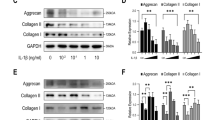
Abstract
Intervertebral disc degeneration (IDD) diseases are common and frequent diseases in orthopedics. The caspase recruitment domain (CARD) and membrane-associated guanylate kinase-like protein 3 (CARMA3) is crucial in the activation of the NF-κB pathway. However, the biological function of CARMA3 in IDD remains unknown. Here, CARMA3 expression was elevated in nucleus pulposus (NP) tissues of IDD rats and nutrient deprivation (ND)-induced NP cells. The main pathological manifestations observed in IDD rats were shrinkage of the NP, reduction of NP cells, fibrosis of NP tissues, and massive reduction of proteoglycans. These changes were accompanied by a decrease in the expression of collagen II and aggrecan, an increase in the expression of the extracellular matrix (ECM) catabolic proteases MMP-3, MMP-13, and metalloprotease with ADAMTS-5, and an increase in the activity of the pro-apoptotic protease caspase-3. The expression of p-IκBαSer32/36 and p-p65Ser536 was also upregulated. However, these effects were reversed with the knockdown of CARMA3. Mechanistically, CARMA3 bound to BCL10 and MALT1 to form a signalosome. Knockdown of CARMA3 reduced the CARMA3-BCL10-MALT1 signalosome-mediated NF-κB activation. CARMA3 activated the NF-κB signaling pathway in a manner that bound to BCL10 and MALT1 to form a signalosome, which affects NP cell damage and is involved in the development of IDD. This supports CARMA3-BCL10-MALT1-NF-κB as a promising targeting axis for the treatment of IDD.







Similar content being viewed by others
Data Availability
Data in this study are available from the corresponding author upon reasonable request.
References
Teraguchi, M., N. Yoshimura, H. Hashizume, S. Muraki, H. Yamada, A. Minamide, H. Oka, Y. Ishimoto, K. Nagata, R. Kagotani, N. Takiguchi, T. Akune, H. Kawaguchi, K. Nakamura, and M. Yoshida. 2014. Prevalence and distribution of intervertebral disc degeneration over the entire spine in a population-based cohort: the Wakayama Spine Study. Osteoarthritis and Cartilage 22 (1): 104–110. https://doi.org/10.1016/j.joca.2013.10.019.
Zhang, Y., F. He, Z. Chen, Q. Su, M. Yan, Q. Zhang, J. Tan, L. Qian, and Y. Han. 2019. Melatonin modulates IL-1β-induced extracellular matrix remodeling in human nucleus pulposus cells and attenuates rat intervertebral disc degeneration and inflammation. Aging 11 (22): 10499–10512. https://doi.org/10.18632/aging.102472.
Chan, D., Y. Song, P. Sham, and K.M.C. Cheung. 2006. Genetics of disc degeneration. European Spine Journal : Official Publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society 15 (Suppl 3): S317–S325.
Kamali, A., R. Ziadlou, G. Lang, J. Pfannkuche, S. Cui, Z. Li, R.G. Richards, M. Alini, and S. Grad. 2021. Small molecule-based treatment approaches for intervertebral disc degeneration: Current options and future directions. Theranostics 11 (1): 27–47. https://doi.org/10.7150/thno.48987.
Raj, P.P. 2008. Intervertebral disc: anatomy-physiology-pathophysiology-treatment. Pain Practice: the Official Journal of World Institute of Pain 8 (1): 18–44. https://doi.org/10.1111/j.1533-2500.2007.00171.x
Vergroesen, P.P.A., I. Kingma, K.S. Emanuel, R.J.W. Hoogendoorn, T.J. Welting, B.J. van Royen, J.H. van Dieën, and T.H. Smit. 2015. Mechanics and biology in intervertebral disc degeneration: A vicious circle. Osteoarthritis and Cartilage 23 (7): 1057–1070. https://doi.org/10.1016/j.joca.2015.03.028.
Shaw, A.S., and E.L. Filbert. 2009. Scaffold proteins and immune-cell signalling. Nature Reviews. Immunology 9 (1): 47–56. https://doi.org/10.1038/nri2473.
Blonska, M., and X. Lin. 2011. NF-κB signaling pathways regulated by CARMA family of scaffold proteins. Cell Research 21 (1): 55–70. https://doi.org/10.1038/cr.2010.182.
McAuley, J.R., T.J. Freeman, P. Ekambaram, P.C. Lucas, and L.M. McAllister-Lucas. 2018. CARMA3 is a critical mediator of G protein-coupled receptor and receptor tyrosine kinase-driven solid tumor pathogenesis. Frontiers In Immunology 9: 1887. https://doi.org/10.3389/fimmu.2018.01887.
Wang, H., Z. Li, Y. Huo, T. Tian, D. Yang, L. Ma, S. Yang, and W. Ding. 2021. 17β-Estradiol alleviates intervertebral disc degeneration by inhibiting NF-κB signal pathway. Life Sciences 284.
Li, Z., X. Wang, H. Pan, H. Yang, X. Li, K. Zhang, H. Wang, Z. Zheng, H. Liu, and J. Wang. 2017. Resistin promotes CCL4 expression through toll-like receptor-4 and activation of the p38-MAPK and NF-κB signaling pathways: implications for intervertebral disc degeneration. Osteoarthritis and Cartilage 25 (2): 341–350. https://doi.org/10.1016/j.joca.2016.10.002.
Grabiner, B.C., M. Blonska, P.-C. Lin, Y. You, D. Wang, J. Sun, B.G. Darnay, C. Dong, and X. Lin. 2007. CARMA3 deficiency abrogates G protein-coupled receptor-induced NF-{kappa}B activation. Genes & Development 21 (8): 984–996.
Cowan, C., C.K. Muraleedharan, J.J. O’Donnell, P.K. Singh, H. Lum, A. Kumar, and S. Xu. 2014. MicroRNA-146 inhibits thrombin-induced NF-κB activation and subsequent inflammatory responses in human retinal endothelial cells. Investigative Ophthalmology & Visual Science 55 (8): 4944–4951. https://doi.org/10.1167/iovs.13-13631.
McAllister-Lucas, L.M., X. Jin, S. Gu, K. Siu, S. McDonnell, J. Ruland, P.C. Delekta, M. Van Beek, and P.C. Lucas. 2010. The CARMA3-Bcl10-MALT1 signalosome promotes angiotensin II-dependent vascular inflammation and atherogenesis. The Journal of Biological Chemistry 285 (34): 25880–25884. https://doi.org/10.1074/jbc.C110.109421.
Chen, S., L. Lei, Z. Li, F. Chen, Y. Huang, G. Jiang, X. Guo, Z. Zhao, H. Liu, H. Wang, C. Liu, Z. Zheng, and J. Wang. 2022. Grem1 accelerates nucleus pulposus cell apoptosis and intervertebral disc degeneration by inhibiting TGF-β-mediated Smad2/3 phosphorylation. Experimental & Molecular Medicine 54 (4): 518–530. https://doi.org/10.1038/s12276-022-00753-9.
Luo, R., G. Li, W. Zhang, H. Liang, S. Lu, J.P.Y. Cheung, T. Zhang, J. Tu, H. Liu, Z. Liao, W. Ke, B. Wang, Y. Song, and C. Yang. 2022. O-GlcNAc transferase regulates intervertebral disc degeneration by targeting FAM134B-mediated ER-phagy. Experimental & Molecular Medicine 54 (9): 1472–1485. https://doi.org/10.1038/s12276-022-00844-7.
Li, Z., H. Yang, Y. Hai, and Y. Cheng. 2023. Regulatory effect of inflammatory mediators in intervertebral disc degeneration. Mediators of Inflammation 2023: 6210885. https://doi.org/10.1155/2023/6210885.
Zhang, G.-Z., M.-Q. Liu, H.-W. Chen, Z.-L. Wu, Y.-C. Gao, Z.-J. Ma, X.-G. He, and X.-W. Kang. 2021. NF-κB signalling pathways in nucleus pulposus cell function and intervertebral disc degeneration. Cell Proliferation 54 (7): e13057. https://doi.org/10.1111/cpr.13057.
Israël, L., M. Bardet, A. Huppertz, N. Mercado, S. Ginster, A. Unterreiner, A. Schlierf, J.F. Goetschy, H.-G. Zerwes, L. Roth, F. Kolbinger, and F. Bornancin. 2018. A CARMA3-dependent tonic signalosome activates MALT1 paracaspase and regulates IL-17/TNF-α-driven keratinocyte inflammation. The Journal of Investigative Dermatology 138 (9): 2075–2079. https://doi.org/10.1016/j.jid.2018.03.1503.
Ni, W., C. Jiang, Y. Wu, H. Zhang, L. Wang, J.H.N. Yik, D.R. Haudenschild, S. Fan, S. Shen, and Z. Hu. 2021. CircSLC7A2 protects against osteoarthritis through inhibition of the miR-4498/TIMP3 axis. Cell Proliferation 54 (6): e13047. https://doi.org/10.1016/j.jid.2018.03.1503.
Parreira, P., C.G. Maher, D. Steffens, M.J. Hancock, and M.L. Ferreira. 2018. Risk factors for low back pain and sciatica: An umbrella review. The Spine Journal : Official Journal of the North American Spine Society 18 (9): 1715–1721. https://doi.org/10.1016/j.spinee.2018.05.018.
Roughley, P.J., L.I. Melching, T.F. Heathfield, R.H. Pearce, and J.S. Mort. 2006. The structure and degradation of aggrecan in human intervertebral disc. European Spine Journal : Official Publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society 15 (Suppl 3): S326–S332.
Liang, H., R. Luo, G. Li, W. Zhang, Y. Song, and C. Yang. 2022. The proteolysis of ECM in intervertebral disc degeneration. International Journal of Molecular Sciences. https://doi.org/10.3390/ijms23031715.
Wang, W.-J., X.-H. Yu, C. Wang, W. Yang, W.-S. He, S.-J. Zhang, Y.-G. Yan, and J. Zhang. 2015. MMPs and ADAMTSs in intervertebral disc degeneration. Clinica Chimica Acta: International Journal of Clinical Chemistry 448: 238–246. https://doi.org/10.1016/j.cca.2015.06.023.
Vo, N.V., R.A. Hartman, T. Yurube, L.J. Jacobs, G.A. Sowa, and J.D. Kang. 2013. Expression and regulation of metalloproteinases and their inhibitors in intervertebral disc aging and degeneration. The Spine Journal : Official Journal of the North American Spine Society 13 (3): 331–341. https://doi.org/10.1016/j.spinee.2012.02.027.
Miyazaki, S., K. Kakutani, T. Yurube, K. Maeno, T. Takada, Z. Zhang, T. Kurakawa, Y. Terashima, M. Ito, T. Ueha, T. Matsushita, R. Kuroda, M. Kurosaka, and K. Nishida. 2015. Recombinant human SIRT1 protects against nutrient deprivation-induced mitochondrial apoptosis through autophagy induction in human intervertebral disc nucleus pulposus cells. Arthritis Research & Therapy 17 (1): 253. https://doi.org/10.1186/s13075-015-0763-6.
Ao, P., W. Huang, J. Li, T. Wu, L. Xu, Z. Deng, W. Chen, C. Yin, and X. Cheng. 2018. 17β-estradiol protects nucleus pulposus cells from serum deprivation-induced apoptosis and regulates expression of MMP-3 and MMP-13 through promotion of autophagy. Biochemical and Biophysical Research Communications 503 (2): 791–797. https://doi.org/10.1016/j.bbrc.2018.06.077.
Wang, J., H. Pan, X. Li, K. Zhang, Z. Li, H. Wang, Z. Zheng, and H. Liu. 2017. Hypoxia suppresses serum deprivation-induced degradation of the nucleus pulposus cell extracellular matrix through the JNK and NF-κB pathways. Journal of Orthopaedic Research : Official Publication of the Orthopaedic Research Society 35 (9): 2059–2066. https://doi.org/10.1002/jor.23486.
Johnson, W.E.B., S. Stephan, and S. Roberts. 2008. The influence of serum, glucose and oxygen on intervertebral disc cell growth in vitro: Implications for degenerative disc disease. Arthritis Research & Therapy 10 (2): R46. https://doi.org/10.1186/ar2405.
Yang, S.-D., Z.-L. Bai, F. Zhang, L. Ma, D.-L. Yang, and W.-Y. Ding. 2014. Levofloxacin increases the effect of serum deprivation on anoikis of rat nucleus pulposus cells via Bax/Bcl-2/caspase-3 pathway. Toxicology Mechanisms and Methods 24 (9): 688–696. https://doi.org/10.3109/15376516.2014.963772.
Fujita, N., J.-I. Imai, T. Suzuki, M. Yamada, K. Ninomiya, K. Miyamoto, R. Iwasaki, H. Morioka, M. Matsumoto, K. Chiba, S. Watanabe, T. Suda, Y. Toyama, and T. Miyamoto. 2008. Vascular endothelial growth factor-A is a survival factor for nucleus pulposus cells in the intervertebral disc. Biochemical and Biophysical Research Communications 372 (2): 367–372. https://doi.org/10.1016/j.bbrc.2008.05.044.
Zelzer, E., R. Mamluk, N. Ferrara, R.S. Johnson, E. Schipani, and B.R. Olsen. 2004. VEGFA is necessary for chondrocyte survival during bone development. Development (Cambridge, England) 131 (9): 2161–2171.
Choi, H., S. Tessier, E.S. Silagi, R. Kyada, F. Yousefi, N. Pleshko, I.M. Shapiro, and M.V. Risbud. 2018. A novel mouse model of intervertebral disc degeneration shows altered cell fate and matrix homeostasis. Matrix Biology : Journal of the International Society For Matrix Biology 70: 102–122. https://doi.org/10.1016/j.matbio.2018.03.019.
Nicolas, S., S. Abdellatef, M.A. Haddad, I. Fakhoury, and M. El-Sibai. 2019. Hypoxia and EGF stimulation regulate VEGF expression in human glioblastoma multiforme (GBM) cells by differential regulation of the PI3K/Rho-GTPase and MAPK pathways. Cells. https://doi.org/10.3390/cells8111397.
Shim, E.-K., J.-S. Lee, D.-E. Kim, S.K. Kim, B.-J. Jung, E.-Y. Choi, and C.-S. Kim. 2016. Autogenous mesenchymal stem cells from the vertebral body enhance intervertebral disc regeneration via paracrine interaction: An in vitro pilot study. Cell Transplantation 25 (10): 1819–1832. https://doi.org/10.3727/096368916X691420.
Wu, J., Y. Chen, Z. Liao, H. Liu, S. Zhang, D. Zhong, X. Qiu, T. Chen, D. Su, X. Ke, Y. Wan, T. Zhou, and P. Su. 2022. Self-amplifying loop of NF-κB and periostin initiated by PIEZO1 accelerates mechano-induced senescence of nucleus pulposus cells and intervertebral disc degeneration. Molecular Therapy : the Journal of the American Society of Gene Therapy 30 (10): 3241–3256. https://doi.org/10.1016/j.ymthe.2022.05.021.
Shao, Z., B. Wang, Y. Shi, C. Xie, C. Huang, B. Chen, H. Zhang, G. Zeng, H. Liang, Y. Wu, Y. Zhou, N. Tian, A. Wu, W. Gao, X. Wang, and X. Zhang. 2021. Senolytic agent quercetin ameliorates intervertebral disc degeneration via the Nrf2/NF-κB axis. Osteoarthritis and Cartilage 29 (3): 413–422. https://doi.org/10.1016/j.joca.2020.11.006.
Yin, H., Y. Zhang, K. Wang, Y. Song, J. Tu, L. Kang, K. Zhao, X. Wu, R. Luo, and C. Yang. 2018. The involvement of regulated in development and DNA damage response 1 (REDD1) in the pathogenesis of intervertebral disc degeneration. Experimental Cell Research 372 (2): 188–197. https://doi.org/10.1016/j.yexcr.2018.10.001.
Wang, J., J. Hu, X. Chen, C. Huang, J. Lin, Z. Shao, M. Gu, Y. Wu, N. Tian, W. Gao, Y. Zhou, X. Wang, and X. Zhang. 2019. BRD4 inhibition regulates MAPK, NF-κB signals, and autophagy to suppress MMP-13 expression in diabetic intervertebral disc degeneration. FASEB Journal : Official Publication of the Federation of American Societies For Experimental Biology 33 (10): 11555–11566. https://doi.org/10.1096/fj.201900703R.
Liao, Z., X. Wu, Y. Song, R. Luo, H. Yin, S. Zhan, S. Li, K. Wang, Y. Zhang, and C. Yang. 2019. Angiopoietin-like protein 8 expression and association with extracellular matrix metabolism and inflammation during intervertebral disc degeneration. Journal of Cellular and Molecular Medicine 23 (8): 5737–5750. https://doi.org/10.1111/jcmm.14488.
Zhongyi, S., Z. Sai, L. Chao, and T. Jiwei. 2015. Effects of nuclear factor kappa B signaling pathway in human intervertebral disc degeneration. Spine 40 (4): 224–232. https://doi.org/10.1097/BRS.0000000000000733.
Wang, L., Y. Guo, W.J. Huang, X. Ke, J.L. Poyet, G.A. Manji, S. Merriam, M.A. Glucksmann, P.S. DiStefano, E.S. Alnemri, and J. Bertin. 2001. Card10 is a novel caspase recruitment domain/membrane-associated guanylate kinase family member that interacts with BCL10 and activates NF-kappa B. The Journal of Biological Chemistry 276 (24): 21405–21409.
Zhang, S., D. Pan, X.-M. Jia, X. Lin, and X. Zhao. 2017. The CARMA3-BCL10-MALT1 (CBM) complex contributes to DNA damage-induced NF-κB activation and cell survival. Protein & Cell 8 (11): 856–860. https://doi.org/10.1007/s13238-017-0441-3.
McAllister-Lucas, L.M., J. Ruland, K. Siu, X. Jin, S. Gu, D.S.L. Kim, P. Kuffa, D. Kohrt, T.W. Mak, G. Nuñez, and P.C. Lucas. 2007. CARMA3/Bcl10/MALT1-dependent NF-kappaB activation mediates angiotensin II-responsive inflammatory signaling in nonimmune cells. Proceedings of the National Academy of Sciences of the United States of America 104 (1): 139–144.
Pan, D., Y. Zhu, Z. Zhou, T. Wang, H. You, C. Jiang, and X. Lin. 2016. The CBM complex underwrites NF-κB activation to promote HER2-associated tumor malignancy. Molecular Cancer Research : MCR. https://doi.org/10.1158/1541-7786.MCR-15-0229-T.
Man, X., T. Liu, Y. Jiang, Z. Zhang, Y. Zhu, Z. Li, C. Kong, and J. He. 2019. Silencing of CARMA3 inhibits bladder cancer cell migration and invasion via deactivating β-catenin signaling pathway. OncoTargets and Therapy 12: 6309–6322. https://doi.org/10.2147/OTT.S191502.
Wu, Y., Y. Qi, Z. Qiu, and W. Chen. 2021. Deficiency of CARMA3 attenuates the development of bleomycin induced pulmonary fibrosis. Biochemical and Biophysical Research Communications 581: 81–88. https://doi.org/10.1016/j.bbrc.2021.10.013.
Hou, Y., X. Xiao, W. Yu, and S. Qi. 2021. Propofol suppresses microglia inflammation by targeting TGM2/NF-κB signaling. Journal of Immunology Research 2021: 4754454. https://doi.org/10.1155/2021/4754454.
Zhang, J., Y. Zheng, Y. Luo, Y. Du, X. Zhang, and J. Fu. 2019. Curcumin inhibits LPS-induced neuroinflammation by promoting microglial M2 polarization via TREM2/ TLR4/ NF-κB pathways in BV2 cells. Molecular Immunology 116: 29–37. https://doi.org/10.1016/j.molimm.2019.09.020.
Feng, X., S. Zhu, J. Qiao, Z. Ji, B. Zhou, and W. Xu. 2023. CX3CL1 promotes M1 macrophage polarization and osteoclast differentiation through NF-κB signaling pathway in ankylosing spondylitis in vitro. Journal of Translational Medicine 21 (1): 573. https://doi.org/10.1186/s12967-023-04449-0.
Sarkar, A., M. Duncan, J. Hart, E. Hertlein, D.C. Guttridge, and M.D. Wewers. 2006. ASC directs NF-kappaB activation by regulating receptor interacting protein-2 (RIP2) caspase-1 interactions. Journal of Immunology (Baltimore, Md. : 1950) 176 (8): 4979–4986.
Author information
Authors and Affiliations
Contributions
YDL and DFY were responsible for the conceptualization and design of this study. YDL, GQZ, JNW, YM, JYH, and HF conducted the experiments and collected the data. YDL, GQZ, and JNW analyzed and interpreted the data. YDL drafted the manuscript. DFY supervised and managed the study. All authors approved the final Manuscript.
Corresponding author
Ethics declarations
Ethics Approval
All experimental procedures were approved by the Ethics Committee of the Dalian Central Hospital (approval number: YN2023-125–01) and were in accordance with the National Institutes of Health guidelines for the care and use of animals.
Consent for Publication
Not applicable.
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Zhang, G., Wu, J. et al. CARMA3 Drives NF-κB Activation and Promotes Intervertebral Disc Degeneration: Involvement of CARMA3-BCL10-MALT1 Signalosome. Inflammation (2024). https://doi.org/10.1007/s10753-024-02016-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10753-024-02016-3