Abstract
Objective and design
Inflammatory bowel disease (IBD) is an idiopathic inflammatory condition of the digestive system marked by oxidative stress, leukocyte infiltration, and elevation of inflammatory mediators. In this study, we demonstrate the protective effect of ethyl gallate (EG), a phytochemical, and propyl gallate (PG), an anti-oxidant, given through normal drinking water (DW) and copper water (CW) in various combinations, which had a positive effect on the amelioration of DSS-induced ulcerative colitis in C57BL/6 J mice.
Materials and methods
We successfully determined the levels of proinflammatory cytokines and anti-oxidant enzymes by ELISA, tracked oxidative/nitrosative stress (RO/NS) by in vivo imaging (IVIS) using L-012 chemiluminescent probe, disease activity index (DAI), and histopathological and morphometric analysis of colon in DSS-induced colitis in a model.
Results
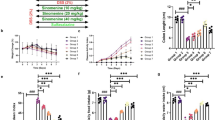
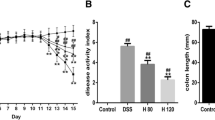
The results revealed that oral administration of ethyl gallate and propyl gallate at a dose of 50 mg/kg considerably reduced the severity of colitis and improved both macroscopic and microscopic clinical symptoms. The level of proinflammatory cytokines (TNF-α, IL-6, IL-1β, and IFN-γ) in colonic tissue was considerably reduced in the DSS + EG-treated and DSS + PG-treated groups, compared to the DSS alone-treated group. IVIS imaging of animals from the DSS + EG and DSS + PG-treated groups showed a highly significant decrease in RO/NS species relative to the DSS control group, with the exception of the DSS + PG/CW and DSS + EG + PG/CW-treated groups. We also observed lower levels of myeloperoxidase (MPO), nitric oxide (NO), and lipid peroxidation (LPO), and restored levels of GST and superoxide dismutase (SOD) in DSS + EG-DW/CW, DSS + PG/DW, and DSS + EG + PG/DW groups compared to DSS alone-treated group. In addition, we showed that the EG, PG, and EG + PG treatment significantly reduced the DAI score, and counteracted the body weight loss and colon shortening in mice compared to DSS alone-treated group. In this 21-day study, mice were treated daily with test substances and were challenged to DSS from day-8 to 14.
Conclusion
Our study highlights the protective effect of ethyl gallate and propyl gallate in various combinations which, in pre-clinical animals, serve as an anti-inflammatory drug against the severe form of colitis, indicating its potential for the treatment of IBD in humans. In addition, propyl gallate was investigated for the first time in this study for its anti-colitogenic effect with normal drinking water and reduced effect with copper water.
Graphical Abstract










Similar content being viewed by others
Data availability
All the data are available in the manuscript. On reasonable request, the corresponding author will provide the datasets used or analyzed during the current work.
Abbreviations
- AGEs:
-
Advanced glycation end products
- ANOVA:
-
Analysis of variance
- BCA:
-
Bicinchoninic acid
- BSA:
-
Bovine serum albumin
- BW:
-
Body weight
- CD:
-
Crohn’s disease
- CDNB:
-
1-Chloro-2: 4-dinitrobenzene
- COX-2:
-
Cycloxygenase-2
- CPCSEA:
-
Committee for the purpose of control and supervision of experiments on animals
- Cu:
-
Copper
- CW:
-
Copper water
- DAI:
-
Disease activity index
- DSS:
-
Dextran sulfate sodium
- DTNB:
-
Dithiobis-nitrobenzoic acid
- DW:
-
Drinking water
- EG:
-
Ethyl gallate
- ELISA:
-
Enzyme-linked immunosorbent assay
- GIT:
-
Gastrointestinal disorder
- GSH:
-
Reduced glutathione
- GSSG:
-
Oxidized glutathione
- GST:
-
Glutathione-S-transferase
- H&E:
-
Hematoxylin and eosin staining
- HO-1:
-
Heme oxygenase 1
- IAEC:
-
Institutional Animal Ethics Committee
- IBD:
-
Inflammatory bowel disease
- IFN-γ:
-
Interferon-gamma
- IL-1β:
-
Interleukin-1 beta
- IL-6:
-
Interleukin 6
- iNOS:
-
Inducible nitric oxide synthase
- IκB:
-
Inhibitor of kappa B
- LPO:
-
Lipid peroxidase
- LD50 :
-
Lethal dose 50
- MDA:
-
Malondialdehyde
- MPO:
-
Myeloperoxidase
- NFκB:
-
Nuclear factor-κB
- NO:
-
Nitric oxide
- Nrf2:
-
Nuclear factor erythroid 2-related factor 2
- PBS:
-
Phosphate-buffered saline
- PC:
-
Positive control (DSS alone-treated group)
- PG:
-
Propyl gallate
- RIPA:
-
Radioimmunoprecipitation assay
- RNS:
-
Reactive nitrogen species
- ROI:
-
Region of interest
- RO/NS:
-
Reactive oxygen/nitrogen species
- ROS:
-
Reactive oxygen species
- SC:
-
Standard control (sulfasalazine drug)
- SEM:
-
Standard error of the mean
- SOD:
-
Superoxide dismutase
- TBARS:
-
Thiobarbituric acid-reactive substances
- TLR-4:
-
Toll-like receptor 4
- TNF-α:
-
Tumor necrosis factor-α
- UC:
-
Ulcerative colitis
- WHO:
-
World Health Organization
References
Abdelmegid MA, Abdo FK, Ahmed FE, Kattaia AAJS (2019) Therapeutic effect of gold nanoparticles on DSS-induced ulcerative colitis in mice with reference to interleukin-17 expression. Sci Rep 9:1–16
Abhirami NR, Laksmi VV, Deepitha AM (2022) A review on prevalence of inflammatory bowel disease in India. J Drug Deliv Therapeutics 12(6):219–223
Ahn D, Kim J, Nam G, Zhao X, Kwon J, Hwang JY, Kim JK, Yoon SY, Chung SJ (2022) Ethyl gallate dual-targeting PTPN6 and PPARγ shows anti-diabetic and anti-obese effects. Int J Mol Sci 23(9):5020
Alavala S, Sangaraju R, Nalban N, SAHU BD, JERALD MK, KILARI EK, SISTLA R. (2019) Stevioside, a diterpenoid glycoside, shows anti-inflammatory property against dextran sulphate sodium-induced ulcerative colitis in mice. European J Pharmacol 855:192–201
Asghar MN, Emani R, Alam C, Helenius TO, Grönroos TJ, Sareila O, Din MU, Holmdahl R, Hänninen A, Toivola DM (2014) In vivo imaging of reactive oxygen and nitrogen species in murine colitis. Inflamm Bowel Dis 20(8):1435–1447
Axelrad JE, Lichtiger S, Yajnik V (2016) Inflammatory bowel disease and cancer: the role of inflammation, immunosuppression, and cancer treatment. World J Gastroenterol 22:4794
Axelsson LG, Landström E, Bylund-Fellenius AC (1998) Experimental colitis induced by dextran sulphate sodium in mice: beneficial effects of sulphasalazine and olsalazine. Aliment Pharmacol Therapeutics 12(9):925–934
Benoit C, Jesse D, Madhu M, Matam VJCPI (2014) Dextran sulfate sodium (DSS)-induced colitis in mice. Current Protocols Immunol 25:1–15
Chen C-H, Liu T-Z, Chen C-H, Wong CH, Chen C-H, Fung-Jou Lu, Chen SC (2007) The efficacy of protective effects of tannic acid, gallic acid, ellagic acid, and propyl gallate against hydrogen peroxide-induced oxidative stress and DNA damages in IMR-90 cells. Mol Nutr Food Res 51:962–968
COSKUN M (2014) Intestinal epithelium in inflammatory bowel disease. Front Med 1:24
Cui H, Yuan J, Du X, Wang M, Yue L, Liu J (2015) Ethyl gallate suppresses proliferation and invasion in human breast cancer cells via Akt-NF-κB signaling. Oncol Reports 33(3):1284–1290
de Mattos B, Garcia M, Nogueira JB, Paiatto LN, Albuquerque CG, Souza CL, Fernandes L, Tamashiro W, Simioni PUJMI (2015) Inflammatory bowel disease: an overview of immune mechanisms and biological treatments. Mediators Inflamm 493012:2015
Gancarcikova S, Lauko S, Hrckova G, Andrejcakova Z, Hajduckova V, Madar M, Fecskeova LK, Mudronova D, Mravcova K, Strkolcova G, Nemcova R, Kacirova J, Staskova A, Vilcek S, Bomba A (2020) Innovative animal model of DSS-induced ulcerative colitis in pseudo germ-free mice. Cell 9:2571
Gaudio E, Taddei G, Vetuschi A, Sfera R, Visconti S, Ricciardi G, Caprilli R (1999) Dextran sulfate sodium (DSS) colitis in rats (clinical, structural, and ultrastructural aspects). Gastroenterology 44:1458–1475
Hsu HC, Lin WC, Chang PJ, Hong CZ, Chen CHJE, Medicine T (2013) Propyl gallate inhibits TPA-induced inflammation via the nuclear factor-κB pathway in human THP-1 monocytes. Exp Therapeutic Med 5:964–968
Jacobi H, Eicke B, Witte I (1998) DNA strand break induction and enhanced cytotoxicity of propyl gallate in the presence of copper (II). Free Radical Biol Med 24(6):972–978
Jacobi H, Hinrichsen ML, Weß D, Witte I (1999) Induction of lipid peroxidation in human fibroblasts by the antioxidant propyl gallate in combination with copper (II). Toxicology Lett 110(3):183–190
Jung HJ, Kim SJ, Jeon WK, Kim BC, Ahn K, Kim K, Kim YM, Park EH, Lim CJ (2011) Anti-inflammatory activity of n-propyl gallate through down-regulation of NF-κB and JNK pathways. Inflammation 34:352–361
Kalaivani T, Rajasekaran C, Mathew L (2011) Free radical scavenging, cytotoxic, and hemolytic activities of an active antioxidant compound ethyl gallate from leaves of Acacia nilotica (L) Wild Ex Delile subsp indica (Benth) Brenan. J Food Sci 76:T144–T149
Karin M (2009) NF-κB as a critical link between inflammation and cancer. Cold Spring Harbor Perspect Biol 1(5):a000141
Kaser A, Zeissig S, Blumberg RSJDD (2010) Genes and environment: how will our concepts on the pathophysiology of IBD develop in the future? Dig Dis 28:395–405
Krishna CM, Kolla JN, Asha S, Reddy TSK (2020) In vitro anti-HIV-1 activity of ethyl gallate. Virus Dis 31:22–27
Macdonald T, Monteleone G, Pender SL (2000) Recent developments in the immunology of inflammatory bowel disease. Scandinavian J Immunol 51:2–9
Mallo N, Lamas J, Leiro JM (2014) Alternative oxidase inhibitors as antiparasitic agents against scuticociliatosis. Parasitology 141(10):1311–1321
Maloy KJ, Powrie F (2011) Intestinal homeostasis and its breakdown in inflammatory bowel disease. Nature 474(7351):298–306
Matricon J (2010) Immunopathogenèse des maladies inflammatoires chroniques de l’intestin. Méd Sci 26(4):405–410
Mehla K, Balwani S, Agrawal A, Ghosh B (2013) Ethyl gallate attenuates acute lung injury through Nrf2 signaling. Biochimie 95(12):2404–2414
Mink SN, Jacobs H, Gotes J, Kasian K, Cheng Z-Q (2011) Ethyl gallate, a scavenger of hydrogen peroxide that inhibits lysozyme-induced hydrogen peroxide signaling in vitro, reverses hypotension in canine septic shock. J Appl Physiol 110:359–374
Mohan S, Thiagarajan K, Chandrasekaran R (2017) Evaluation of ethyl gallate for its antioxidant and anticancer properties against chemical-induced tongue carcinogenesis in mice. Biochem J 474(17):3011–3025
National center for biotechnology information (2023a) PUBCHEM Compound Summary For CID 4947, P. G. R. J., 2023
National center for biotechnology information (2023b) Pubchem compound summary for CID 13250, E. G. R. J., 2023
Ng SC, Shi HY, Hamidi N, Underwood FE, Tang W, Benchimol EI, Panaccione R, Ghosh S, Wu JC, Chan FK, Sung JJ (2017) Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet 390(10114):2769–2778
Sahu BD, Tatireddy S, Koneru M, Borkar RM, Kumar JM, Kuncha M, Srinivas R, Sistla R (2014) Naringin ameliorates gentamicin-induced nephrotoxicity and associated mitochondrial dysfunction, apoptosis and inflammation in rats: possible mechanism of nephroprotection. Toxicol Appl Pharmacol 277(1):8–20
Sanchez-Muñoz F, Dominguez-Lopez A, Yamamoto-Furusho JK (2008) Role of cytokines in inflammatory bowel disease. World J Gastroenterol 14(27):4280
Sangaraju R, Nalban N, Alavala S, Rajendran V, Jerald MK, Sistla R (2019) Protective effect of galangin against dextran sulfate sodium (DSS)-induced ulcerative colitis in Balb/c mice. Inflamm Res 68:691–704
Sangaraju R, Alavala S, Nalban N, Jerald MK, Sistla RJII (2021) Galangin ameliorates Imiquimod-Induced psoriasis-like skin inflammation in BALB/c mice via down regulating NF-κB and activation of Nrf2 signaling pathways. Int Immunopharmacol 96:107754
Shen G, Kong AN (2009) Nrf2 plays an important role in coordinated regulation of Phase II drug metabolism enzymes and Phase III drug transporters. Biopharmaceut Drug Dispos 30(7):345–355
Singer II, Kawka DW, Schloemann S, Tessner T, Riehl T, Stenson WF (1998) Cyclooxygenase 2 is induced in colonic epithelial cells in inflammatory bowel disease. Gastroenterology 115(2):297–306
Singh R, Chandrashekharappa S, Bodduluri SR, Baby BV, Hegde B, Kotla NG, Hiwale AA, Saiyed T, Patel P, Vijay-Kumar M, Langille MG (2019) Enhancement of the gut barrier integrity by a microbial metabolite through the Nrf2 pathway. Nat Commun 10(1):89
Strober W, Fuss IJ (2011) Proinflammatory cytokines in the pathogenesis of inflammatory bowel diseases. Gastroenterology 140(6):1756–1767
Sudha VP, Singh KO, Prasad SR, Venkatasubramanian P (2009) Killing of enteric bacteria in drinking water by a copper device for use in the home: laboratory evidence. Trans Royal Soc Tropical Med Hygiene 103(8):819–822
Sudha VP, Sheeba Ganesan GP, Pazhani TR, Nair GB, Venkatasubramanian P (2009) Killing of enteric bacteria in drinking water by a copper device for use in the home: laboratory evidence. Trans Royal Soc Tropical Med Hygiene 103(8):819–822
Tatiya-aphiradee N, Chatuphonprasert W, Jarukamjorn K (2019) Immune response and inflammatory pathway of ulcerative colitis. J Basic Clin Physiol Pharmacol 30:1–10
Tun X, Yasukawa K, Yamada K-I (2014) Involvement of nitric oxide with activation of toll-like receptor 4 signaling in mice with dextran sodium sulfate-induced colitis. Free Radical Biol Med 74:108–117
Viennois E, Chen F, Merlin D (2013) NF-κB pathway in colitis-associated cancers. Trans Gastrointest Cancer 2(1):21
Wang Z, Xue R, Lv M, Qi Y, Wei Yu, Xie Z, Chen W, Wang X, Tian X, Han Bo (2022) Stepwise tracking strategy to screen ingredient from Galla Chinensis based on the “mass spectrometry guided preparative chromatography coupled with systems pharmacology.” J Ethnopharmacol 284:114533
Wei PL, Huang CY, Chang YJ (2019) Propyl gallate inhibits hepatocellular carcinoma cell growth through the induction of ROS and the activation of autophagy. PLoS ONE 14(1):e0210513
Wu TW, Fung KP, Zeng LH, Wu J, Nakamura H (1994) Propyl gallate as a hepatoprotector in vitro and in vivo. Biochem Pharmacol 48(2):419–422
Xu Y, Pang GR, Lü XF, Chen ZJ, Sun ST, Song JZ (2006) The effect of propyl gallate on the activity of various antifungal drugs against filamentous fungi in vitro. Chin J Ophthalmol 42(4):309–312
Acknowledgements
The authors would like to thank The Indian Council of Medical Research for funding this study [(ICMR) No. of File: 61/03/2021-IMM/BMS] and also the Director, National Institute of Nutrition for the support and motivation to work on the project. Priyanka Raju Chougule thanks Department of Science & Technology, New Delhi, India for providing the DST-INSPIRE Senior Research Fellowship (No: DST/INSPIRE Fellowship/[IF190480]), and Sangaraju Rajendra thanks ICMR, New Delhi, India for providing the ICMR-Research Associate (No. 3/1/2/300/2021-Nut). All authors thank everyone who contributed technical assistance to our research.
Author information
Authors and Affiliations
Contributions
The experiment's conception and design, data analysis, and writing the original manuscript were all done by SNS, PRC, and RS. The experiments such as IVIS, anti-oxidant assays, and ELISA were carried out by SNS, PRC, RS, PBP, SSYHQ, VVP, and MB, while the paper was reviewed by SNS, PRC, RS, PBP, SSYHQ, VVP, SG, SKM, and MB. Performers of the experiments and data analysts were SNS, PRC, RS, PBP, SSYHQ, VVP, and MB. Statistical analysis was carried out by SNS, PRC, and RS. All authors reviewed the paper.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no competing financial interest.
Ethics approval and consent to participate
The protocols for the use of animals were approved by the Institutional Animal Ethics Committee (IAEC) of the ICMR-NIN, and the IAEC approval number is ICMR-NIN/IAEC/02/018/2020.
Consent for publication
All listed authors have approved the manuscript before submission, including the names and order of authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chougule, P.R., Sangaraju, R., Patil, P.B. et al. Effect of ethyl gallate and propyl gallate on dextran sulfate sodium (DSS)-induced ulcerative colitis in C57BL/6 J mice: preventive and protective. Inflammopharmacol 31, 2103–2120 (2023). https://doi.org/10.1007/s10787-023-01254-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01254-5