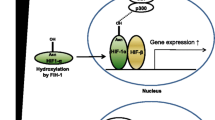
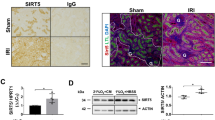
Abstract
The kidneys are the most vulnerable organs to severe ischemic insult that results in cellular hypoxia under pathophysiological conditions. Large amounts of oxygen are consumed by the kidneys, mainly to produce energy for tubular reabsorption. Beyond high oxygen demand and the low oxygen supply, different other factors make kidneys vulnerable to ischemia which is deemed to be a major cause of acute kidney injury (AKI). On the other hand, kidneys are capable of sensing and responding to oxygen alternations to evade harms resulting from inadequate oxygen. The hypoxia-inducible factor (HIF) is the main conserved oxygen-sensing mechanism that maintains homeostasis under hypoxia through direct/indirect regulation of several genes that contribute to metabolic adaptation, angiogenesis, energy conservation, erythropoiesis, and so on. In response to oxygen availability, prolyl-hydroxylases (PHDs) control the HIF stability. This review focuses on the oxygen-sensing mechanisms in kidneys, particularly in proximal tubular cells (PTCs) and discusses the molecules involved in ischemic response and metabolic reprogramming. Moreover, the possible roles of non-coding RNAs (microRNAs and long non-coding RNAs) in the development of ischemic AKI are put forward.



Similar content being viewed by others
Data availability
The review article did not utilize a dataset. All statements are supported by related references.
References
Biju MP, Akai Y, Shrimanker N, Haase VH (2005) Protection of HIF-1-deficient primary renal tubular epithelial cells from hypoxia-induced cell death is glucose dependent. Am J Physiol Renal Physiol 289:F1217–F1226. https://doi.org/10.1152/ajprenal.00233.2005
Bonventre JV, Yang L (2011) Cellular pathophysiology of ischemic acute kidney injury. J Clin Investig 121:4210–4221
Brandenburger T, Lorenzen JM (2020) Diagnostic and therapeutic potential of microRNAs in acute kidney injury. Front Pharmacol 11:657
Cao H, Luo J, Zhang Y, Mao X, Wen P, Ding H et al (2020) Tuberous sclerosis 1 (Tsc1) mediated mTORC1 activation promotes glycolysis in tubular epithelial cells in kidney fibrosis. Kidney Int 98:686–698. https://doi.org/10.1016/j.kint.2020.03.035
Chen F, Hu Y, Xie Y, Zhao Z, Ma L, Li Z et al (2020a) Total glucosides of paeony alleviate cell apoptosis and inflammation by targeting the long noncoding RNA XIST/MicroRNA-124-3p/ITGB1 axis in renal ischemia/reperfusion injury. Mediat Inflamm 2020:8869511. https://doi.org/10.1155/2020/8869511
Chen H, Cheng Q, Wang J, Zhao X, Zhu S (2021) Long-term efficacy and safety of hypoxia-inducible factor prolyl hydroxylase inhibitors in anaemia of chronic kidney disease: a meta-analysis including 13,146 patients. J Clin Pharm 46:999–1009
Chen J, Zheng Y, Li L (2020b) LncRNA RPSAP52 regulates miR-423–5p/GSTM1 axis to suppress hypoxia-induced renal proximal tubular epithelial cell apoptosis. Arch Physiol Biochem. https://doi.org/10.1080/13813455.2020.1750657
Combe C, Burton CJ, Dufourcq P, Weston S, Horsburgh T, Walls J et al (1997) Hypoxia induces intercellular adhesion molecule-1 on cultured human tubular cells. Kidney Int 51:1703–1709. https://doi.org/10.1038/ki.1997.235
De Laplanche E, Gouget K, Cléris G, Dragounoff F, Demont J, Morales A et al (2006) Physiological oxygenation status is required for fully differentiated phenotype in kidney cortex proximal tubules. Am J Physiol Renal Physiol 291:F750–F760. https://doi.org/10.1152/ajprenal.00022.2006
Ding H, Jiang L, Xu J, Bai F, Zhou Y, Yuan Q et al (2017) Inhibiting aerobic glycolysis suppresses renal interstitial fibroblast activation and renal fibrosis. Am J Physiol Renal Physiol 313:F561-f575. https://doi.org/10.1152/ajprenal.00036.2017
El Awad B, Kreft B, Wolber E-M, Hellwig-Bürgel T, Metzen E, Fandrey J et al (2000) Hypoxia and interleukin-1β stimulate vascular endothelial growth factor production in human proximal tubular cells. Kidney Int 58:43–50. https://doi.org/10.1046/j.1523-1755.2000.00139.x
Evans RG, Smith DW, Lee CJ, Ngo JP, Gardiner BS (2020) What makes the kidney susceptible to hypoxia? Anat Rec (hoboken) 303:2544–2552. https://doi.org/10.1002/ar.24260
Feng J, Kong R, Xie L, Lu W, Zhang Y, Dong H et al (2019) Clemaichinenoside protects renal tubular epithelial cells from hypoxia/reoxygenation injury in vitro through activating the Nrf2/HO-1 signalling pathway. Clin Exp Pharmacol Physiol 47:495–502. https://doi.org/10.1111/1440-1681.13219
Fernández-Martínez AB, Arenas Jiménez MI, Lucio Cazaña FJ (2012) Retinoic acid increases hypoxia-inducible factor-1α through intracrine prostaglandin E2 signaling in human renal proximal tubular cells HK-2. Biochimica et Biophysica Acta (BBA) Mol Cell Biol Lipids 1821:672–683. https://doi.org/10.1016/j.bbalip.2012.01.010
Fernandez-Martínez AB, Torija AV, Carracedo J, Ramirez R, De Lucio-Cazaña FJ (2014) Microparticles released by vascular endothelial cells increase hypoxia inducible factor expression in human proximal tubular HK-2 cells. Int J Biochem Cell Biol 53:334–342. https://doi.org/10.1016/j.biocel.2014.05.025
Funk JA, Schnellmann RG (2012) Persistent disruption of mitochondrial homeostasis after acute kidney injury. Am J Physiol Renal Physiol 302:F853–F864. https://doi.org/10.1152/ajprenal.00035.2011
Garcia-Pastor C, Benito-Martinez S, Bosch RJ, Fernandez-Martinez AB, Lucio-Cazana FJ (2021) Intracellular prostaglandin E2 contributes to hypoxia-induced proximal tubular cell death. Sci Rep 11:7047. https://doi.org/10.1038/s41598-021-86219-w
Geng X, Song N, Zhao S, Xu J, Liu Y, Fang Y et al (2020) LncRNA GAS5 promotes apoptosis as a competing endogenous RNA for miR-21 via thrombospondin 1 in ischemic AKI. Cell Death Discov 6:19. https://doi.org/10.1038/s41420-020-0253-8
Godwin JG, Ge X, Stephan K, Jurisch A, Tullius SG, Iacomini J (2010) Identification of a microRNA signature of renal ischemia reperfusion injury. Proc Natl Acad Sci 107:14339–14344
Haase VH (2013) Mechanisms of hypoxia responses in renal tissue. J Am Soc Nephrol 24:537–541. https://doi.org/10.1681/asn.2012080855
Haddad G, Kölling M, Wegmann UA, Dettling A, Seeger H, Schmitt R et al (2021) Renal AAV2-mediated overexpression of long non-coding RNA H19 attenuates ischemic acute kidney injury through sponging of microRNA-30a-5p. J Am Soc Nephrol 32:323–341. https://doi.org/10.1681/asn.2020060775
Hansell P, Welch WJ, Blantz RC, Palm F (2013) Determinants of kidney oxygen consumption and their relationship to tissue oxygen tension in diabetes and hypertension. Clin Exp Pharmacol Physiol 40:123–137. https://doi.org/10.1111/1440-1681.12034
Hesp AC, Schaub JA, Prasad PV, Vallon V, Laverman GD, Bjornstad P et al (2020) The role of renal hypoxia in the pathogenesis of diabetic kidney disease: a promising target for newer renoprotective agents including SGLT2 inhibitors? Kidney Int 98:579–589. https://doi.org/10.1016/j.kint.2020.02.041
Holdstock L, Meadowcroft AM, Maier R, Johnson BM, Jones D, Rastogi A et al (2016) Four-week studies of oral hypoxia-inducible factor–prolyl hydroxylase inhibitor GSK1278863 for treatment of anemia. J Am Soc Nephrol 27:1234–1244
Hong YA, Jung SY, Yang KJ, Im DS, Jeong KH, Park CW et al (2020) Cilastatin preconditioning attenuates renal ischemia-reperfusion injury via hypoxia inducible factor-1α activation. Int J Mol Sci. https://doi.org/10.3390/ijms21103583
Jiang X, Li D, Shen W, Shen X, Liu Y (2019) LncRNA NEAT1 promotes hypoxia-induced renal tubular epithelial apoptosis through downregulating miR-27a-3p. J Cell Biochem 120:16273–16282. https://doi.org/10.1002/jcb.28909
Kang HM, Ahn SH, Choi P, Ko YA, Han SH, Chinga F et al (2015) Defective fatty acid oxidation in renal tubular epithelial cells has a key role in kidney fibrosis development. Nat Med 21:37–46. https://doi.org/10.1038/nm.3762
Kaucsár T, Révész C, Godó M, Krenács T, Albert M, Szalay CI et al (2013) Activation of the miR-17 family and miR-21 during murine kidney ischemia-reperfusion injury. Nucleic Acid Ther 23:344–354
Kölling M, Genschel C, Kaucsar T, Hübner A, Rong S, Schmitt R et al (2018) Hypoxia-induced long non-coding RNA Malat1 is dispensable for renal ischemia/reperfusion-injury. Sci Rep 8:3438
Kroening S, Neubauer E, Wessel J, Wiesener M, Goppelt-Struebe M (2009) Hypoxia interferes with connective tissue growth factor (CTGF) gene expression in human proximal tubular cell lines. Nephrol Dial Transplant 24:3319–3325. https://doi.org/10.1093/ndt/gfp305
Lan R, Geng H, Singha PK, Saikumar P, Bottinger EP, Weinberg JM et al (2016) Mitochondrial pathology and glycolytic shift during proximal tubule atrophy after ischemic AKI. J Am Soc Nephrol 27:3356–3367. https://doi.org/10.1681/asn.2015020177
Lash Lh TJ, Eb W, Bm P (1993) Hypoxia and oxygen dependence of cytotoxicity in renal proximal tubular and distal tubular cells. Biochem Pharmacol 45:191–200
Leonard MO, Cottell DC, Godson C, Brady HR, Taylor CT (2003) The role of HIF-1α in transcriptional regulation of the proximal tubular epithelial cell response to hypoxia. J Biol Chem 278:40296–40304. https://doi.org/10.1074/jbc.M302560200
Leong CL, Anderson WP, O’connor PM, Evans RG (2007) Evidence that renal arterial-venous oxygen shunting contributes to dynamic regulation of renal oxygenation. Am J Physiol Renal Physiol 292:F1726–F1733. https://doi.org/10.1152/ajprenal.00436.2006
Levy MN, Sauceda G (1959) Diffusion of oxygen from arterial to venous segments of renal capillaires. Am J Physiol 196:1336–1339. https://doi.org/10.1152/ajplegacy.1959.196.6.1336
Li X, Wu Z, Yang J, Zhang D (2022) LncRNA 148400 promotes the apoptosis of renal tubular epithelial cells in ischemic AKI by targeting the miR-10b-3p/GRK4 axis. Cells. https://doi.org/10.3390/cells11243986
Li Z, Lu S, Li X (2021) The role of metabolic reprogramming in tubular epithelial cells during the progression of acute kidney injury. Cell Mol Life Sci 78:5731–5741. https://doi.org/10.1007/s00018-021-03892-w
Liao Y, Peng X, Li X, Wu D, Qiu S, Tang X et al (2022) CircRNA_45478 promotes ischemic AKI by targeting the miR-190a-5p/PHLPP1 axis. Faseb J 36:e22633. https://doi.org/10.1096/fj.202201070R
Liu D, Liu Y, Zheng X, Liu N (2021) c-MYC-induced long noncoding RNA MEG3 aggravates kidney ischemia-reperfusion injury through activating mitophagy by upregulation of RTKN to trigger the Wnt/β-catenin pathway. Cell Death Dis 12:191. https://doi.org/10.1038/s41419-021-03466-5
Liu J, Li X, Yang J, Zhang D (2022) LncRNA ENSMUST_147219 mediates the progression of ischemic acute kidney injury by targeting the miR-221-5p/IRF6 axis. Apoptosis 27:531–544. https://doi.org/10.1007/s10495-022-01730-3
Liu L-L, Li D, He Y-L, Zhou Y-Z, Gong S-H, Wu L-Y et al (2017) miR-210 protects renal cell against hypoxia-induced apoptosis by targeting HIF-1 alpha. Mol Med 23:258–271
Manotham K, Tanaka T, Matsumoto M, Ohse T, Inagi R, Miyata T et al (2004) Transdifferentiation of cultured tubular cells induced by hypoxia. Kidney Int 65:871–880. https://doi.org/10.1111/j.1523-1755.2004.00461.x
Markham A (2021) Enarodustat: first approval. Drugs 81:169–174
Maxwell PH, Osmond MK, Pugh CW, Heryet A, Nicholls LG, Tan CC et al (1993) Identification of the renal erythropoietin-producing cells using transgenic mice. Kidney Int 44:1149–1162. https://doi.org/10.1038/ki.1993.362
Miao A-F, Liang J-X, Yao L, Han J-L, Zhou L-J (2021) Hypoxia-inducible factor prolyl hydroxylase inhibitor roxadustat (FG-4592) protects against renal ischemia/reperfusion injury by inhibiting inflammation. Ren Fail 43:803–810
Miao M, Wu M, Li Y, Zhang L, Jin Q, Fan J, et al (2022) Clinical potential of hypoxia inducible factors prolyl hydroxylase inhibitors in treating nonanemic diseases. Frontiers Pharmacol 13:837249. https://doi.org/10.3389/fphar.2022.837249
Nakagawa TLH, Hj Z, Kang Dh, Gf S, Rj J (2004) Differential regulation of VEGF by TGF-β and hypoxia in rat proximal tubular cells. Am J Physiol Renal Physiol 287:F658–F664
Nakamura M, Yamabe H, Osawa H, Nakamura N, Shimada M, Kumasaka R et al (2006) Hypoxic conditions stimulate the production of angiogenin and vascular endothelial growth factor by human renal proximal tubular epithelial cells in culture. Nephrol Dial Transplant 21:1489–1495. https://doi.org/10.1093/ndt/gfl041
Nakatsuji M, Urakami-Takebayashi Y, Miyadokoro S, Ikeda T, Takehara I, Sun H et al (2020) Fatty acids bound to albumin induce prostaglandin E2 production in human renal proximal tubular epithelial cell line HK-2. Biochem Biophys Res Commun 530:273–277. https://doi.org/10.1016/j.bbrc.2020.07.033
Nangaku M, Kondo K, Ueta K, Kokado Y, Kaneko G, Matsuda H et al (2021) Efficacy and safety of vadadustat compared with darbepoetin alfa in Japanese anemic patients on hemodialysis: a phase 3, multicenter, randomized, double-blind study. Nephrol Dial Transpl 36:1731–1741
Ndibalema A, Kabuye D, Wen S, Li L, Li X, Fan Q (2020) Empagliflozin protects against proximal renal tubular cell injury induced by high glucose via regulation of hypoxia-inducible factor 1-Alpha. Diabetes, Metab Syndr Obes Targets Ther 13:1953–1967. https://doi.org/10.2147/dmso.S243170
Nourbakhsh N, Singh P (2014) Role of renal oxygenation and mitochondrial function in the pathophysiology of acute kidney injury. Nephron Clin Pract 127:149–152. https://doi.org/10.1159/000363545
Orphanides C, Fine LG, Norman JT (1997) Hypoxia stimulates proximal tubular cell matrix production via a TGF-β1-independent mechanism. Kidney Int 52:637–647. https://doi.org/10.1038/ki.1997.377
Packer M (2021) Mechanisms leading to differential hypoxia-inducible factor signaling in the diabetic kidney: modulation by SGLT2 inhibitors and hypoxia mimetics. Am J Kidney Dis 77:280–286. https://doi.org/10.1053/j.ajkd.2020.04.016
Pan J, Zhang G, Hu Y, Jiang H, Tang X, Zhang D (2022) MiR-6918–5p prevents renal tubular cell apoptosis by targeting MBD2 in ischemia/reperfusion-induced AKI. Life Sci 308:120921. https://doi.org/10.1016/j.lfs.2022.120921
Piao SG, Kang SH, Lim SW, Chung BH, Doh KC, Heo SB et al (2013) Influence of N-acetylcysteine on klotho expression and its signaling pathway in experimental model of chronic cyclosporine nephropathy in mice. Transplantation 96:146–153
Provenzano R, Besarab A, Sun CH, Diamond SA, Durham JH, Cangiano JL et al (2016) Oral hypoxia–inducible factor prolyl hydroxylase inhibitor roxadustat (FG-4592) for the treatment of anemia in patients with CKD. Clin J Am Soc Nephrol 11:982–991
Provenzano R, Tumlin J, Zabaneh R, Chou J, Hemmerich S, Neff TB et al (2020) Oral hypoxia-inducible factor prolyl hydroxylase inhibitor roxadustat (FG-4592) for treatment of anemia in chronic kidney disease: a placebo-controlled study of pharmacokinetic and pharmacodynamic profiles in hemodialysis patients. J Clin Pharmacol 60:1432–1440
Romano AH, Conway T (1996) Evolution of carbohydrate metabolic pathways. Res Microbiol 147:448–455. https://doi.org/10.1016/0923-2508(96)83998-2
Rudnicki M, Perco P, Enrich J, Eder S, Heininger D, Bernthaler A et al (2009) Hypoxia response and VEGF-A expression in human proximal tubular epithelial cells in stable and progressive renal disease. Lab Invest 89:337–346. https://doi.org/10.1038/labinvest.2008.158
Schödel J, Ratcliffe P (2019) Mechanisms of hypoxia signalling: new implications for nephrology. Nat Rev Nephrol 15:641–659
Seagroves TN, Ryan HE, Lu H, Wouters BG, Knapp M, Thibault P et al (2001) Transcription factor HIF-1 is a necessary mediator of the pasteur effect in mammalian cells. Mol Cell Biol 21:3436–3444. https://doi.org/10.1128/mcb.21.10.3436-3444.2001
Shen Y, Jiang L, Wen P, Ye Y, Zhang Y, Ding H et al (2020) Tubule-derived lactate is required for fibroblast activation in acute kidney injury. Am J Physiol Renal Physiol 318:F689-f701. https://doi.org/10.1152/ajprenal.00229.2019
Shu S, Wang Y, Zheng M, Liu Z, Cai J, Tang C et al (2019) Hypoxia and hypoxia-inducible factors in kidney injury and repair. Cells 8:207
Simon N, Hertig A (2015) Alteration of fatty acid oxidation in tubular epithelial cells: from acute kidney injury to renal fibrogenesis. Front Med (lausanne) 2:52. https://doi.org/10.3389/fmed.2015.00052
Singh A, Rattan S (2021) BDNF rescues aging-associated internal anal sphincter dysfunction. Am J Physiol Gastrointest Liver Physiol 321:G87–G97
Singh A, Singh J, Rattan SJN, Motility, (2022) Evidence for the presence and release of BDNF in the neuronal and non-neuronal structures of the internal anal sphincter. Neurogastroenterology 34:e14099
Song H, Han I-Y, Kim Y, Kim YH, Choi I-W, Seo S-K et al (2015) The NADPH oxidase inhibitor DPI can abolish hypoxia-induced apoptosis of human kidney proximal tubular epithelial cells through Bcl2 up-regulation via ERK activation without ROS reduction. Life Sci 126:69–75. https://doi.org/10.1016/j.lfs.2015.02.004
Tanaka S, Tanaka T, Nangaku M (2016) Hypoxia and hypoxia-inducible factors in chronic kidney disease. Renal Replace Ther 2:1–8
Tanaka T, Hanafusa N, Ingelfinger JR, Ohse T, Fujita T, Nangaku M (2003) Hypoxia induces apoptosis in SV40-immortalized rat proximal tubular cells through the mitochondrial pathways, devoid of HIF1-mediated upregulation of Bax. Biochem Biophys Res Commun 309:222–231. https://doi.org/10.1016/s0006-291x(03)01557-2
Tang B, Li W, Ji T, Li X, Qu X, Feng L et al (2020) Downregulation of XIST ameliorates acute kidney injury by sponging miR-142-5p and targeting PDCD4. J Cell Physiol 235:8852–8863. https://doi.org/10.1002/jcp.29729
Tao YS, Piao SG, Jin YS, Jin JZ, Zheng HL, Zhao HY et al (2018) Expression of brain-derived neurotrophic factor in kidneys from normal and cyclosporine-treated rats. BMC Nephrol 19:1–12
Wang IK, Palanisamy K, Sun KT, Yu SH, Yu TM, Li CH et al (2020) The functional interplay of lncRNA EGOT and HuR regulates hypoxia-induced autophagy in renal tubular cells. J Cell Biochem 121:4522–4534. https://doi.org/10.1002/jcb.29669
Welch WJ, Baumgärtl H, Lübbers D, Wilcox CS (2001) Nephron pO2 and renal oxygen usage in the hypertensive rat kidney. Kidney Int 59:230–237. https://doi.org/10.1046/j.1523-1755.2001.00483.x
Wenger RH, Hoogewijs D (2010) Regulated oxygen sensing by protein hydroxylation in renal erythropoietin-producing cells. Am J Physiol Renal Physiol 298:F1287–F1296. https://doi.org/10.1152/ajprenal.00736.2009
Xi X, Zou C, Ye Z, Huang Y, Chen T, Hu H (2019) Pioglitazone protects tubular cells against hypoxia/reoxygenation injury through enhancing autophagy via AMPK-mTOR signaling pathway. Eur J Pharmacol 863:172695. https://doi.org/10.1016/j.ejphar.2019.172695
Xiang H, Xue W, Li Y, Zheng J, Ding C, Dou M et al (2020) Knockdown of ANGPTL2 protects renal tubular epithelial cells against hypoxia/reoxygenation-induced injury via suppressing TLR4/NF-kappaB signaling pathway and activating Nrf2/HO-1 signaling pathway. Cell Transplant 29:963689720946663. https://doi.org/10.1177/0963689720946663
Xie LB, Chen B, Liao X, Chen YF, Yang R, He SR et al (2020) LINC00963 targeting miR-128-3p promotes acute kidney injury process by activating JAK2/STAT1 pathway. J Cell Mol Med 24:5555–5564. https://doi.org/10.1111/jcmm.15211
Xu J, Wang B, Zhang D (2022) LncRNA ENSMUST00000171502 induced by HIF-1α ameliorates ischemic acute kidney injury via targeting the miR-130b-3p/Mybl-1 axis. Cells. https://doi.org/10.3390/cells11233747
Yamamoto H, Nobori K, Matsuda Y, Hayashi Y, Hayasaki T, Akizawa T (2021) Molidustat for renal anemia in nondialysis patients previously treated with erythropoiesis-stimulating agents: a randomized, open-label, phase 3 study. Am J Nephrol 52:884–893
Yan Z, Zang B, Gong X, Ren J, Wang RJ (2020) MiR-214-3p exacerbates kidney damages and inflammation induced by hyperlipidemic pancreatitis complicated with acute renal injury. Life Sci 241:117118
Yang L, Wang B, Ma L, Fu PJFIP (2022) An update of long-noncoding RNAs in acute kidney injury. Front Physiol. https://doi.org/10.3389/fphys.2022.849403
Yaqoob MEC, Rw S (1996) Role of nitric oxide and superoxide balance in hypoxia–reoxygenation proximal tubular injury. Nephrol Dial Transplant 11:1743–1746
Yaqoob M, Edelstein CL, Wieder ED, Alkhunaizi AM, Gengaro PE, Nemenoff RA et al (1996) Nitric oxide kinetics during hypoxia in proximal tubules: effects of acidosis and glycine. Kidney Int 49:1314–1319. https://doi.org/10.1038/ki.1996.187
Yu WSM, Xu R, Yu J, Cui K, Tong J, Shi L, Ren H, Du H (2013) Berberine protects human renal proximal tubular cells from hypoxia/reoxygenation injury via inhibiting endoplasmic reticulum and mitochondrial stress pathways. J Transl Med 11:1–10
Yuan Q, Lv Y, Ding H, Ke Q, Shi C, Luo J et al (2021a) CPT1α maintains phenotype of tubules via mitochondrial respiration during kidney injury and repair. Cell Death Dis 12:792. https://doi.org/10.1038/s41419-021-04085-w
Yuan Xp LL, Cb C, Zhou J, Yt Z, Xp W, Han M, Cx W (2017) MicroRNA-423-5p facilitates hypoxia/reoxygenation-induced apoptosis in renal proximal tubular epithelial cells by targeting GSTM1 via endoplasmic reticulum stress. Oncotarget 8:82064–82077
Yuan Y, Li X, Chu Y, Ye G, Yang L, Dong Z (2021b) Long non-coding RNA H19 augments hypoxia/reoxygenation-induced renal tubular epithelial cell apoptosis and injury by the miR-130a/BCL2L11 pathway. Front Physiol 12:632398
Zager RA, Johnson AC, Lund S, Randolph-Habecker J (2007) Toll-like receptor (TLR4) shedding and depletion: acute proximal tubular cell responses to hypoxic and toxic injury. Am J Physiol Renal Physiol 292:F304–F312. https://doi.org/10.1152/ajprenal.00237.2006
Zager RA, Johnson ACM, Hanson SY (2004) Proximal tubular cytochrome c efflux: determinant, and potential marker, of mitochondrial injury. Kidney Int 65:2123–2134. https://doi.org/10.1111/j.1523-1755.2004.00638.x
Zhang H, Xu R, Wang Z (2021) Contribution of oxidative stress to HIF-1-mediated profibrotic changes during the kidney damage. Oxid Med Cell Longev 2021:6114132. https://doi.org/10.1155/2021/6114132
Zhang L, Liu L, Bai M, Liu M, Wei L, Yang Z et al (2019) Hypoxia-induced HE4 in tubular epithelial cells promotes extracellular matrix accumulation and renal fibrosis via NF-κB. FASEB J 34:2554–2567. https://doi.org/10.1096/fj.201901950R
Zhang M, Dong R, Yuan J, Da J, Zha Y, Long Y et al (2022) Roxadustat (FG-4592) protects against ischaemia/reperfusion-induced acute kidney injury through inhibiting the mitochondrial damage pathway in mice. Clin Experiment Pharmacol 49:311–318
Zhao H, Jiang N, Han Y, Yang M, Gao P, Xiong X et al (2020) Aristolochic acid induces renal fibrosis by arresting proximal tubular cells in G2/M phase mediated by HIF-1alpha. FASEB J 34:12599–12614. https://doi.org/10.1096/fj.202000949R
Funding
This work was supported by the Kidney Research Center at Tabriz University of Medical Sciences, Tabriz, Iran (Grant # 71526).
Author information
Authors and Affiliations
Contributions
MA and SZV designed the article. YRS, SMHKh, and AS wrote the manuscript. SZV and MA revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rahbar Saadat, Y., Hosseiniyan Khatibi, S.M., Sani, A. et al. Ischemic tubular injury: Oxygen-sensitive signals and metabolic reprogramming. Inflammopharmacol 31, 1657–1669 (2023). https://doi.org/10.1007/s10787-023-01232-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-023-01232-x