Abstract
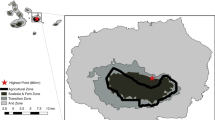
Microhabitat preference among primates, which provides them with the niche they need to survive, often conditions primate diversity, abundance, and coexistence. Vegetation alteration and recovery have built heterogeneous forest landscapes that may influence primates’ microhabitat preferences. We compared the diversity and size of trees/shrubs and the presence of lianas in 132 sites where we captured the rufous mouse lemur (Microcebus rufus), with that of 240 sites where we did not capture this species, to investigate the aspects of microhabitat structure they might prefer. We then examined how this structural preference varies across a heterogeneous landscape of forests with different disturbance levels. Overall, microhabitats used by M. rufus differed significantly from unused ones in densities of small size, understory, and midstory plants. Microcebus rufus frequented microhabitats with significantly denser small- and medium-size (diameter at breast height [DBH] 2.5–10 cm) trees/shrubs without lianas in the primary forest, and small-size plants (DBH 2.5–4.9 cm) with one liana in other forest types. Compared to the microhabitats they used in the primary forest, the microhabitats in other forest types had lower densities of trees/shrubs with lianas. Additionally, the secondary forests and forest fragments also had significantly lower DBH. Although this variation in microhabitat use may represent an opportunity for M. rufus to live in disturbed habitats, it may expose them to additional threats, affecting their long-term survival. These findings emphasize the need to examine potential changes in microhabitat use among primates living in anthropogenic landscapes, which could help optimize long-term conservation and management of threatened primate species in heterogeneous landscapes.
Abstract in Malagasy
Ny fahasamihafana sy habetsahana ary firaisa-monin’ireo gidro ao anaty ala dia mazàna voafehin’ny fananana toeram-ponenana tandrify izay mamaritra ny eram-piveloman’izy ireo. Ny fahasimbana sy fiavaozan’ny tamba-java-maniry dia mahatonga ny ala ho lasa tontolo iray misy karazam-toeram-ponenana maro, izay mety hanova ihany koa ny rafitr’ io toeram-ponenana tandrify io. Mba hamantarana ny rafitry ny toeram-ponenana tandrifin’ny antsidy mena (Microcebus rufus) dia nanao fikarohana mahakasika ny fahasamihafana sy haben’ireo hazo ary ny fisian’ny vahy amin’ny toerana 132 nahafandrihana sy 240 tsy nahafandrihana antsidy mena izahay. Nijery ihany koa ny mety fiovaovan’io rafitra io eo amin’ny tontolo iray misy karazana ala manana sokajim-pahasimbana samihafa. Ny toeram-ponenana tandrifin’ny M. rufus ary dia miavaka amin’ny fananany hazo madinika savaivo ary hazo iva sy salatsalany. Raha dinihina akaiky kokoa dia mampiasa matetika toeram-ponenana be hazo manana savaivo madinika sy salatsalany (savaivo 2.5–10 sm) tsy misy vahy izy ireo any amin’ny ala voajanahary; ary be hazo madini-tsavaivo (savaivo 2.5–4.9 sm) any amin’ireo karazana ala hafa. Raha ampitahaina amin’ny toeram-ponenana any amin’ny ala voajanahary kosa dia nahitana fitotonganan’ny isan’ny hazo misy vahy any amin’ny karazana ala hafa, ary miampy fidinan’ny savaivo izany any amin’ny ala savoka sy vakivakinala. Na dia afaka manararaotra miaina amin’io fiovan-drafitry ny toeram-ponenana tandrify io ary ny M. rufus manoloana ny fahasimbana, dia mety hampiharihary azy ireo amin’ny ambana hafa izany, izay mety hanahirana ny fikajiana azy ireo ho lovain-jafy. Ity voka-pikarohana ity dia mampiseho ny ilàna ny fandalinana ny fiovaovan’nyfapampiasan’ny gidro ny toeram-ponenana tandrifiny anaty tontolo voatsemban’olombelona, izay heverina fa hanatsara ny fikajiana lovain-jafy’ny ala sy fitantanana ireo gidro tandindomin-doza any amin’ny tontolo misy karazam-ponenana maro.








Similar content being viewed by others
References
Anderson, J., Rowcliffe, J. M., & Cowlishaw, G. (2007). Does the matrix matter? A forest primate in a complex agricultural landscape. Biodiversity and Conservation, 135, 121–222.
Andriamandimbiarisoa, L., Blanthorn, T. S., Ernest, R., Ramanamanjato, J.-B., Randriantafika, F., et al (2015). Habitat corridor utilization by the gray mouse lemur, Microcebus murinus, in the littoral forest fragments of southeastern Madagascar. Madagascar Conservation & Development, 10(3), 144–150.
Andriatsitohaina, B., Ramsay, M. S., Kiene, F., Lehman, S. M., Rasoloharijaona, S., et al. (2020). Ecological fragmentation effects in mouse lemurs and small mammals in northwestern Madagascar. American Journal of Primatology, 82(4), e23059. https://doi.org/10.1002/ajp.23059.
Atsalis, S. (1999). Diet of the brown mouse lemur (Microcebus rufus) in Ranomafana National Park, Madagascar. International Journal of Primatology, 20(2), 193–199.
Benchimol, M., & Venticinque, E. M. (2014). Responses of primates to landscape change in Amazonian land-bridge islands: A multi-scale analysis. Biotropica, 46(4), 470–478.
Boyle, S. A., & Smith, A. T. (2010). Can landscape and species characteristics predict primate presence in forest fragments in Brazilian Amazon? Biological Conservation, 143, 1134–1143.
Brown, K. A., & Guveritch, J. (2004). Long-term impacts of logging on forest diversity in Madagascar. Proceedings of the National Academy of Sciences of the USA, 101(16), 6045–6049.
Cabeza, M., Terraube, J., Burgas, D., Temba, E.M. & Rakoarijaoana, M. (2019). Gold is not green: Artisanal gold mining threatens Ranomafana National Park's biodiversity. Animal Conservation, 22(5), pp.417–419.
Cameron, A., & Gould, L. (2013). Fragment-adaptive behavioral strategies and intersite variation in the ring-tailed lemur (Lemur catta) in south-central Madagascar. In L. K. Marsh & C. A. Chapman (Eds.), Primates in fragments: Complexity and resilience (pp. 227–244). Developments in primatology: progress and prospects. Springer Science+Business Media.
Chang, C. C., & Turner, B. L. (2019). Ecological succession in a changing world. The Journal of Ecology, 107(2), 503–509.
Chapman, C. A. (1995). Primate seed dispersal: Coevolution and conservation implications. Evolutionary Anthropology, 4, 74–82.
Chapman, C. A., & Dunham, A. E. (2018). Primate seed dispersal and forest restoration: An African perspective for a brighter future. International Journal of Primatology, 39, 427–442.
Chapman, C. A., Lawes, M. J., & Heley, H. A. C. (2006). What hope for African primate diversity? African Journal of Ecology, 44, 116–133.
Dammhahn, M. (2009). Females go where the food is: Does the socio-ecological model explain variation in social organization of solitary foragers? Behavioral Ecology and Sociobiology, 63, 939–952.
DeSisto, C. M. M., Park, D. S., Davis, C. C., Ramananjato, V., Tonos, J., & Razafindratsima, O. H. (2020). An invasive species spread by threatened diurnal lemurs impacts rainforest structure in Madagascar. Biological Invasions, 22, 2845–2858. https://doi.org/10.1007/s10530-020-02293-7.
Dunham, A. E., Erhart, E. M., & Wright, P. C. (2011). Global climate cycles and cyclones: consequences for rainfall patterns and lemur reproduction in southeastern Madagascar. Global Change Biology, 17, 219–227.
Dunham, A. E., Razafindratsima, O. H., Rakotonirina, P., & Wright, P. C. (2018). Fruiting phenology is linked to rainfall variability in a tropical rainforest. Biotropica, 50, 396–404.
Ehbrecht, M., Seidel, D., Annighöfer, P., Kreft, H., Köhler, M., et al. (2021). Global patterns and climatic controls of forest structural complexity. Nature Communications, 12(519). https://doi.org/10.1038/s41467-020-20767-z.
Enstam, K. L., & Isbell, L. A. (2004). Microhabitat preference and vertical use of space by Patas monkeys (Erythrocebus patas) in relation to predation risk and habitat structure. Folia Primatologica, 75, 70–84.
Estrada, A., Raboy, B. E., & Oliveira, L. C. (2012). Agroecosystems and primate conservation in the tropics: A review. American Journal of Primatology, 74, 696–711.
Ganzhorn, J. U., Malcomber, S., Andrianantoanina, O., & Goodman, S. M. (1997). Habitat characteristics and lemur species richness in Madagascar. Biotropica, 29(3), 331–343.
Gillespie, T. R., & Chapman, C. A. (2006). Prediction of parasite infection dynamics in primate metapopulations based on attributes to forest fragmentation. Conservation Biology, 20(2), 441–448.
Harrell, F. E. (2015). Ordinal logistic regression. In Regression modeling strategies. Springer series in statistics. Springer. https://doi.org/10.1007/978-3-319-19425-7_13.
Herrera, J. P., Wright, P. C., Lauterbur, E., Ratovonjanahary, L., & Taylor, L. L. (2011). The effects of habitat disturbance on lemurs at Ranomafana National Park, Madagascar. International Journal of Primatology, 32, 1091–1108.
Hill, M. O. (1971). Diversity and evenness: A unifying notation and its consequences. Ecology, 54, 427–432.
Irwin, M. T., Wright, P. C., Birkinshaw, C. R., Fisher, B. L., Gardner, C. J., et al (2010). Patterns of species change in anthropogenically disturbed forests of Madagascar. Biological Conservation, 143, 2351–2362.
Johnson, S. E., Ingraldi, C., Ralainasolo, F. B., Andriamaharoa, H. E., Ludovic, R., et al (2011). Gray-headed lemur (Eulemur cinnereiceps) abundance and forest structure dynamics at Manombo, Madagascar. Biotropica, 43(3), 371–379.
Kappeler, P. M., & Rasoloarison, R. M. (2003). Microcebus, mouse lemur, Tsidy. In M. Goodman & J. P. Benstead (Eds.), The natural history of Madagascar (pp. 1310–1315). University of Chicago Press.
Knoop, S., Chikhi, L., & Salmona, J. (2018). Mouse lemurs’ use of degraded habitat: A review of the literature. Lemur News, 21, 20–31.
Louis, E. E., Coles, M. S., Andriantompohavana, R., Sommer, J. A., Engberg, S. E., et al (2006). Revision of the mouse lemurs (Microcebus) of Eastern Madagascar. International Journal of Primatology, 27(2), 347–389.
Mansourian, S., Razafimahatratra, A., & Valauri, D. (2018). Experiences in forest landscape restoration—Lessons learnt from 13 years of restoration in a moist tropical forest: The Fandriana-Marolambo landscape in Madagascar (WWF Field Series.). Antananarivo.
Marsh, L. K., & Chapman, C. A. (Eds.). (2013). Primates in fragments: Complexity and resilience. Developments in primatology: progress and prospects. Springer.
Mbora, D. N., & McPeek, M. A. (2009). Host density and human activities mediate increased paratsite prevalence and richness in primates threatened by habitat loss and fragmentation. Journal of Animal Ecology, 78(1), 210–2018.
Mittermeier, R. A., Louis, E. E., Richardson, M., Schwitzer, C., Langrand, O., & Rylands, A. (2010). Lemurs of Madagascar, 3rd ed. (Tropical Field Guide Series). Conservation International.
Nakagawa, S., & Cuthill, I. C. (2007). Effect size, confidence interval and statistical significance: A practical guide for biologists. Biological Reviews, 82, 591–605.
Nekaris, & Stengel, C. J. (2013). Where are they? Quantification, distribution and microhabitat use of fragments by the Red Slender Loris (Loris tardigradus tardigradus) in Sri Lanka. In L. K. Marsh & C. A. Chapman (Eds.), Primates in fragments: Complexity and resilience (pp. 371–384). Developments in primatology: progress and prospects. Springer.
Nowak, K., & Lee, P. C. (2013). “Specialist” primates can be flexible in response to habitat alteration. In L. K. Marsh & C. A. Chapman (Eds.), Primates in fragments: Complexity and resilience (pp. 199–212). Springer.
Peres, C. A. (1993). Structure and spatial organization of an Amazonian terra firme forest primate community. Journal of Tropical Ecology, 9, 259–276.
Pyritz, L. W., Büntge, A. B. S., Herzog, S. K., & Kessler, M. (2010). Effects of habitat structure and fragmentation on diversity and abundance of Primates in tropical deciduous forests in Bolivia. International Journal of Primatology, 31, 796–8112.
R Core Team. (2018). R: A language an environment for statistical computing. Austria. https://www.R-project.org/.
Rakotondranary, S. J., & Ganzhorn, J. U. (2011). Habitat separation of sympatric Microcebus spp. in dry spiny forest of south-eastern Madagascar. Folia Primatologica, 82, 212–223.
Ramananjato, V., Rakotomalala, Z., Park, D. S., DeSisto, C. M. M., Raoelinjanakolona, N. N., et al (2020). The role of nocturnal omnivorous lemurs as seed dispersers in Malagasy rain forests. Biotropica, 52(4), 758–765. https://doi.org/10.1111/btp.12789.
Randrianambinina, B., Rasoloharijaona, S., Rakotondravony, R., Zimmerman, E., & Radespiel, U. (2010). Abundance and conservation status of two newly described lemur species in northwestern Madagascar (Microcebus danfossi, Lepilemur grewcockorum). Madagascar Conservation & Development, 5(2), 95–102.
Razafimahaimodison, J.-C. (2003). Biodiversity and ecotourism: Impacts of habitat disturbance on an endangered bird species in Madagascar. Biodiversity, 4, 9–16.
Razafindratsima, O. H. (2017). Post-dispersal seed removal by rodents in Ranomafana rain forest, Madagascar. Journal of Tropical Ecology, 33, 232–236.
Razafindratsima, O. H., & Dunham, A. E. (2015). Assessing the impacts of nonrandom seed dispersal by multiple frugivore partners on plant recruitment. Ecology, 96(1), 24–30.
Razafindratsima, O. H., Jones, T. A., & Dunham, A. E. (2014). Patterns of movement and seed dispersal by three lemur species. American Journal of Primatology, 76, 84–96.
Rendigs, A., Radespiel, U., Wrogemann, D., & Zimmerman, E. (2003). Relationships between microhabitat structure and distribution of mouse lemurs (Microcebus spp.) in northwestern Madagascar. International Journal of Primatology, 24(1), 47–64.
Schreier, B. M., Harcourt, A. H., Coppeto, S. A., & Somi, S. F. (2009). Interspecific competition and niche separation in primates: A global analysis. Biotropica, 41, 283–291.
Schwitzer, N., Randriantahina, G. H., Kaumanns, W., Hoffmeister, D., & Schwitzer, C. (2007). Habitat utilization of blue-eyed black lemurs, Eulemur macaco flavifrons (Gray, 1867), in primary and altered forest fragments. Primate Conservation, 22, 79–87.
Seiler, M., Holderied, M., & Schwitzer, C. (2013). Effects of habitat alteration on resting behaviour of the Sahamalaza sportive lemur in North West Madagascar. In L. K. Marsh & C. A. Chapman (Eds.), Primates in fragments: Complexity and resilience (pp. 259–271). Springer.
Shanubhogue, A., & Gore, A. P. (1987). Using logistic regression in ecology. Current Science, 56(18), 933–935.
Steffens, T. S., & Lehman, S. M. (2016). Factors determining Microcebus abundance in a fragmented landscape in Ankarafantsika National Park, Madagascar. In S. M. Lehman, U. Radespiel, & E. Zimmermann (Eds.), The dwarf and mouse lemurs of Madagascar: Biology, behavior and conservation biogeography of Cheirogaleidae (pp. 477–497). Cambridge University Press.
Steffens, T. S., & Lehman, S. M. (2019). Species-area relationships of lemurs in a fragmented landscape in Madagascar. American Journal of Primatology, 81(4), e22972. https://doi.org/10.1002/ajp.22972.
Webber, A. D., Solofondranohatra, J. S., Razafindramoana, S., Fernández, D., Parker, C. A., et al. (2020). Lemurs in cacao: Presence and abundance within the shade plantations of northern Madagascar. Folia Primatologica, 91(2), 97–107. https://doi.org/10.1159/000501987.
Wilmé, L., Waeber, P. O., Moutou, F., Gardner, C. J., Razafindratsima, O. H., et al (2016). A proposal for ethical research conduct in Madagascar. Madagascar Conservation & Development, 11(1), 36–39.
Wright, P. C., Erhart, E. M., Tecot, S., Baden, A. L., Arrigo-Nelson, S. J., et al. (2012). Long-term lemur research at Centre Valbio, Ranomafana National Park. In P. M. Kappeler & D. P. Watts (Eds.), Long-term field studies of Primates (pp. 67–100). Springer-Verlag.
Wright, P. C., Hearthstone, E., Zakamanana, F., Andrianoely, D., & Donohue, M. E. (2020). Microcebus rufus, Rufous mouse lemur. In The IUCN red list of threatened species 2020. IUCN SSC Primate Specialist: Group. https://doi.org/10.2305/IUCN.UK.2020-2.RLTS.T59544947A115588845.en.
Wright, P. C., & Johnson, S. E. (2013). Ranomafana National Park. In C. Schwitzer, R. A. Mittermeier, N. Davies, S. E. Johnson, J. H. Ratsimbazafy, et al. (Eds.), Lemurs of Madagascar: A strategic plan for their conservation 2013–2016 (pp. 89–91). IUCN SSC Primate Specialist Group, Bristol Conservation and Science Foundation, and Conservation International.
Acknowledgments
We thank the Rufford Small Grant for Nature Conservation (Grant No. 24419-1) and Idea Wild for generously funding the research that led to these observations. We thank Finaritra Randimbiarison and Dr. Zafimahery Rakotomalala for their insightful comments that improved an earlier version of this manuscript. We also thank Ary Saina Writing Group for their advice, comments, and suggestions when preparing and writing this article and Centre Valbio and Mention Zoologie and Biodiversité Animale at the University of Antananarivo for their administrative and logistical support during fieldwork. We are grateful to Nérée Raharo Beson and Aimé “Meme” Andriantiana and all local guides for their valuable help during data collection. We also thank Dr. Andrea Baden, two anonymous reviewers, and the editor-in-chief Dr. Joanna Setchell for their helpful feedback in the development of this article.
Author information
Authors and Affiliations
Contributions
VR and OHR conceived and designed the project. VR conducted fieldwork. VR analyzed the data with inputs from OHR. VR wrote the first draft of the manuscript. Both authors contributed equally to later writing of the manuscript.
Corresponding author
Additional information
Handling Editor: Joanna Setchell.

Badge earned for open practices: Open Data. Experiment materials and data are available at: https://doi.org/10.5061/dryad.2280gb5rs.
Supplementary Information
ESM 1
(DOCX 404 kb)
Rights and permissions
About this article
Cite this article
Ramananjato, V., Razafindratsima, O.H. Structure of Microhabitats Used by Microcebus rufus Across a Heterogeneous Landscape. Int J Primatol 42, 682–700 (2021). https://doi.org/10.1007/s10764-021-00224-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10764-021-00224-4