Abstract
Background
Immunization with killed Leishmania promastigotes without adjuvant was considered as safe, but gave variable rates of protection. Taking advantage of the immuno-modulatory effect of caffeic acid (CA), a natural polyphenolic antioxidant, we investigated its potentiating effect in autoclaved Leishmania major (ALM)-induced protection of Leishmania major-infected BALB/c.
Methods
First, BALB/c mice were sensitized for 6 weeks either with CA, or ALM alone or combined with caffeic acid (ALM–CA) or Freund’s adjuvant (ALM–FA), and subsequently infected with L. major promastigotes. Second, to test the curative effect, CA was given daily for 5 weeks to susceptible mice, starting on week 4 post-infection. Sera, footpads and lymph nodes (LNs) were collected at week 9 post-infection and submitted to biochemical or histological analyses.
Results
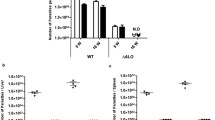
Compared to the respective controls, our results showed that CA directly healed footpad lesions and reduced the hallmarks of cutaneous leishmaniasis including oxidative inflammation, parasite load, and phagocytes influx and infestation. In sensitized mice, the protection enhanced gradually from ALM–FA, CA, ALM to ALM–CA in parallel to decreased seric IgGt levels. In contrast to ALM–FA, the combined effect of ALM and CA increased specific isotype IgG2, and decreased IL-17 and MCP-1, and phagocyte influx, as attested by the concomitant reduction in myeloperoxidase (MPO) and α-naphthyl acetate esterase (ANAE) activities. ALM–CA shifted IFN-γ/TGF-β and iNO synthase/arginase1 (iNOS/Arg1) balances in a Th1 immune response that control efficiently cutaneous lesions and LNs hypertrophy, and reactivate the death of infected phagocytes.
Conclusions
Therefore, CA combined with ALM synergizes with L. major antigens for priming innate cells, through early polarization to optimal Th1 response that leads to IFN-γ and iNOS-dependent leishmanicidal activity of neutrophils and macrophages.
Graphical Abstract








Similar content being viewed by others
Abbreviations
- Arg1:
-
Arginase1
- ALM:
-
Autoclaved Leishmania major
- ANAE:
-
α-Naphthyl acetate esterase
- CA:
-
Caffeic acid
- DCs:
-
Dendritic cells
- iNOS:
-
Inducible NO synthase
- IFN-γ:
-
Interferon-γ
- IL-17A:
-
Interleukin 17A
- LNs:
-
Lymph nodes
- MCP-1:
-
Monocyte chemoattractant protein 1
- MO:
-
Macrophages
- MPO:
-
Myeloperoxidase
- PN:
-
Neutrophils
- ROS:
-
Reactive oxygen species
- TGF-β:
-
Tumor growth factor-β
References
Abi Abdallah DS, Denkers ET (2012) Neutrophils cast extracellular traps in response to protozoan parasites. Front Immunol 3:1–6
Bahar K, Dowlati LY, Shidani B, Alimohammadian MH, Khamesipour A, Ehsasi S et al (1996) Comparative safety and immunogenicity trial of two killed Leishmania major vaccines with or without BCG in human volunteers. Clin Dermatol 14(5):489–495
Belazzoug S (1985) A new enzymatic variant of Leishmania infantum Nicolle, 1908, agent of cutaneous leishmaniasis in northern Algeria. Ann Parasitol Hum Comp 60(1):1–3
Belkhelfa-Slimani R, Djerdjouri B (2017) Caffeic acid and quercetin exert caspases-independent apoptotic effects on Leishmania major promastigotes, and reactivate the death of infected phagocytes derived from BALB/c mice. Asian Pac J Trop Biomed 7(4):321–331
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brunner R, Jensen-Jarolim E, Pali-Schöll I (2010) The ABC of clinical and experimental adjuvants—a brief overview. Immunol Lett 128(1):29–35
Buffet PA, Sulahian A, Garin YJ, Nassar N, Derouin F (1995) Culture microtitration: a sensitive method for quantifying Leishmania infantum in tissues of infected mice. Antimicrob Agents Chemother 39(9):2167–2168
Conrad SM, Strauss-Ayali D, Field AE, Mack M, Mosser DM (2007) Leishmania-derived murine monocyte chemoattractant protein 1 enhances the recruitment of a restrictive population of CC Chemokine receptor 2-positive macrophages. Infect Immun 75(2):653–665
Corraliza IM, Soler G, Eichmann K, Modolell M (1995) Arginase induction by suppressors of nitric oxide synthesis (IL-4, IL-10 and PGE2) in murine bone marrow-derived macrophages. Biochem Biophys Res Commun 206:667–673
Darrah PA, Patel DT, De Luca PM, Lindsay RW, Davey DF, Flynn BJ et al (2007) Multifunctional TH1 cells define a correlate of vaccine-mediated protection against Leishmania major. Nat Med 13:843–850
De Luca PM, Barreto Macedo AB (2016) Cutaneous leishmaniasis vaccination: a matter of quality. Front Immun 7:151
Desjeux P (2004) Leishmaniasis: current situation and new perspectives. Comp Immunol Microb Infect Dis 27:305–318
Ding AH, Nathan CF, Stuehr DJ (1988) Release of reactive nitrogen intermediates and reactive oxygen intermediates from mouse peritoneal macrophages: comparison of activating cytokines and evidence for independent production. J Immunol 141:2407–2412
Gantt KR, Schultz-Cherry S, Rodriguez N, Jeronimo SMB, Nascimento ET, Goldman TL et al (2003) Activation of TGF-β by Leishmania chagasi: importance for parasite survival in macrophages. J Immunol 170(5):2613–2620
Gonzalez-Lombana C, Gimblet C, Bacellar O, Oliveira WW, Passos S et al (2013) IL-17 mediates immunopathology in the absence of IL-10 following Leishmania major infection. PLoS Pathog 9(3):e1003243
Gutiérrez-Kobeh L, de Oyarzabal E, Argueta J, Wilkins A, Salaiza N, Fernandez E et al (2013) Inhibition of dendritic cell apoptosis by Leishmania mexicana amastigotes. Parasitol Res 112(4):1755–1762
Hadmman E (2001) Leishmaniasis: current status of vaccine development. Clin Microb Rev 14(2):229–243
Harrat Z, Boubidi SC, Pratlong F, Benikhlef R, Selt B, Dedet JP et al (2009) Description of a dermatropic Leishmania close to L. killicki (Rioux, Lanotte & Pratlong 1986) in Algeria. Trans R Soc Trop Med Hyg 103:16–20
Hurrell BP, Schuster S, Grün E, Coutaz M, Williams RA, Held W et al (2015) Rapid sequestration of Leishmania mexicana by neutrophils contributes to the development of chronic lesion. PLoS Pathog 11(5):e1004929
Iniesta V, Carcelen J, Molano I, Peixoto PMV, Redondo E, Parra P, Mangas M et al (2005) Arginase I induction during Leishmania major infection mediates the development of disease. Infect Immun 73(9):6085–6090
Jochim RC, Teixeira C (2009) Leishmania commandeers the host inflammatory response through neutrophils. Trends Parasitol 25(4):145–147
Katara GK, Ansari NA, Singh A, Ramesh V, Salotra P (2012) Evidence for involvement of Th17 type responses in post Kala Azar dermal Leishmaniasis (PKDL). PLoS Negl Trop Dis 6(6):e1703
Khamesipour A, Rafati S, Davoudi N, Maboudi F, Modabber F (2006) Leishmaniasis vaccine candidates for development: a global overview. Ind J Med Res 123:423–438
Krawisz JE, Sharon P, Stenson WF (1984) Quantitative assay for acute intestinal inflammation based on myeloperoxidase activity. Assessment of inflammation in rat and hamster models. Gastroenterology 87(6):1344–1350
Kumar R, Bumb RA, Salotra P (2009) Evaluation of localized and systemic immune responses in cutaneous leishmaniasis caused by Leishmania tropica: interleukin-8, monocyte chemotactic protein-1 and nitric oxide are major regulatory factors. Immunology 130:193–201
Lagranderie MRR, Balazuc AM, Deriaud E, Leclerc CD, Gheorghiu M (1996) Comparison of immune responses of mice immunized with five different Mycobacterium bovis BCG vaccine strains. Infect Immun 64(1):1–9
Laskay T, van Zandbergen G, Solbach W (2008) Neutrophil granulocytes as host cells and transport vehicles for intracellular pathogens: apoptosis as infection-promoting factor. Immunobiology 213:183–191
Li J, Hunter CA, Farrell JP (1999) Anti-TGF-β treatment promotes rapid healing of Leishmania major infection in mice by enhancing in vivo nitric oxide production. J Immunol 162:974–979
Lopes MF, Costa-da-Silva AC, DosReis GA (2014) Innate immunity to Leishmania infection: within phagocytes. Mediat Inflam 2014:754965
Lopez Kostka S, Dinges S, Griewank K, Iwakura Y, Udey MC, von Stebut E (2009) IL-17 promotes progression of cutaneous leishmaniasis in susceptible mice. J Immunol 182:3039–3046
Mukherjee P, Sen PC, Ghose AC (2006) Lymph node cells from BALB/c mice with chronic visceral leishmaniasis exhibiting cellular anergy and apoptosis: involvement of Ser/Thr phosphatase. Apoptosis 11(11):2013–2029
Mutiso JM, Macharia JC, Mutisya RM, Taracha E (2010) Subcutaneous immunization against Leishmania major-infection in mice: efficacy of formalin-killed promastigotes combined with adjuvants. Rev Inst Med Trop Sao Paulo 52(2):95–100
Ng LG, Hsu A, Mandell MA, Roediger B, Hoeller C, Mrass P et al (2008) Migratory dermal dendritic cells act as rapid sensors of protozoan parasites. PLoS Pathog 4:e1000222
Okwor I, Mou Z, Liu D, Uzonna J (2012) Protective immunity and vaccination against cutaneous Leishmaniasis. Front Immunol 3:1–13
Paiva CN, Feijo DF, Dutra FF, Carneiro VC, Freitas GB, Alves LS et al (2012) Oxidative stress fuels Trypanosoma cruzi infection in mice. J Clin Invest 122(7):2531–2542
Parody N, Soto M, Requena JM, Alonso C (2004) Adjuvant guided polarization of the immune humoral response against a protective multicomponent antigenic protein (Q) from Leishmania infantum. A CpG + Q mix protects BALB/c mice from infection. Parasite Immunol 26:283–293
Pereira L, Abbehusen M, Teixeira C, Cunha J, Nascimento IP, Fukutani K et al (2015) Vaccination with Leishmania infantum acidic ribosomal P0 but not with nucleosomal histones proteins controls Leishmania infantum infection in hamsters. PLoS Negl Trop Dis 9(2):e0003490
Peters NC, Sacks D (2009) The impact of vector mediated neutrophil recruitment on cutaneous Leishmaniasis. Cell Microbiol 11(9):1290–1296
Peters NC, Egen JG, Secundino N, Debrabant A, Kimblin N, Kamhawi S et al (2008) In vivo imaging reveals an essential role for neutrophils in Leishmaniasis transmitted by sand flies. Science 321(5891):970–974
Peters NC, Bertholet S, Lawyer PG, Charmoy M, Romano A, Ribeiro-Gomes FL et al (2012) Evaluation of recombinant Leishmania polyprotein plus glucopyranosyl lipid a stable emulsion vaccines against sand fly-transmitted Leishmania major in C57BL/6 Mice. J Immunol 189:4832–4841
Pinheiro RO, Pinto EF, Lopes JRC, Guedes HLM, Fentanes RF, Rossi-Bergmann B (2005) TGF-β-associated enhanced susceptibility to leishmaniasis following intramuscular vaccination of mice with Leishmania amazonensis antigens. Microbes Infect 7(13):1317–1323
Radtke OA, Foo LY, Lu Y, Kiderlen AF, Kolodziej H (2003) Evaluation of sage phenolics for their antileishmanial activity and modulatory effects on interleukin-6, interferon and tumour necrosis factor-alpha-release in RAW 264.7 cells. Z Naturforschung C58(5–6):395–400
Ribeiro-Gomes FL, Peters NC, Debrabant A, Sacks DL (2012) Efficient capture of infected neutrophils by dendritic cells in the skin inhibits the early anti-Leishmania response. PLoS Pathog 8(2):e1002536
Ribeiro-Gomes FL, Romano A, Lee S, Roffê E, Peters NC, Debrabant A, Sacks D (2015) Apoptotic cell clearance of Leishmania major-infected neutrophils by dendritic cells inhibits CD8+ T-cell priming in vitro by Mer tyrosine kinase-dependent signaling. Cell Death Dis 6:e2018
Rosa R, Marques C, Rodrigues OR, Santos-Gomes GM (2007) Immunization with Leishmania infantum released proteins confers partial protection against parasite infection with a predominant Th1 specific immune response. Vaccine 25:4525–4532
Sacks D, Anderson C (2004) Re-examination of the immunosuppressive mechanisms mediating non-cure of Leishmania infection in mice. Immunol Rev 201(1):225–238
Sarkar A, Aga E, Bussmeyer U, Bhattacharyya A, Möller S, Hellberg L et al (2013) Infection of neutrophil granulocytes with Leishmania major activates ERK 1/2 and modulates multiple apoptotic pathways to inhibit apoptosis. Med Microbiol Immunol 202:25–35
Scott P (2005) Micro review: immunologic memory in cutaneous Leishmaniasis. Cell Microb 12:1707–1713
Soares-Silva M, Diniz F, Gomes GN, Diana B (2016) The mitogen activated protein kinase (MAPK) pathway: role in immune evasion by trypanosomatids. Front Microb doi: 10.3389/fmicb.2016.00183
Tounsi N, Meghari S, Moser M, Djerdjouri B (2015) Lysophosphatidylcholine exacerbates Leishmania major-dendritic cell infection through interleukin-10 and a burst in arginase1 and indoleamine 2,3-dioxygenase activities. Int Immunopharmacol 25(1):1–9
Van Zandbergen G, Klinger M, Mueller A, Dannenberg S, Gebert A, Solbach W, Laskay T (2004) Cutting edge: neutrophil granulocyte serves as a vector for Leishmania entry into macrophages. J Immunol. 173(11):6521–6525
Wahl SM, Swisher J, McCartney-Francis N, Chen W (2004) TGF-β: the perpetrator of immune suppression by regulatory T cells and suicidal T cells. J Leukoc Biol 76:15–24
Zhang WW, Matlashewski G (2008) Immunization with a toll-like receptor 7 and/or 8 agonist vaccine adjuvant increases protective immunity against Leishmania major in BALB/c Mice. Infect Immun 76(8):3777–3783
Zhou X, Scharf ME, Meinke LJ, Chandler LD, Siegfried BD (2003) Characterization of general esterases from methyl parathion-resistant and -susceptible populations of western corn rootworm (Coleoptera: Chrysomelidae). J Econ Entomol 96(6):1855–1863
Acknowledgements
We are grateful to Pr Zoubir Harrat from “Laboratoire d’Eco-Epidémiologie et Génétique des Populations, Institut Pasteur, Algiers, Algeria”, for her kind gift of Leishmania major inoculum.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Belkhelfa-Slimani, R., Djerdjouri, B. Caffeic acid combined with autoclaved Leishmania major boosted the protection of infected BALB/c mice by enhancing IgG2 production, IFN-γ/TGF-β and iNO synthase/arginase1 ratios, and the death of infected phagocytes. Inflammopharmacol 26, 621–634 (2018). https://doi.org/10.1007/s10787-017-0399-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-017-0399-z