Abstract
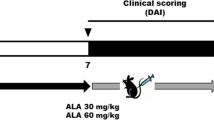
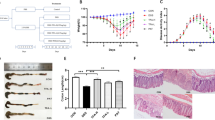
The aim of this investigation was to explore the protective impacts and mechanisms of Anastatica hierochuntica essential oil (EOAH) against dextran sulfate sodium (DSS)-induced experimental colitis in mice. EOAH demonstrated a reduction in DSS-induced body weight decline, disease activity index (DAI), colon length reduction, colonic tissue damage, and myeloperoxidase (MPO) activity. The essential oil significantly mitigated the production of pro-inflammatory agents including TNF-α, IL-1β, and IL-12. Further analysis revealed that EOAH’s anti-inflammatory effects involved the regulation of NF-κB and PPARγ pathways, as well as the inhibition of NLRP3 activation in colitis mice. Notably, EOAH treatment elevated the levels of beneficial commensal bacteria such as Lactobacillus and Bifidobacteria, while reducing Escherichia coli levels in the mice’s feces. In addition, EOAH restored the expression of occludin and ZO-1 proteins in colonic tissues affected by ulcerative colitis (UC). These findings indicate that supplementing with EOAH might offer a novel therapeutic approach for UC prevention.







Similar content being viewed by others
Data availability
Data created/analyzed associated with this research is available from the corresponding author upon reasonable request.
References
Adachi M, Kurotani R, Morimura K et al (2006) Peroxisome proliferator activated receptor γ in colonic epithelial cells protects against experimental inflammatory bowel disease. Gut 55:1104–1113. https://doi.org/10.1136/GUT.2005.081745
Alex P, Zachos NC, Nguyen T et al (2009) Distinct cytokine patterns identified from multiplex profiles of murine DSS and TNBS-induced colitis. Inflamm Bowel Dis 15:341–352. https://doi.org/10.1002/IBD.20753
Ali BH, Baker RK, Mohammd TU, Hassn HA (2013) Anastatica hierochuntica L. used as an alternative of conjugated estrogen (premarin) in rabbit females council for innovative research. J J Adv Chem 9:1783–1786
Alqudah A, AbuDalo R, Qnais E et al (2023) Potential anti-inflammatory activity of the Anastatica hierochuntica essential oil. J Essent Oil Res 35:1–10. https://doi.org/10.1080/10412905.2022.2118878
Bashashati M, Habibi RH, Keshavarzian A et al (2015) Intestinal microbiota: a regulator of intestinal inflammation and cardiac ischemia?. Curr Drug Targets 16(3):199–208
Bauer C, Duewell P, Mayer C et al (2010) Colitis induced in mice with dextran sulfate sodium (DSS) is mediated by the NLRP3 inflammasome. Gut 59:1192–1199. https://doi.org/10.1136/GUT.2009.197822
Celinski K, Dworzanski T, Korolczuk A et al (2011) Effects of peroxisome proliferator-actvated receptors-γamma ligands on dextran sodium sulphate-induced colitis in rats. J Physiol Pharmacol 62:347–356
Coskun M, Vermeire S, Nielsen OH (2017) Novel targeted therapies for inflammatory bowel disease. Trends Pharmacol Sci 38:127–142. https://doi.org/10.1016/J.TIPS.2016.10.014
Cosnes J, Gowerrousseau C, Seksik P, Cortot A (2011) Epidemiology and natural history of inflammatory bowel diseases. Gastroenterology 140:1785-1794.e4. https://doi.org/10.1053/J.GASTRO.2011.01.055
Dahham SS, Tabana YM, Ahamed MBK, Majid AMSA (2016) In vivo anti-inflammatory activity of β-caryophyllene evaluated by molecular imaging. Mol Med Chem. https://doi.org/10.14800/mmc.1001
Desreumaux P, Dubuquoy L, Nutten S et al (2001) Attenuation of colon inflammation through activators of the retinoid X Receptor (Rxr)/peroxisome proliferator-activated receptor γ (Pparγ) heterodimera basis for new therapeutic strategies. J Exp Med 193:827–838. https://doi.org/10.1084/JEM.193.7.827
Dieleman LA, Hoentjen F, Qian BF et al (2004) Reduced ratio of protective versus proinflammatory cytokine responses to commensal bacteria in HLA-B27 transgenic rats. Clin Exp Immunol 136:30–39. https://doi.org/10.1111/J.1365-2249.2004.02410.X
Dinarello CA (2009) Immunological and inflammatory functions of the interleukin-1 family. Annu Rev Immunol 27:519–550. https://doi.org/10.1146/ANNUREV.IMMUNOL.021908.132612
Dubuquoy L, Rousseaux C, Thuru X et al (2006) PPARγ as a new therapeutic target in inflammatory bowel diseases. Gut 55:1341–1349. https://doi.org/10.1136/GUT.2006.093484
Feng Y, Wang Y, Wang P et al (2018) Short-chain fatty acids manifest stimulative and protective effects on intestinal barrier function through the inhibition of NLRP3 inflammasome and autophagy. Cell Physiol Biochem 49:190–205. https://doi.org/10.1159/000492853
Franchi L, Eigenbrod T, Muñoz-Planillo R, Nuñez G (2009) The inflammasome: a caspase-1-activation platform that regulates immune responses and disease pathogenesis. Nat Immunol 103(10):241–247. https://doi.org/10.1038/ni.1703
Fukuda T, Majumder K, Zhang H et al (2016) Adenine inhibits TNF-α signaling in intestinal epithelial cells and reduces mucosal inflammation in a dextran sodium sulfate-induced colitis mouse model. J Agric Food Chem 64:4227–4234. https://doi.org/10.1021/ACS.JAFC.6B00665/SUPPL_FILE/JF6B00665_SI_001.PDF
Garside P (1999) Cytokines in experimental colitis. Clin Exp Immunol 118:337. https://doi.org/10.1046/J.1365-2249.1999.01088.X
Gerova VA, Stoynov SG, Katsarov S et al (2011) Increased intestinal permeability in inflammatory bowel diseases assessed by iohexol test. World J Gastroenterol 17:2211. https://doi.org/10.3748/wjg.v17.i17
Håkansson Å, Tormo-Badia N, Baridi A et al (2015) Immunological alteration and changes of gut microbiota after dextran sulfate sodium (DSS) administration in mice. Clin Exp Med 15:107–120. https://doi.org/10.1007/S10238-013-0270-5/FIGURES/7
Haque MR, Ansari SH (2019) Aromatic aldehyde compound cuminaldehyde protects nonalcoholic fatty liver disease in rats feeding high fat diet. Hum Exp Toxicol 823–832. https://doi.org/10.1177/0960327119842248
Ji KL, Gan XQ, Xu YK et al (2016) Protective effect of the essential oil of Zanthoxylum myriacanthum var. pubescens against dextran sulfate sodium-induced intestinal inflammation in mice. Phytomedicine 23:883–890. https://doi.org/10.1016/J.PHYMED.2016.05.006
Khor B, Gardet A, Xavier RJ (2011) Genetics and pathogenesis of inflammatory bowel disease. Nat 4747351(474):307–317. https://doi.org/10.1038/nature10209
Kim DH, Cheon JH (2017) Pathogenesis of inflammatory bowel disease and recent advances in biologic therapies. Immune Netw 17:25–40. https://doi.org/10.4110/IN.2017.17.1.25
Kim H, Banerjee N, Sirven MA et al (2017) Pomegranate polyphenolics reduce inflammation and ulceration in intestinal colitis—involvement of the miR-145/p70S6K1/HIF1α axis in vivo and in vitro. J Nutr Biochem 43:107–115. https://doi.org/10.1016/J.JNUTBIO.2017.02.005
Kim MG, Kim SM, Min JH et al (2019) Anti-inflammatory effects of linalool on ovalbumin-induced pulmonary inflammation. Int Immunopharmacol 74:105706. https://doi.org/10.1016/J.INTIMP.2019.105706
Kobayashi Y, Kovacs-Nolan J, Matsui T, Mine Y (2015) The Anti-atherosclerotic dipeptide, Trp-His, reduces intestinal inflammation through the blockade of L-type Ca2+ channels. J Agric Food Chem 63:6041–6050. https://doi.org/10.1021/ACS.JAFC.5B01682/SUPPL_FILE/JF5B01682_SI_001.PDF
Korinek M, Handoussa H, Tsai YH et al (2021) Anti-inflammatory and antimicrobial volatile oils: fennel and cumin inhibit neutrophilic inflammation via regulating calcium and MAPKs. Front Pharmacol 12:674095. https://doi.org/10.3389/FPHAR.2021.674095/BIBTEX
Kucharzik T, Walsh SV, Chen J et al (2001) Neutrophil transmigration in inflammatory bowel disease is associated with differential expression of epithelial intercellular junction proteins. Am J Pathol 159:2001–2009. https://doi.org/10.1016/S0002-9440(10)63051-9
Laukoetter MG, Bruewer M, Nusrat A (2006) Regulation of the intestinal epithelial barrier by the apical junctional complex. Curr Opin Gastroenterol 22:85–89. https://doi.org/10.1097/01.MOG.0000203864.48255.4F
Li D, Feng Y, Tian M et al (2021) Gut microbiota-derived inosine from dietary barley leaf supplementation attenuates colitis through PPARγ signaling activation. Microbiome 9:1–22. https://doi.org/10.1186/S40168-021-01028-7/FIGURES/7
Lin T, Chen L, Yang X et al (2022) NLRP3 and gut microbiota homeostasis: progress in research. Cells 11:3758. https://doi.org/10.3390/CELLS11233758
Liu K, Chen Q, Liu Y et al (2012) Isolation and biological activities of decanal, linalool, valencene, and octanal from sweet orange oil. J Food Sci. https://doi.org/10.1111/J.1750-3841.2012.02924.X
Lu A, Magupalli VG, Ruan J et al (2014) Unified polymerization mechanism for the assembly of asc-dependent inflammasomes. Cell 156:1193–1206. https://doi.org/10.1016/j.cell.2014.02.008
MamdouhHashiesh H, Sheikh A, Meeran MFN et al (2023) β-Caryophyllene, a dietary phytocannabinoid, alleviates diabetic cardiomyopathy in mice by inhibiting oxidative stress and inflammation activating cannabinoid type-2 receptors. ACS Pharmacol Transl Sci 6:1129–1142. https://doi.org/10.1021/ACSPTSCI.3C00027/SUPPL_FILE/PT3C00027_SI_001.PDF
Matsuoka K, Kanai T (2015) The gut microbiota and inflammatory bowel disease. Semin Immunopathol 37:47–55. https://doi.org/10.1007/S00281-014-0454-4/TABLES/2
Matsuoka K, Saito E, Fujii T et al (2015) Tacrolimus for the treatment of ulcerative colitis. Intest Res 13:219–226. https://doi.org/10.5217/IR.2015.13.3.219
McGuckin MA, Eri R, Simms LA et al (2009) Intestinal barrier dysfunction in inflammatory bowel diseases. Inflamm Bowel Dis 15:100–113. https://doi.org/10.1002/IBD.20539
Mine Y, Zhang H (2015a) Anti-inflammatory effects of poly-L-lysine in intestinal mucosal system mediated by calcium-sensing receptor activation. J Agric Food Chem 63:10437–10447. https://doi.org/10.1021/ACS.JAFC.5B03812/SUPPL_FILE/JF5B03812_SI_001.PDF
Mine Y, Zhang H (2015b) Calcium-sensing receptor (CaSR)-mediated anti-inflammatory effects of L-amino acids in intestinal epithelial cells. J Agric Food Chem 63:9987–9995. https://doi.org/10.1021/ACS.JAFC.5B03749/ASSET/IMAGES/MEDIUM/JF-2015-03749C_0008.GIF
Nakajima A, Wada K, Miki H et al (2001) Endogenous PPARγ mediates anti-inflammatory activity in murine ischemia-reperfusion injury. Gastroenterology 120:460–469. https://doi.org/10.1053/GAST.2001.21191
Nakashima S, Matsuda H, Oda Y et al (2010) Melanogenesis inhibitors from the desert plant Anastatica hierochuntica in B16 melanoma cells. Bioorg Med Chem 18:2337–2345. https://doi.org/10.1016/J.BMC.2010.01.046
Oshitani N, Watanabe K, Nakamura S et al (2005) Dislocation of tight junction proteins without F-actin disruption in inactive Crohn’s disease. Int J Mol Med 15:407–410. https://doi.org/10.3892/IJMM.15.3.407/HTML
Peana AT, D’Aquila PS, Panin F et al (2002) Anti-inflammatory activity of linalool and linalyl acetate constituents of essential oils. Phytomedicine 9:721–726. https://doi.org/10.1078/094471102321621322
Pulla Reddy ChA, Lokesh BR (1994) Studies on anti-inflammatory activity of spice principles and dietary n-3 polyunsaturated fatty acids on carrageenan-induced inflammation in rats. Ann Nutr Metab 38:349–358. https://doi.org/10.1159/000177833
Qnais E, Modallal N, Bseiso Y et al (2017) Evaluation of the antinociceptive effects of the essential oil from aerial parts of anastatica hierochuntica in experimental models. Pharmacologyonline 3:112–122
Rand TG, Sun M, Gilyan A et al (2010) Dectin-1 and inflammation-associated gene transcription and expression in mouse lungs by a toxic (1,3)-β-D glucan. Arch Toxicol 84:205–220. https://doi.org/10.1007/S00204-009-0481-4/FIGURES/5
Ruyssers NE, De Winter BY, De Man JG et al (2009) Therapeutic potential of helminth soluble proteins in TNBS-induced colitis in mice. Inflamm Bowel Dis 15:491–500. https://doi.org/10.1002/IBD.20787
Scandiffio R, Geddo F, Cottone E et al (2020) Protective effects of (E)-β-Caryophyllene (BCP) in chronic inflammation. Nutr 12:3273. https://doi.org/10.3390/NU12113273
Shaban F, Al-Azzawie HF, Sadiq Mohammed A (2011) EFFECT of alcoholic anastatica hierochuntica extract on some biochemical and histological parameters in alloxan induced diabetic rats. Iraqi J Sci 52:445–455
Shah YM, Morimura K, Gonzalez FJ (2007) Expression of peroxisome proliferator-activated receptor-γ in macrophage suppresses experimentally induced colitis. Am J Physiol Gastrointest Liver Physiol 292:657–666. https://doi.org/10.1152/AJPGI.00381.2006/ASSET/IMAGES/LARGE/ZH30030746680005.JPEG
Su CG, Wen X, Bailey ST et al (1999) A novel therapy for colitis utilizing PPAR-γ ligands to inhibit the epithelial inflammatory response. J Clin Invest 104:383–389. https://doi.org/10.1172/JCI7145
Tomy MJ, Dileep KV, Prasanth S et al (2014) Cuminaldehyde as a lipoxygenase inhibitor. in vitro and in silico validation. Appl Biochem Biotechnol 174:388–397. https://doi.org/10.1007/S12010-014-1066-0/TABLES/2
Yadav PN, Liu Z, Rafi MM (2003) A diarylheptanoid from lesser galangal (Alpinia officinarum) inhibits proinflammatory mediators via inhibition of mitogen-activated protein kinase, p44/42, and transcription factor nuclear factor-κB. J Pharmacol Exp Ther 305:925–931. https://doi.org/10.1124/JPET.103.049171
Yao X, Zhang C, Xing Y et al (2017) (2017) Remodelling of the gut microbiota by hyperactive NLRP3 induces regulatory T cells to maintain homeostasis. Nat Commun 81(8):1–17. https://doi.org/10.1038/s41467-017-01917-2
Zhang J, Fu S, Sun S et al (2014) Inflammasome activation has an important role in the development of spontaneous colitis. Mucosal Immunol 7:1139–1150. https://doi.org/10.1038/MI.2014.1
Zhang Z, Shen P, Liu J et al (2017) In vivo study of the efficacy of the essential oil of Zanthoxylum bungeanum pericarp in dextran sulfate sodium-induced murine experimental colitis. J Agric Food Chem 65:3311–3319. https://doi.org/10.1021/ACS.JAFC.7B01323/SUPPL_FILE/JF7B01323_SI_001.PDF
Zhao J, Zhao R, Cheng L et al (2018) Peroxisome proliferator-activated receptor gamma activation promotes intestinal barrier function by improving mucus and tight junctions in a mouse colitis model. Dig Liver Dis 50:1195–1204. https://doi.org/10.1016/J.DLD.2018.04.016
Acknowledgements
The authors would like to thank the Deanship of Scientific Research at the Hashemite University for sponsoring this research.
Funding
The authors would like to extend their sincere appreciation to the Deanship of Scientific Research at the Hashemite University, Zarqa, Jordan (Grant number: 34/2020).
Author information
Authors and Affiliations
Contributions
Conceptualization, A.A. and E.S.A.; methodology, Y.B.; software, O.G.; validation, M.A., S.A. and A.A.; formal analysis, M.O.; investigation, A.A.; resources, E.Q.; data curation, R.A.D; writing—original draft preparation, A.A.; writing—review and editing, A.A.; visualization, M.W.; supervision, O.G.; project administration, E.Q.; funding acquisition, A.A. All the authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Animal experimental procedures were approved by the animal ethics committee at the Hashemite University (IRB number: 14/4/2021/2022, 14 April 2022), and were in accordance with the guidelines of the US National Institutes of Health on the use and care of laboratory animals, and with the Animal Research: Reporting of in Vivo Experiments (ARRIVE) guidelines Experiment design and induction of colitis in mice.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alqudah, A., Qnais, E., Gammoh, O. et al. Exploring the therapeutic potential of Anastatica hierochuntica essential oil in DSS-induced colitis. Inflammopharmacol (2024). https://doi.org/10.1007/s10787-024-01449-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10787-024-01449-4