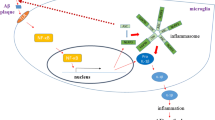
Abstract
Alzheimer’s disease (AD) is a progressive type of neurodegenerative disease characterized by successive loss of the conventional structure and functions of neurons. In addition to dead neurons type detected within AD brain tissues, there are a predominantly varying number of deteriorating neurons (DTNs). As the number of deteriorating neurons increases, they exaggerate the release of inflammatory factors and oxidative stress that trigger the cascade of neuroinflammation. Triggering receptor expressed on myeloid cells 1 (TREM-1) which is a transmembrane immune receptor type regularly expressed by phagocytic cells, may act as a stimulating factor for neuroinflammation. Once TREM-1 is activated, it directly activates spleen tyrosine kinase (SYK) downstream signaling cascades, which can be considered an initiating phase for AD pathology and AD progression. Sequentially, SYK activates the pro-inflammatory microglia M1 phenotype which executes several inflammatory actions, leading to neurotoxicity. These released neurotoxins promote neuronal cell death, synaptic dysfunctions, and memory impairments. Thus, the current review outlines the direct etiological and pathologic features of Alzheimer’s disease linked with deteriorating neurons, TREM-1, and SYK.
Graphical abstract


Similar content being viewed by others
Data availability
No data were used for the research described in the article.
Abbreviations
- AD:
-
Alzheimer’s disease
- DTNs:
-
Deteriorating neurons
- TREM-1:
-
Triggering receptor expressed on myeloid cells 1
- SYK:
-
Spleen tyrosine kinase (SYK)
- LOAD:
-
Late-onset type of AD
- NF-κB:
-
Nuclear factor kappa B
- CBF:
-
Cerebral blood flow
- MAP kinase:
-
Mitogen-activated protein kinase
- TNF-α:
-
Tumor necrosis factor-alpha
- IL-6:
-
Interleukin 6
- TLR-4:
-
Toll-like receptor
- PTK:
-
Protein–tyrosine kinase
- PI3K:
-
Phosphoinositide 3-kinase
- JAK:
-
Janus kinase
- GSK3β:
-
Glycogen Synthase Kinase 3 Beta
- NLRP3:
-
NOD-, LRR- and pyrin domain-containing protein 3
- DAP12:
-
DNAX activation protein of 12 kDa
- PLCγ:
-
Phosphoinositide phospholipase C-gamma
- PRRs:
-
Pattern recognition receptors
- MyD88:
-
Myeloid differentiation factor 88
- BACE1:
-
Beta-secretase
References
Alsop DC, Detre JA, Grossman M (2000) Assessment of cerebral blood flow in Alzheimer’s disease by spin-labeled magnetic resonance imaging. Ann Neurol 47:93–100
Anwar M, Fathi M (2022) The cross-talk between microglia-derived exosomes and brain printing biometrics as a diagnostic approach for Parkinson’s disease prior to motor symptoms implications [abstract]. Mov Disord 37(S2):S647–S648
Anwar MM (2021a) Brain-printing biometrics underlying mechanism as an early diagnostic technique for Alzheimer’s disease neurodegenerative type. Curr Res Physiol 4:216–222
Anwar MM (2021b) Oxidative stress-A direct bridge to central nervous system homeostatic dysfunction and Alzheimer’s disease. Cell Biochem Funct. https://doi.org/10.1002/cbf.3673
Anwar MM, Fathi MH (2023) Early approaches of YKL-40 as a biomarker and therapeutic target for Parkinson’s disease. Neurodegener Dis Manag. https://doi.org/10.2217/nmt-2022-0010
Anwar MM, Mabrouk AA (2023) Hepatic and cardiac implications of increased toxic amyloid-beta serum level in lipopolysaccharide-induced neuroinflammation in rats: new insights into alleviating therapeutic interventions. Inflammopharmacology. https://doi.org/10.1007/s10787-023-01202-3
Anwar MM, Ali OS, Rashed LA, Badawi AM, Eltablawy NA (2018) Regulation of miRNA-124, nuclear factor-kappa B and β-catenin expression in response to novel therapeutic protocol in LPS induced Alzheimer’s disease in rats. Res Neurosci 7:17
Anwar MM, Ali OS, Rashed LA, Badawi AM, Eltablawy NA (2019) The neuro engraftment and neuroregenerative effects of hydrogen sulphide donor, intracerebral MSCs, Ginko Biloba and Kefir in attenuating neuropathological hallmarks of lipopolysaccharide induced Alzheimer’s disease rat models. Int J Aging Res 2:38
Anwar MM, Özkan E, Gürsoy-Özdemir Y (2021) The role of extracellular matrix alterations in mediating astrocyte damage and pericyte dysfunction in Alzheimer’s disease: a comprehensive review. Eur J Neurosci. https://doi.org/10.1111/ejn.15372
Anwar MM, Özkan E, Shomalizadeh N, Sapanci S, Özler C, Kesibi J, Gürsoy-Özdemir Y (2023) Assessing the role of primary healthy microglia and gap junction blocker in hindering Alzheimer’s disease neuroinflammatory type: Early approaches for therapeutic intervention. Front Neurosci. https://doi.org/10.3389/fnins.2022.1041461
Aoki N, Kimura S, Xing Z (2003) Role of DAP12 in innate and adaptive immune responses. Curr Pharm Des 9:7–10
Association AS (2019) 2019 Alzheimer’s disease facts and figures. Alzheimers Dement 15:321–387
Birkle TJY, Brown GC (2023) Syk inhibitors protect against microglia-mediated neuronal loss in culture. Front Aging Neurosci 15:1120952
Chaplin DD (2010) Overview of the immune response. J Allergy Clin Immunol 125:S3-23
Colonna M (2003) TREMs in the immune system and beyond. Nat Rev Immunol 3:445–453
de Oliveira Matos A, dos Santos Dantas PH, Silva-Sales FM, Sales-Campos H (2020) The role of the triggering receptor expressed on myeloid cells-1 (TREM-1) in non-bacterial infections. Crit Rev Microbiol 46:237–252
de Oliveira Matos A, dos Santos Dantas PH, Colmenares MTC, Sartori GR, Silva-Sales M, da Silva JHM, Neves BJ, Andrade CH, Sales-Campos H (2023) The CDR3 region as the major driver of TREM-1 interaction with its ligands, an in silico characterization. Comput Struct Biotechnol J 21:2579–2590
Derkinderen P, Scales TM, Hanger DP, Leung KY, Byers HL, Ward MA, Lenz C, Price C, Bird IN, Perera T, Kellie S, Williamson R, Noble W, van Etten RA, Leroy K, Brion JP, Reynolds CH, Anderton BH (2005) Tyrosine 394 is phosphorylated in Alzheimer’s paired helical filament tau and in fetal tau with c-Abl as the candidate tyrosine kinase. J Neurosci 25:6584–6593
Deture MA, Dickson DW (2019) The neuropathological diagnosis of Alzheimer’s disease. Mol Neurodegener 14:32
Faruki S, Geahlen RL, Asai DJ (2000) Syk-dependent phosphorylation of microtubules in activated B-lymphocytes. J Cell Sci 113(Pt 14):2557–2565
Feng C-W, Chen N-F, Sung C-S, Kuo H-M, Yang S-N, Chen C-L, Hung H-C, Chen B-H, Wen Z-H, Chen W-F (2019) Therapeutic effect of modulating TREM-1 via anti-inflammation and autophagy in Parkinson’s disease. Front Neurosci 13:769
Fortin CF, Lesur O, Fulop T (2007) Effects of TREM-1 activation in human neutrophils: activation of signaling pathways, recruitment into lipid rafts and association with TLR4. Int Immunol 19:41–50
Geahlen RL (2014) Getting Syk: spleen tyrosine kinase as a therapeutic target. Trends Pharmacol Sci 35:414–422
Goldsmith HS (2014) Benefit of omental blood flow in Alzheimer’s disease: effect on deteriorating neurons. J Alzheimers Dis 42(Suppl 3):S277–S280
Goldsmith HS (2022) Alzheimer’s disease: a decreased cerebral blood flow to critical intraneuronal elements is the cause. J Alzheimers Dis 85:1419–1422
He X, Huang Y, Liu Y, Zhang X, Yue P, Ma X, Miao Z, Long X, Yang Y, Wan X, Lei J, Shu K, Lei T, Gan C, Zhang H (2022) BAY61-3606 attenuates neuroinflammation and neurofunctional damage by inhibiting microglial Mincle/Syk signaling response after traumatic brain injury. Int J Mol Med 49:1–3
Kapasouri EM, Ioannidis DC, Cameron D, Vassiliou VS, Hornberger M (2022) The utility of arterial spin labeling MRI in medial temporal lobe as a vascular biomarker in Alzheimer’s disease spectrum: a systematic review and meta-analysis. Diagnostics 12:2967
Kelker MS, Foss TR, Peti W, Teyton L, Kelly JW, Wüthrich K, Wilson IA (2004) Crystal structure of human triggering receptor expressed on myeloid cells 1 (TREM-1) at 1.47 A. J Mol Biol 342:1237–1248
Kim MW, Choe K (2022) Pharmacological inhibition of spleen tyrosine kinase suppressed neuroinflammation and cognitive dysfunction in LPS-induced neurodegeneration model. Cells 11:1777
Li Z, Wu F, Xu D, Zhi Z, Xu G (2019) Inhibition of TREM1 reduces inflammation and oxidative stress after spinal cord injury (SCI) associated with HO-1 expressions. Biomed Pharmacother 109:2014–2021
Marchetti G, Dessì A (2020) Syk inhibitors: new computational insights into their intraerythrocytic action in plasmodium falciparum malaria. Int J Mol Sci 21:7009
Mcadoo SP, Tam FW (2011) Fostamatinib disodium. Drugs Fut 36:273
Paris D, Ait-Ghezala G, Bachmeier C, Laco G, Beaulieu-Abdelahad D, Lin Y, Jin C, Crawford F, Mullan M (2014) The spleen tyrosine kinase (Syk) regulates Alzheimer amyloid-β production and Tau hyperphosphorylation. J Biol Chem 289:33927–33944
Radsak MP, Salih HR, Rammensee HG, Schild H (2004) Triggering receptor expressed on myeloid cells-1 in neutrophil inflammatory responses: differential regulation of activation and survival. J Immunol 172:4956–4963
Roe K, Gibot S, Verma S (2014) Triggering receptor expressed on myeloid cells-1 (TREM-1): a new player in antiviral immunity? Front Microbiol 5:627
Ruzza P, Biondi B, Calderan A (2009) Therapeutic prospect of Syk inhibitors. Expert Opin Ther Pat 19:1361–1376
Sagulenko V, Thygesen SJ, Sester DP, Idris A, Cridland JA, Vajjhala PR, Roberts TL, Schroder K, Vince JE, Hill JM, Silke J, Stacey KJ (2013) AIM2 and NLRP3 inflammasomes activate both apoptotic and pyroptotic death pathways via ASC. Cell Death Differ 20:1149–1160
Sehar U, Rawat P, Reddy AP, Kopel J, Reddy PH (2022) Amyloid beta in aging and Alzheimer’s disease. Int J Mol Sci 23:12924
Shi X, Wei T, Hu Y, Wang M, Tang Y (2022) The associations between plasma soluble Trem1 and neurological diseases: a Mendelian randomization study. J Neuroinflammation 19:218
Tessarz AS, Weiler S, Zanzinger K, Angelisová P, Horejsí V, Cerwenka A (2007) Non-T cell activation linker (NTAL) negatively regulates TREM-1/DAP12-induced inflammatory cytokine production in myeloid cells. J Immunol 178:1991–1999
Tsuruga K, Sugawara N, Yasui-Furukori N, Takahashi I, Tsuchimine S, Kaneda A, Nakaji S, Nakamura K (2014) A positive correlation between serum amyloid β levels and depressive symptoms among community-dwelling elderly individuals in Japan. Neuropsychiatr Dis Treat 10:1621–1627
Venegas C, Kumar S, Franklin BS, Dierkes T, Brinkschulte R, Tejera D, Vieira-Saecker A, Schwartz S, Santarelli F, Kummer MP, Griep A, Gelpi E, Beilharz M, Riedel D, Golenbock DT, Geyer M, Walter J, Latz E, Heneka MT (2017) Microglia-derived ASC specks cross-seed amyloid-β in Alzheimer’s disease. Nature 552:355–361
Wu X, Zeng H, Xu C, Chen H, Fan L, Zhou H, Yu Q, Fu X, Peng Y, Yan F, Yu X, Chen G (2021) TREM1 regulates neuroinflammatory injury by modulate proinflammatory subtype transition of microglia and formation of neutrophil extracellular traps via interaction with SYK in experimental subarachnoid hemorrhage. Front Immunol. https://doi.org/10.3389/fimmu.2021.766178
Xu P, Zhang X, Liu Q, Xie Y, Shi X, Chen J, Li Y, Guo H, Sun R, Hong Y, Liu X, Xu G (2019) Microglial TREM-1 receptor mediates neuroinflammatory injury via interaction with SYK in experimental ischemic stroke. Cell Death Dis 10:555
Yamaguchi T, Hamano T, Sada K, Kanaan NM, Sasaki H, Yen S-H, Asano R, Enomoto S, Shirafuji N, Ueno A, Ikawa M, Yamamura O, Nakamoto Y (2020) Syk inhibitor reduces oligomeric tau associated with GSK3β inactivation and autophagy activation. Alzheimers Dement 16:e042633
Zhang C, Kan X, Zhang B, Ni H, Shao J (2022) The role of triggering receptor expressed on myeloid cells-1 (TREM-1) in central nervous system diseases. Mol Brain 15:84
Funding
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
MMA organized the main ideas of the current review, finalized, and submitted the final version for review.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Not applicable.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Consent for publication was obtained for every individual person’s data included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Anwar, M.M. The orchestrating role of deteriorating neurons and TREM-1 in crosstalk with SYK in Alzheimer’s disease progression and neuroinflammation. Inflammopharmacol 31, 2303–2310 (2023). https://doi.org/10.1007/s10787-023-01270-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01270-5