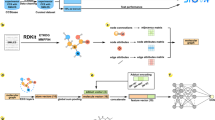
Abstract
The conductor-like screening model with the segment activity coefficient (COSMO-SAC) model enables us to predict the liquid phase behavior based on quantum chemical calculations when experimental information is unavailable. However, the quantum chemical COSMO calculations require significant computational resources (software, hardware, experience, and time) for successful results. In this work, we suggest machine learning models for replacing COSMO calculations to alleviate the computational burden. The machine learning (ML) models include graph convolutional neural networks. The ML models were constructed based on the molecular level structural information and previous COSMO calculation databases. Additional COSMO calculations were generated to train several missing functional groups, which are not included in the existing databases. The ML prediction abilities were shown by comparison of generated cavity volume and sigma profile with the original COSMO-SAC. The vapor–liquid equilibrium (VLE) prediction results were also compared with the original COSMO-SAC and the UNIQUAC functional-group activity coefficients (UNIFAC) model. It is shown that the suggested ML models can predict VLE with reasonable accuracy comparable with the original COSMO-SAC and UNIFAC. The new ML models can effectively replace the time-consuming COSMO-SAC calculations and experimental data-dependent UNIFAC models.
Graphical Abstract




Similar content being viewed by others
Abbreviations
- \({A}_{i}\) :
-
Cavity area of component \(i\) [Å2]
- \({a}_{eff}\) :
-
Effective area parameter [Å2]
- \({A}_{ES}\) :
-
Electrostatic interaction parameter A [kcal Å2·mol−1·e−2]
- \({B}_{ES}\) :
-
Electrostatic interaction parameter B [kcal Å4 K2 mol−1·e−2]
- \({c}_{hb}\) :
-
Hydrogen bonding interaction parameter [kcal Å4·mol−1·e−2]
- \({c}_{OH}\) :
-
OH interaction parameter [kcal Å4·mol−1·e−2]
- \({c}_{OH-OT}\) :
-
OH-OT interaction parameter [kcal Å4·mol−1·e−2]
- \({c}_{OT}\) :
-
OT interaction parameter [kcal Å4·mol−1·e−2]
- \({\widehat{f}}_{i}\) :
-
Partial fugacity of component \(i\) [–]
- \(N\) :
-
Number of learned molecules [–], number of total experimental data points [–]
- \(n\) :
-
Number of grids of screening charge [–]
- \(P\) :
-
Pressure [\({\text{kPa}}\)]
- \({P}_{i}^{sat}\) :
-
Saturation pressure of component i [\({\text{kPa}}\)]
- \(p\left(\sigma \right)\) :
-
Sigma profile [–]
- \({q}_{0}\) :
-
Area normalization parameter [Å2]
- \({r}_{0}\) :
-
Volume normalization parameter [Å3]
- R2 :
-
R-squared score [–]
- \({V}_{i}\) :
-
Cavity volume of component \(i\) [Å3]
- \({x}_{i}\) :
-
Liquid mole fraction of component \(i\) [–]
- \({y}_{i}\) :
-
Vapor mole fraction of component \(i\) [–]
- \(z\) :
-
Coordination number [–]
- \(\Delta P\) :
-
Relative root mean error of pressure [%]
- \(\Delta W\) :
-
Exchange energy [kcal Å4·mol−1]
- \(\Delta y\) :
-
Root mean error of vapor mole fraction [–]
- \(\Gamma \left(\sigma \right)\) :
-
Segment activity coefficient [–]
- \({\gamma }_{i}\) :
-
Activity coefficient of component \(i\) [–]
- \(\widehat{{\phi }_{i}}\) :
-
Partial fugacity coefficient of component \(i\) [–]
- \(\sigma\) :
-
Charge density [e Å2]
- COSMO:
-
Conductor-like screening MOdel
- COSMO-RS:
-
Conductor-like screening MOdel with real solvent
- COSMO-SAC:
-
Conductor-like screening MOdel with segment activity coefficient
- GC:
-
Group contribution
- GCM:
-
Group contribution model
- GC-GCN:
-
Group contribution graph convolution neural network
- HDNN:
-
High-dimensional neural network
- LLE:
-
Liquid–liquid equilibrium
- LSTM:
-
Long short-term memory
- M-GCN:
-
Molecular graph convolution neural network
- ML:
-
Machine learning
- RMSE:
-
Root mean square error
- RRMSE:
-
Relative root mean square error
- SLE:
-
Solid–liquid equilibrium
- UNIFAC:
-
UNIQUAC functional-group activity coefficients
- VLE:
-
Vapor–liquid equilibrium
References
A. Klamt, J. Phys. Chem.-US 99, 2224 (1995). https://doi.org/10.1021/j100007a062
S.-T. Lin, S.I. Sandler, Ind. Eng. Chem. Res. 41, 899 (2002)
I.H. Bell, E. Mickoleit, C.M. Hsieh, S.T. Lin, J. Vrabec, C. Breitkopf, A. Jager, J. Chem. Theory Comput. 16, 2635 (2020). https://doi.org/10.1021/acs.jctc.9b01016
R.P. de Soares, L.F. Baladão, P.B. Staudt, Fluid Phase Equilib. 488, 13 (2019). https://doi.org/10.1016/j.fluid.2019.01.015
S.S. Kang, J. Lee, J.W. Kang, Fluid Phase Equilib. (2020). https://doi.org/10.1016/j.fluid.2020.112673
E. Mullins, R. Oldland, Y. Liu, S. Wang, S.I. Sandler, C.-C. Chen, M. Zwolak, K.C. Seavey, Ind. Eng. Chem. Res. 45, 4389 (2006)
A. Klamt, COSMO-RS: From Quantum Chemistry to Fluid Phase Thermodynamics and Drug Design (Elsevier, Amsterdam, 2005)
T. Mu, J. Rarey, J. Gmehling, AIChE J. 53, 3231 (2007). https://doi.org/10.1002/aic.11338
T. Mu, J. Rarey, J. Gmehling, AIChE J. 55, 3298 (2009). https://doi.org/10.1002/aic.11933
Q.L. Liu, L. Zhang, K. Tang, L.L. Liu, J. Du, Q.W. Meng, R. Gani, Aiche J. (2021). https://doi.org/10.1002/aic.17110
M. Haghighatlari, J. Hachmann, Curr. Opin. Chem. Eng. 23, 51 (2019). https://doi.org/10.1016/j.coche.2019.02.009
J.-J. Chang, J.-L. Kang, D.S.-H. Wong, C.-H. Chou, H.-H. Hsu, C.-H. Huang, S.-T. Lin, in 29th European Symposium on Computer Aided Process Engineering, (2019), pp. 787–792
J. Marrero, R. Gani, Fluid Phase Equilib. 183, 183 (2001). https://doi.org/10.1016/S0378-3812(01)00431-9
S. Kearnes, K. McCloskey, M. Berndl, V. Pande, P. Riley, J. Comput. Aided Mol. Des. 30, 595 (2016). https://doi.org/10.1007/s10822-016-9938-8
S.Y. Hwang, J.W. Kang, Int. J. Thermophys. (2022). https://doi.org/10.1007/s10765-022-03060-7
S. Wang, S.I. Sandler, C.C. Chen, Ind. Eng. Chem. Res. 46, 7275 (2007). https://doi.org/10.1021/ie070465z
C.M. Hsieh, S.I. Sandler, S.T. Lin, Fluid Phase Equilib. 297, 90 (2010). https://doi.org/10.1016/j.fluid.2010.06.011
C.M. Hsieh, S.T. Lin, J. Vrabec, Fluid Phase Equilib. 384, 14 (2014). https://doi.org/10.1016/j.fluid.2014.10.019
R.C. Xiong, S.I. Sandler, R.I. Burnett, Ind. Eng. Chem. Res. 53, 8265 (2014). https://doi.org/10.1021/ie404410v
R. Fingerhut, W.L. Chen, A. Schedemann, W. Cordes, J. Rarey, C.M. Hsieh, J. Vrabec, S.T. Lin, Ind. Eng. Chem. Res. 56, 9868 (2017). https://doi.org/10.1021/acs.iecr.7b01360
F. Chollet et al., Keras. (Keras, 2015) https://keras.io/. Accessed 07 Oct 2022
Y.-H. Song, X.-M. Hou, J. Song, Y. Zhang, J. Wang, P.-H. Wei, C.-F. Li, J. Chem. Eng. Data 63, 138 (2017). https://doi.org/10.1021/acs.jced.7b00740
H. Zheng, X. Luo, G. Yin, J. Chen, S. Zhao, J. Chem. Eng. Data 63, 49 (2017). https://doi.org/10.1021/acs.jced.7b00574
D.-C. Wang, C.-L. Wang, Y.-J. Luo, H. Song, X.-P. Liu, J. Chem. Eng. Data (2018). https://doi.org/10.1021/acs.jced.8b00158
X. Man, T. Jiao, Z. Wang, X. Zhang, X. Zhang, J. Chem. Eng. Data 64, 5038 (2019). https://doi.org/10.1021/acs.jced.8b01247
S. Naicker, S.A. Iwarere, P. Naidoo, D. Ramjugernath, J. Chem. Eng. Data 64, 2388 (2019). https://doi.org/10.1021/acs.jced.8b01196
D. Wei, L. Kong, X. Tan, S. Zheng, J. Chem. Eng. Data 64, 736 (2019). https://doi.org/10.1021/acs.jced.8b00966
J. Wu, L. Wang, X. Chen, X. Wei, J. Liang, Z. Tong, J. Chem. Eng. Data 64, 905 (2019). https://doi.org/10.1021/acs.jced.8b00672
J. Xu, S. Li, Z. Zeng, W. Xue, J. Chem. Eng. Data 65, 81 (2019). https://doi.org/10.1021/acs.jced.9b00757
T. Sommer, M. Zapletal, J. Trejbal, J. Chem. Eng. Data 65, 2291 (2020). https://doi.org/10.1021/acs.jced.9b00746
S.M. Jiao, Y. Gao, J. Feng, T. Lei, X.C. Yuan, Opt. Express 28, 3717 (2020). https://doi.org/10.1364/Oe.382319
M.A. Schulz, B.T.T. Yeo, J.T. Vogelstein, J. Mourao-Miranada, J.N. Kather, K. Kording, B. Richards, D. Bzdok, Nat. Commun. (2020). https://doi.org/10.1038/s41467-020-18037-z
Acknowledgements
This work was supported by National Research Foundation of Korea (NRF) grants funded by the Korean government (MSIT) [Grant Numbers NRF- 2021R1A5A6002853 and NRF-2019M3E6A1064876]. This work was also supported by Korea Environment Industry & Technology Institute (KEITI) through Technology Development Project for Safety Management of Household Chemical Products Program, funded by Korea Ministry of Environment (MOE) (ARQ202201483001).
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
CREdiT authorship contribution statement. BCR: Conceptualization, Methodology, Investigation, Writing—original draft. SSK: Methodology. SYH: Methodology. JWK: Supervision, Writing—review & editing.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ryu, B.C., Hwang, S.Y., Kang, S.S. et al. Group Contribution Based Graph Convolution Network: Predicting Vapor–Liquid Equilibrium with COSMO-SAC-ML. Int J Thermophys 44, 49 (2023). https://doi.org/10.1007/s10765-022-03141-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-022-03141-7