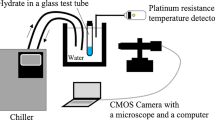
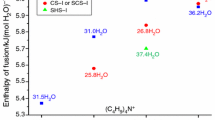
Abstract
Ionic semiclathrate hydrates mainly consist of water typically together with tetrabutylammonium and tetrabutylphosphonium salts. Since ionic semiclathrate hydrates have the large dissociation heat under ambient pressure and temperature conditions, various ionic semiclathrate hydrates have been studied as safety and eco-friendly phase change materials. In this study, tetrabutylammonium acetate hydrates and tetrabutylammonium hydroxide hydrates were proposed as thermal energy storage media for air conditioning and cooling lithium-ion batteries. The dissociation heat, which was a significant thermophysical property to design thermal energy storage systems, were measured at various mass fractions. The largest dissociation heats of tetrabutylammonium acetate hydrates and tetrabutylammonium hydroxide hydrates were 212.9 ± 0.9 kJ⋅kg−1 and 200.4 ± 2.2 kJ⋅kg−1. As a result of the comparison of the dissociation heats of tetrabutylammonium acetate hydrates and tetrabutylammonium hydroxide hydrates with those of other ionic semiclathrate hydrates, it was found that tetrabutylammonium acetate hydrates and tetrabutylammonium hydroxide hydrates had the promising thermophysical properties as thermal energy storage media for air conditioning and cooling lithium-ion batteries, respectively.







Similar content being viewed by others
Data Availability
All datasets presented in this study are included in the article/supplementary files.
References
Y.A. Dyadin, K.A. Udachin, J. Incl. Phenom. 2, 61–72 (1984). https://doi.org/10.1007/BF00663240
G.A. Jefferey, Acc. Chem. Res. 2, 344–352 (1969). https://doi.org/10.1021/ar50023a004
K. Sato, H. Tokutomi, R. Ohmura, Fluid Phase Equilib. 337, 115–118 (2013). https://doi.org/10.1016/j.fluid.2012.09.016
Y. Arai, R. Koyama, F. Endo, A. Hotta, R. Ohmura, J. Chem. Thermodyn. 131, 330–335 (2019). https://doi.org/10.1016/j.jct.2018.11.017
Y. Yamauchi, T. Yamasaki, F. Endo, A. Hotta, R. Ohmura, Chem. Eng. Technol. 40, 1810–1816 (2017). https://doi.org/10.1002/ceat.201600459
T. Iwai, T. Miyamoto, N. Kurokawa, A. Hotta, R. Ohmura, J. Energy Storage 52, 104801 (2022). https://doi.org/10.1016/j.est.2022.104801
J. Gholinezhad, A. Chapoy, B. Tohidi, Chem. Eng. Res. Des. 89, 1747–1751 (2011). https://doi.org/10.1016/j.cherd.2011.03.008
K. Iino, Y. Sakakibara, T. Suginaka, R. Ohmura, J. Chem. Thermodyn. 71, 133–136 (2014). https://doi.org/10.1016/j.jct.2013.12.001
T. Kobori, S. Muromachi, R. Ohmura, J. Chem. Eng. Data 60, 299–303 (2015). https://doi.org/10.1021/je500589z
H. Akiba, R. Ohmura, J. Chem. Thermodyn. 97, 83–87 (2016). https://doi.org/10.1016/j.jct.2016.01.014
R.V. Belosludov, R.K. Zhdanov, K.V. Gets, Y.Y. Bozhko, V.R. Belosludov, Y. Kawazoe, J. Phys. Chem. 124, 18474–18481 (2020). https://doi.org/10.1021/acs.jpcc.0c05947
M. Maruyama, S. Kao, H. Kiyokawa, S. Takeya, R. Ohmura, Energy Fuels 36, 10601–10609 (2022). https://doi.org/10.1021/acs.energyfuels.2c01355
A.A. Karimi, O. Dolotko, D. Dalmazzone, Fluid Phase Equib. 361, 175–180 (2014). https://doi.org/10.1016/j.fluid.2013.10.043
ASHRAE TC 9.9 2011 Thermal Guidelines for Data Processing Environments—Expanded Data Center Classes and Usage Guidance. (American Society of Heating, Refrigerating and Air-Conditioning Engineers Inc., 2011), https://www.ashrae.org/. Accessed 2 Nov 2022
B. Shabani, M. Biju, Energies 8, 10153–10177 (2015). https://doi.org/10.3390/en80910153
O. Nashed, J.C.H. Koh, B. Lal, Procedia Eng. 148, 1351–1356 (2016). https://doi.org/10.1016/j.proeng.2016.06.586
H. Nakayama, S. Torigata, Bull. Chem. Soc. Jpn. 57, 171–174 (1984). https://doi.org/10.1246/bcsj.57.171
R. Feistel, W. Wagner, J. Phys. Chem. Ref. Data 35, 1021–1047 (2006). https://doi.org/10.1063/1.2183324
T.V. Rodinova, I.S. Terekhova, G.V. Villevald, T.D. Karpova, A.Y. Manakov, J. Therm. Anal. Calorim. 128, 1165–1174 (2017). https://doi.org/10.1007/s10973-016-6023-4
T. Iwai, I. Ohta, K. Hiraga, K. Kashima, A. Hotta, R. Ohmura, Fluid Phase Equilib. 562, 113561 (2022). https://doi.org/10.1016/j.fluid.2022.113561
P.G. Lafond, K.A. Olcott, E.D. Sloan, C.A. Koh, A.K. Sum, J. Chem. Thermodyn. 48, 1–6 (2012). https://doi.org/10.1016/j.jct.2011.12.023
W. Lin, D. Dalmazzone, W. Fürst, A. Delahaye, L. Fournaison, P. Clain, J. Chem. Thermodyn. 61, 132–137 (2013). https://doi.org/10.1016/j.jct.2013.02.005
T. Suginaka, H. Sakamoto, K. Iino, S. Takeya, M. Nakajima, R. Ohmura, Fluid Phase Equilib. 317, 25–28 (2012). https://doi.org/10.1016/j.fluid.2011.12.010
J. Shimada, M. Shimada, T. Sugahara, K. Tsunashima, Fluid Phase Equilib. 485, 61–66 (2019). https://doi.org/10.1016/j.fluid.2018.11.038
T.V. Rodinova, A.Y. Manakov, Y.G. Stenin, G.V. Villevald, T.D. Karpova, J. Incl. Macrocycl. Chem. 61, 107–111 (2008). https://doi.org/10.1007/s10847-007-9401-5
Y.A. Dyadin, I.S. Terekhova, T.M. Polyanskaya, L.S. Aladko, J. Struct. Chem. 17, 566–571 (1976). https://doi.org/10.1007/BF00753438
Acknowledgments
The authors gratefully acknowledge the Ohmura Laboratory members for valuable discussion and support.
Funding
This study was supported by a Keirin-racing-based research-promotion fund from the JKA Foundation (2022M-270).
Author information
Authors and Affiliations
Contributions
Conceptualization was contributed by TI, RO; methodology was contributed by TI, ST; formal analysis and investigation were contributed by TI, ST, RO; writing—original draft preparation, was contributed by TI; writing—review and editing, was contributed by RO; funding acquisition was contributed by RO; Resources were contributed by AH, RO; supervision was contributed by RO.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Ethical Approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Iwai, T., Takamura, S., Hotta, A. et al. Measurements of the Dissociation Heats of Tetrabutylammonium Acetate and Tetrabutylammonium Hydroxide Ionic Semiclathrate Hydrates. Int J Thermophys 44, 42 (2023). https://doi.org/10.1007/s10765-022-03150-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-022-03150-6