016/j.celrep.2018.01.040
Molendijk I, Bonsing BA, Roelofs H et al (2015) Allogeneic bone marrow-derived mesenchymal stromal cells promote healing of refractory perianal fistulas in patients with Crohn’s disease. Gastroenterology 149:918–27.e6. https://doi.org/10.1053/j.gastro.2015.06.014
Mrahleh MA, Matar S, Jafar H et al (2021) Human Wharton’s Jelly-derived mesenchymal stromal cells primed by tumor necrosis factor-α and interferon-γ modulate the innate and adaptive immune cells of type 1 diabetic patients. Front Immunol 12:732549. https://doi.org/10.3389/fimmu.2021.732549
Pan T, Sun S, Chen Y et al (2022) Immune effects of PI3K/Akt/HIF-1alpha-regulated glycolysis in polymorphonuclear neutrophils during sepsis. Crit Care (Lond, Engl) 26:29. https://doi.org/10.1186/s13054-022-0389
Ren W, Chai M, Jiang M et al (2022) High glucose mediates apoptosis and osteogenesis of MSCs via downregulation of AKT-Sirt1-TWIST. Mol Biol Rep 49:2723–2733. https://doi.org/10.1007/s11033-021-07082-6
Ryu DB, Lim JY, Lee SE et al (2016) Induction of Indoleamine 2,3-dioxygenase by pre-treatment with Poly(I:C) may enhance the efficacy of MSC treatment in DSS-induced colitis. Immune Netw 16:358–365. https://doi.org/10.4110/in.2016.16.6.358
Strober W, Fuss IJ (2011) Proinflammatory cytokines in the pathogenesis of inflammatory bowel diseases. Gastroenterology 140(6):1756–1767. https://doi.org/10.1053/j.gastro.2011.02.016
Wang W, Xia T, Yu X (2015) Wogonin suppresses inflammatory response and maintains intestinal barrier function via TLR4-MyD88-TAK1-mediated NF-kappaB pathway in vitro. Inflamm Res Off J Eur Histamine Res Soc 64:31–423. https://doi.org/10.1007/s00011-015-0822-0
Wu Q, Xie S, Zhu Y et al (2021) Wogonin strengthens the therapeutic effects of mesenchymal stem cells in dss-induced colitis via promoting IL-10 production. Oxid Med Cell Longev 2021:5527935. https://doi.org/10.1155/2021/5527935
Xu C, Feng C, Huang P et al (2022) TNFalpha and IFNgamma rapidly activate PI3K-AKT signaling to drive glycolysis that confers mesenchymal stem cells enhanced anti-inflammatory property. Stem Cell Res Ther 13:491. https://doi.org/10.1186/s13287-022-03178-3
Zhang X, Wang S, Ding X et al (2020) Potential methods for improving the efficacy of mesenchymal stem cells in the treatment of inflammatory bowel diseases. Scand J Immunol 92:e12897. https://doi.org/10.1111/sji.12897
Zhou Y, Dou F, Song H et al (2022) Anti-ulcerative effects of wogonin on ulcerative colitis induced by dextran sulfate sodium via Nrf2/TLR4/NF-kappaB signaling pathway in BALB/c mice. Environ Toxicol 37:954–963. https://doi.org/10.1002/tox.23457
Funding
This work is supported by the National Natural Science Foundation of China (NO. 82170216), Fundamental and Applied Basic Research of Guangzhou City-University (Institution) Projects (NO. 202201020461), Science and Technology Program of Guangzhou (NO. 202102020738, NO. 202102080468), Sanming Project of Medicine in Shenzhen Nanshan (NO. SZSM202103012), Shenzhen Nanshan District Science and Technology Program (NO. NS2022023).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Mengye Wu, Cuiping Li and Xue Zhou performed the research and interpreted the data. Ming, Pan, Zhiyong Wu and Xiaorong Lai established a mouse model of IBD. Qinghai Lian performed metabolic analysis. Jianqi Feng, Xiaolu Guo and Rui Fang performed RNA-seq analysis, qPCR and western blotting. Yanwen Peng and Mengye Wu wrote the manuscript and all authors commented on previous versions of the manuscript. Yanwen Peng supervised the project. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interests to report.
Ethics approval
All of the experimental animal protocols were approved by the Sun Yat-sen University Institutional Animal Care and Use Committee. (reference number: SYSU-IACUC-2020-000423, approval date: 2020.11.23).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, M., Li, C., Zhou, X. et al. Wogonin preconditioning of MSCs improved their therapeutic efficiency for colitis through promoting glycolysis. Inflammopharmacol (2024). https://doi.org/10.1007/s10787-024-01491-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10787-024-01491-2