Abstract
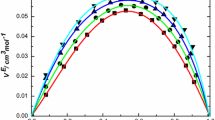
Concentration dependences of the thermal effusivity, isentropic compressibility coefficient, and molar volume were investigated experimentally for aqueous solutions of ethanol, 1-propanol, and 2-propanol. The thermal effusivity was determined using a photoacoustic method. The excess molar volume was found from measured densities, while the isentropic compressibility coefficient was calculated based on density and ultrasound velocity measurements. It has been shown that the dependence of the effusivity on concentration, expressed in mass fraction units, is nonlinear in the case of all the alcohols used. Moreover, the location of extreme deviations from linearity for the thermal effusivity, Δe, agrees well with that of characteristic points for the isentropic compressibility coefficient, κ S , and the excess molar volume, \({V_{\rm m}^{\rm E}}\), as a function of the concentration.
Similar content being viewed by others
References
Neamtu C., Dadarlat D., Chirtoc M., Hadj Sahraoui A., Longuemart S., Bicanic D.: Instrum. Sci. Technol. 34, 225 (2006)
Franks F.: Water: A Matrix of Life, 2nd edn, pp. 53–68. The Royal Society of Chemistry, Cambridge (2000)
Jerie K., Baranowski A., Ernst S., Glinski J.: Acta Phys. Polon. A69, 81 (1986)
Marczak W., Spurek M.: J. Solution Chem. 33, 99 (2004)
Jeffrey G.A., McMullan R.K.: Progr. Inorg. Chem. 8, 43 (1967)
Wakisaka A., Matsuura K.: J. Mol. Liq. 129, 25 (2006)
Almond D.P., Patel P.M.: Photothermal Science and Techniques. Chapman & Hall, London (1996)
Dadarlat D., Neamtu C., Tosa V., Streza M.: Acta Chim. Slov. 54, 149 (2007)
Sikorska A., Dadarlat D., Linde B.B.J., Streza M., Neamtu C.: J. Phys. IV 137, 341 (2006)
Sikorska A., Linde B.B.J., Żwirbla W.: Chem. Phys. 320, 31 (2005)
Sikorska A., Linde B.: Chem. Phys. 354, 148 (2008)
Gonzalez C., Iglesias M., Lanz J., Marino G., Orge B., Resa J.M.: J. Food Eng. 50, 29 (2001)
J.A. Riddick, W.B. Bunger, T.K. Sokano (eds.), Organic Solvents. Physical Properties and Methods of Purification (Techniques of Chemistry) , 4th edn. (Wiley InterScience, New York, 1986)
D.R. Lide (ed.), CRC Handbook of Chemistry and Physics on CD-ROM, 86th edn. (CRC Press, Boca Raton, FL, 2006)
Savaroglu G., Aral E.: Int. J. Thermophys. 26, 1525 (2005)
Savaroglu G., Aral E.: Fluid Phase Equilib. 215, 253 (2004)
Papaloannou D., Zakklus D., Panayiotou C.: J. Chem. Eng. Data 36, 35 (1991)
Kiyohara O., Benson G.C.: J. Solution Chem. 10, 281 (1981)
Del Grosso V.A., Mader C.W.: J. Acoust. Soc. 52, 1442 (1972)
Perondi L.F., Miranda L.C.M.: J. Appl. Phys. 62, 2955 (1987)
Dupre A., Page F.T.M.: Nature 2, 210 (1870)
Assael M.J., Charitidou E., Wakeham W.: Int. J. Thermophys. 10, 793 (1989)
Wang J., Fielbig M.: Int. J. Thermophys. 16, 1353 (1994)
Sato T., Chiba A., Nozaki R.: J. Chem. Phys. 110, 2508 (1999)
Nishi N., Takahashi S., Morimoto M., Tanaka A., Muraya K.: J. Phys. Chem. 99, 462 (1995)
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor Dr. Stefan Ernst on the occasion of his 75th birthday.
Rights and permissions
About this article
Cite this article
Sikorska, A., Ponikwicki, N., Koniecko, A. et al. Comparative Studies of the Mixing Effect on the Thermal Effusivity, Compressibility, and Molar Volume for Aqueous Solutions of Alcohols. Int J Thermophys 31, 131–142 (2010). https://doi.org/10.1007/s10765-009-0639-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10765-009-0639-6