Abstract
Recent studies have shown that, coupled with other environmental factors, aluminium exposure may lead to neurodegeneration resulting in cognitive impairment resembling Alzheimer's disease. Menaquinone, a form of vitamin K2, aids in maintaining healthy bones and avoids coronary calcification. It also has anti-inflammatory and antioxidant properties. Here, we study the neuroprotective effects of vitamin K2 (MK-7) using the animal model of Alzheimer's disease (AD). Aluminium chloride (AlCl3; 100 mg/kg for 3 weeks orally) was administered to Swiss albino mice to induce neurodegeneration and Vitamin K2 (100 g/kg for 3 weeks orally) was applied as treatment. This was followed by behavioural studies to determine memory changes. The behavioural observations correlated with proinflammatory, oxidative, and brain histopathological changes in AlCl3-treated animals with or without vitamin K2 treatment. AlCl3 administration led to memory decline which was partially restored in Vitamin K2 treated animals. Myeloperoxidase levels in the brain increased due to AlCl3-mediated inflammation, which Vitamin K2 prevented. The acetylcholine esterase and oxidative stress markers induced by AlCl3 were reversed by Vitamin K2. Also, Vitamin K2 helps to restore hippocampal BDNF levels and reduced the amyloid β accumulation in AlCl3-administered animals. Additionally, Vitamin K2 protected the hippocampal neurons against AlCl3-mediated damage as observed in histopathological studies. We conclude that Vitamin K2 could partially reverse AlCl3-mediated cognitive decline. It increases acetylcholine and BDNF levels while reducing oxidative stress, neuroinflammation, and β-amyloid deposition, thus protecting the hippocampal neurons from AlCl3-mediated damage.
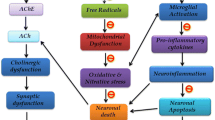
Graphical abstract







Similar content being viewed by others
Data availability
The available data has been shared. Further quaries ma be directed to authors.
References
Abbas F, Eladl MA, El-Sherbiny M, Abozied N, Nabil A, Mahmoud SM, Mokhtar HI, Zaitone SA, Ibrahim D (2022) Celastrol and thymoquinone alleviate aluminum chloride-induced neurotoxicity: Behavioral psychomotor performance, neurotransmitter level, oxidative-inflammatory markers, and BDNF expression in rat brain. Biomed Pharmacother 151:113072. https://doi.org/10.1016/j.biopha.2022.113072
Akhtar A, Dhaliwal J, Saroj P, Uniyal A, Bishnoi M, Sah SP (2020) Chromium picolinate attenuates cognitive deficit in ICV-STZ rat paradigm of sporadic Alzheimer’s-like dementia via targeting neuroinflammatory and IRS-1/PI3K/AKT/GSK-3β pathway. Inflammopharmacol 28:385–400. https://doi.org/10.1007/s10787-019-00681-7
Ameen N, Shafi S (2016) Biochemical and in-vivo antioxidant parameters for evaluation of memory enhancing activity. Int J Pharm Chem Biol Sci 6(3):265–270
Antunes M, Biala, (2012) The novel object recognition memory: neurobiology, test procedure, and its modifications Cogn. Process 13(2):93–110. https://doi.org/10.1007/s10339-011-0430-z
Borai IH, Ezz MK, Rizk MZ, Aly HF, El-Sherbiny M, Matloub AA, Fouad GI (2017) Therapeutic impact of grape leaves polyphenols on certain biochemical and neurological markers in AlCl3-induced Alzheimer’s disease. Biomed Pharmacother 93:837–851. https://doi.org/10.1016/j.biopha.2017.07.038
Chiroma SM, MohdMoklas MA, Mat Taib CN, Baharuldin MTH, Amon Z (2018) D-galactose and aluminium chloride induced rat model with cognitive impairments. Biomed Pharmacother 103:1602–1608
del Blanco B, Barco A (2018) Impact of environmental conditions and chemicals on the neuronal epigenome. Curr Opin Chem Biol 45:157–165. https://doi.org/10.1016/j.cbpa.2018.06.003
Ellman EL, Courtney KD, Andres V Jr, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7(2):88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Ennaceur A (2010) One-trial object recognition in rats and mice: methodological and theoretical issues. Behav Brain Res 215(2):244–254. https://doi.org/10.1016/j.bbr.2009.12.036
Exley C (2017) Aluminum should now be considered a primary etiological factor in Alzheimer’s disease. J Alzheimers Dis Rep 1(1):23–25. https://doi.org/10.3233/ADR-170010
Exley C, Esiri MM (2006) Severe cerebral congophilic angiopathy coincident with increased brain aluminium in a resident of Camelford, Cornwall, UK. J Neurol Neurosurg Psychiatry 77(7):877–879. https://doi.org/10.1136/jnnp.2005.086553
Firdaus Z, Kumar D, Singh SK, Singh TD (2022) Centella asiatica alleviates AlCl3-induced cognitive impairment, oxidative stress, and neurodegeneration by modulating cholinergic activity and oxidative burden in rat brain. Biol Trace Elem Res 200:5115–5126. https://doi.org/10.1007/s12011-021-03083-5
Gareau MG, Wine E, Rodrigues DM, Cho JH, Whary MT, Philpott DJ, MacQueen G, Sherman PM (2011) Bacterial infection causes stress-induced memory dysfunction in mice. Gut 60(3):307–317 (gut.2009.202515)
Gupta A, Jalhan SH, Jindal A, Upmanyu N (2012) Various animal models to check learning and memory—a review. Int J Pharm 4:91–95
Hadipour E, Tayarani-Najaran Z, Fereidoni M (2020) Vitamin K2 protects PC12 cells against Aβ (1–42) and H2O2-induced apoptosis via p38 MAP kinase pathway. Nutr Neurosci 23(5):343–352. https://doi.org/10.1080/1028415X.2018.1504428
Heijtz RD, Wang S, Anuar F, Qian Y, Bjorkholm B, Samuelsson A, Hibberd ML, Forssberg H, Pettersson S (2011) Normal gut microbiota modulates brain development and behaviour. Proc Natl Acad Sci 108(7):3047–3052. https://doi.org/10.1073/pnas.1010529108
Komada M, Takao K, Miyakawa T (2008) Elevated plus maze for mice. JoVE-J vis Exp 22:e1088. https://doi.org/10.3791/1088
McLachlan DRC, Bergeron C, Alexandrov PN, Walsh WJ, Pogue AI, Percy ME, Kruck TPA, Fang Z, Sharfman NM, Jaber V, Zhao Y, Li W, Lukiw WJ (2019) Aluminum in neurological and neurodegenerative disease. Mol Neurobiol 56:1531–1538. https://doi.org/10.1007/s12035-018-1441-x
Miranda M, Morici JF, Zanoni MB, Bekinschtein P (2019) Brain-derived neurotrophic factor: a key molecule for memory in the healthy and the pathological brain. Front Cell Neurosci. https://doi.org/10.3389/fncel.2019.00363
Moron MS, Depierre JW, Mannervik B (1979) Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. BBA-Gen Subjects 582(1):67–78. https://doi.org/10.1016/0304-4165(79)90289-7
Pulli B, Ali M, Forghani R, Schob S, Hsieh KL, Wojtkiewicz G, Linnoila JJ, Chen JW (2013) Measuring myeloperoxidase activity in biological samples. PLoS ONE. https://doi.org/10.1371/journal.pone.0067976
Saba K, Rajnala N, Veeraiah P, Tiwari V, Rana RK, Lakhotia SC, Patel AB (2017) Energetics of excitatory and inhibitory neurotransmission in aluminum chloride model of Alzheimer’s disease: reversal of behavioral and metabolic deficits by Rasa Sindoor. Front Mol Neurosci 10:323. https://doi.org/10.3389/fnmol.2017.00323
Saputra WD, Aoyama N, Komai M, Shirakawa H (2019) Menaquinone-4 suppresses lipopolysaccharide-induced inflammation in MG6 mouse microglia-derived cells by inhibiting the NF-κB signaling pathway. Int J Mol Sci 20(9):2317. https://doi.org/10.3390/ijms20092317
Sarkar SR, Mazumder PM, Chatterjee K, Sarkar A, Adhikary M, Mukhopadhyay K, Banerjee S (2021) Saccharomyces boulardii ameliorates gut dysbiosis associated cognitive decline. Physiol Behav 236:113411. https://doi.org/10.1016/j.physbeh.2021.113411
Shunan D, Yu M, Guan H, Zhou Y (2021) Neuroprotective effect of Betalain against AlCl3-induced Alzheimer’s disease in Sprague Dawley Rats via putative modulation of oxidative stress and nuclear factor kappa B (NF-κB) signaling pathway. Biomed Pharmacothe 137:111369. https://doi.org/10.1016/j.biopha.2021.111369
Tanaka S, Kondo H, Kanda K, Ashino T, Nakamachi T, Sekikawa K, Iwakura Y, Shioda S, Numazawa S, Yoshida T (2011) Involvement of interleukin-1 in lipopolysaccaride-induced microglial activation and learning and memory deficits. J Neurosci Res 89(4):506–514. https://doi.org/10.1002/jnr.22582
Wang T, Hu X, Liang S, Li W, Wu X, Wang L, Jin (2015) Lactobacillus fermentum NS9 restores the antibiotic induced physiological and psychological abnormalities in rats. Benef Microbes 6(5):707–717. https://doi.org/10.3920/BM2014.0177
Wang CS, Kavalali ET, Monteggia LM (2021) BDNF signaling in context: from synaptic regulation to psychiatric disorders. Cell. https://doi.org/10.1016/j.cell.2021.12.003
Wei Y, Liu D, Zheng Y, Li H, Hao C, Ouyang W (2017) Protective effects of kinetin against aluminum chloride and D-galactose induced cognitive impairment and oxidative damage in mouse. Brain Res Bull 134:262–272. https://doi.org/10.1016/j.brainresbull.2017.08.014
Yang WN, Hu XD, Han H, Shi LL, Feng GF, Liu Y, Qian YH (2014) The effects of valsartan on cognitive deficits induced by aluminum trichloride and d-galactose in mice. Neurol Res 36(7):651–658. https://doi.org/10.1179/1743132813Y.0000000295
Zameer S, Najmi AK, Vohora D, Akhtar M (2018) Bisphosphonates: future perspective for neurological disorders. Curr Pharmacol Rep PR 70(5):900–907. https://doi.org/10.1016/j.pharep.2018.03.011
Funding
The work is not funded by any agency.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chatterjee, K., Mazumder, P.M. & Banerjee, S. Vitamin K2 protects against aluminium chloride-mediated neurodegeneration. Inflammopharmacol 31, 2675–2684 (2023). https://doi.org/10.1007/s10787-023-01290-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01290-1