Abstract—
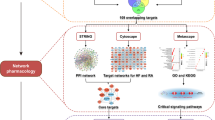
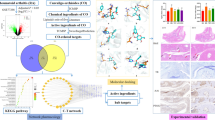
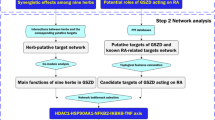
Kaempferol is a common flavonoid aglycone widely found in plants. It exhibits beneficial therapeutic effects in the treatment of arthritis. However, the effects of kaempferol on gouty arthritis (GA) have not been verified. This study aimed to explore the potential mechanisms by which kaempferol regulates GA by network pharmacology and experimental validation. Potential drug targets for GA were identified with a protein–protein interaction network. Then, we performed a KEGG pathway analysis to elucidate the major pathway involved in the kaempferol-mediated treatment of GA. In addition, the molecular docking was performed. A rat model of GA was constructed to verify the results of network pharmacology analysis and investigate the mechanism of kaempferol against GA. The network pharmacology study indicated that there were 275 common targets of kaempferol and GA treatment. Kaempferol exerted therapeutic effects on GA, in part, by regulating the IL-17, AGE-RAGE, p53, TNF, and FoxO signaling pathways. Molecular docking results showed that kaempferol stably docked with the core MMP9, ALB, CASP3, TNF, VEGFA, CCL2, CXCL8, AKT1, JUN, and INS. Experimental validation suggested that kaempferol eased MSU-induced mechanical allodynia, ankle edema, and inflammation. It significantly suppressed the expression of IL-1β, IL-6, TNF-α, and TGF-β1 and restored Th17/Treg imbalance in MSU-induced rats and IL-6-induced PBMCs. Kaempferol also affected RORγt and Foxp3 through IL-17 pathway. The present study clarifies the mechanism of kaempferol against GA and provides evidence to support its clinical use.









Similar content being viewed by others
Data Availability
The data used to support the findings of this study will be available from the corresponding author upon request.
Abbreviations
- BC:
-
Betweenness centrality
- BCA:
-
Bicinchoninic acid
- BP:
-
Biological process
- CC:
-
Closeness centrality
- DC:
-
Degree centrality
- DMSO:
-
Dimethyl sulfoxide
- EDTA:
-
Ethylenediaminetetraacetic acid
- Foxp3:
-
Forkhead box p3
- GA:
-
Gouty arthritis
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- GO:
-
Gene ontology
- H&E:
-
Hematoxylin and eosin
- IL:
-
Interleukin
- KEGG:
-
Kyoto encyclopedia of genes and genomes
- MCC:
-
Maximal clique centrality
- MF:
-
Molecular function
- MSU:
-
Monosodium urate
- NF-κB:
-
Nuclear factor-κ-gene binding
- PBMC:
-
Peripheral blood mononuclear cell
- PBS:
-
Phosphate-buffered saline
- PPI:
-
Protein–protein interaction
- RORγt:
-
Retinoid-related orphan nuclear receptor γt
- SD:
-
Sprague Dawley
- TCMSP:
-
Traditional chinese medicine database and analysis platform;
- TGF-β:
-
Transforming growth facter-β1
- TLR:
-
Toll-like receptors
- TNF:
-
Tumor necrosis factor
- TTD:
-
Therapeutic target database
References
Dalbeth, N., H.K. Choi, L.A.B. Joosten, P.P. Khanna, H. Matsuo, F. Perez-Ruiz, and L.K. Stamp. 2019. Gout. Nature reviews. Disease primers 5 (1): 69.
Xiao, N., J. Qu, S. He, P. Huang, Y. Qiao, G. Li, T. Pan, H. Sui, and L. Zhang. 2019. Exploring the therapeutic composition and mechanism of Jiang-Suan-Chu-Bi recipe on gouty arthritis using an integrated approach based on chemical profile, network pharmacology and experimental support using molecular cell biology. Frontiers in pharmacology 10: 1626.
Du, X., L. Zhao, Y. Yang, Z. Zhang, J. Hu, H. Ren, Z. Chen, and Y. Li. 2020. Investigation of the mechanism of action of Porana sinensis Hemsl. against gout arthritis using network pharmacology and experimental validation. Journal of ethnopharmacology 252: 112606.
Rajendran, P., T. Rengarajan, N. Nandakumar, R. Palaniswami, Y. Nishigaki, and I. Nishigaki. 2014. Kaempferol, a potential cytostatic and cure for inflammatory disorders. European journal of medicinal chemistry 86: 103–112.
Imran, M., B. Salehi, J. Sharifi-Rad, T. Aslam Gondal, F. Saeed, A. Imran, M. Shahbaz, P. V. Tsouh Fokou, M. Umair Arshad, H. Khan, S. G. Guerreiro, N. Martins, and L. M. Estevinho. 2019. Kaempferol: A Key Emphasis to Its Anticancer Potential. Molecules (Basel, Switzerland) 24 (12).
Devi, K.P., D.S. Malar, S.F. Nabavi, A. Sureda, J. Xiao, S.M. Nabavi, and M. Daglia. 2015. Kaempferol and inflammation: From chemistry to medicine. Pharmacological research 99: 1–10.
Ren, J., Y. Lu, Y. Qian, B. Chen, T. Wu, and G. Ji. 2019. Recent progress regarding kaempferol for the treatment of various diseases. Experimental and therapeutic medicine 18 (4): 2759–2776.
Pan, D., N. Li, Y. Liu, Q. Xu, Q. Liu, Y. You, Z. Wei, Y. Jiang, M. Liu, T. Guo, X. Cai, X. Liu, Q. Wang, M. Liu, X. Lei, M. Zhang, X. Zhao, and C. Lin. 2018. Kaempferol inhibits the migration and invasion of rheumatoid arthritis fibroblast-like synoviocytes by blocking activation of the MAPK pathway. International immunopharmacology 55: 174–182.
Wang, S., J. Gao, Q. Li, W. Ming, Y. Fu, L. Song, and J. Qin. 2020. Study on the regulatory mechanism and experimental verification of icariin for the treatment of ovarian cancer based on network pharmacology. Journal of ethnopharmacology 262: 113189.
Chen, Y., Y. Jia, Y. Li, Y. Zheng, G. Chen, and Y. Shi. 2022. Investigation on the antitumor effects of paeonol against renal cell carcinoma based on network pharmacology and experimental validation. Journal of ethnopharmacology 285: 114857.
Trevisan, G., C. Hoffmeister, M.F. Rossato, S.M. Oliveira, M.A. Silva, R.P. Ineu, G.P. Guerra, S. Materazzi, C. Fusi, R. Nassini, P. Geppetti, and J. Ferreira. 2013. Transient receptor potential ankyrin 1 receptor stimulation by hydrogen peroxide is critical to trigger pain during monosodium urate-induced inflammation in rodents. Arthritis and rheumatism 65 (11): 2984–2995.
Yin, C., B. Liu, P. Wang, X. Li, Y. Li, X. Zheng, Y. Tai, C. Wang, and B. Liu. 2020. Eucalyptol alleviates inflammation and pain responses in a mouse model of gout arthritis. British journal of pharmacology 177 (9): 2042–2057.
Samarpita, S., and M. Rasool. 2021. Cyanidin restores Th17/Treg balance and inhibits T follicular helper cell differentiation via modulation of ROCK2 signaling in an experimental model of rheumatoid arthritis. International immunopharmacology 101 (Pt B): 108359.
Tu, L.Y., H.H. Bai, J.Y. Cai, and S.P. Deng. 2016. The mechanism of kaempferol induced apoptosis and inhibited proliferation in human cervical cancer SiHa cell: From macro to nano. Scanning 38 (6): 644–653.
Imran, M., A. Rauf, Z.A. Shah, F. Saeed, A. Imran, M.U. Arshad, B. Ahmad, S. Bawazeer, M. Atif, D.G. Peters, and M.S. Mubarak. 2019. Chemo-preventive and therapeutic effect of the dietary flavonoid kaempferol: A comprehensive review. Phytotherapy research : PTR 33 (2): 263–275.
Wang, J., J. Mao, R. Wang, S. Li, B. Wu, and Y. Yuan. 2020. Kaempferol protects against cerebral ischemia reperfusion injury through intervening oxidative and inflammatory stress induced apoptosis. Frontiers in pharmacology 11: 424.
Shrestha, R., K. Mohankumar, G. Martin, A. Hailemariam, S. O. Lee, U. H. Jin, R. Burghardt, and S. Safe. 2021. Flavonoids kaempferol and quercetin are nuclear receptor 4A1 (NR4A1, Nur77) ligands and inhibit rhabdomyosarcoma cell and tumor growth. Journal of experimental & clinical cancer research CR 40 (1): 392.
Jia, Z., A. Chen, C. Wang, M. He, J. Xu, H. Fu, X. Zhang, W. Lv, and Z. Guo. 2019. Amelioration effects of Kaempferol on immune response following chronic intermittent cold-stress. Research in veterinary science 125: 390–396.
Sharma, A.R., and J.S. Nam. 2019. Kaempferol stimulates WNT/β-catenin signaling pathway to induce differentiation of osteoblasts. The Journal of nutritional biochemistry 74: 108228.
Hopkins, A.L. 2008. Network pharmacology: The next paradigm in drug discovery. Nature chemical biology 4 (11): 682–690.
Bai, L., X. Li, L. He, Y. Zheng, H. Lu, J. Li, L. Zhong, R. Tong, Z. Jiang, J. Shi, and J. Li. 2019. Antidiabetic potential of flavonoids from traditional chinese medicine: A review. The American journal of Chinese medicine 47 (5): 933–957.
Sok Yen, F., C. Shu Qin, S. Tan Shi Xuan, P. Jia Ying, H. Yi Le, T. Darmarajan, B. Gunasekaran, and S. Salvamani. 2021. Hypoglycemic Effects of Plant Flavonoids: A Review. Evidence-based complementary and alternative medicine : eCAM 2021, 2057333.
Wong, S.K., K.Y. Chin, and S. Ima-Nirwana. 2019. The osteoprotective effects of kaempferol: the evidence from in vivo and in vitro studies. Drug design, development and therapy 13: 3497–3514.
Singh, J.A., and A. Gaffo. 2020. Gout epidemiology and comorbidities. Seminars in arthritis and rheumatism 50 (3s): S11-s16.
Dehlin, M., L. Jacobsson, and E. Roddy. 2020. Global epidemiology of gout: Prevalence, incidence, treatment patterns and risk factors. Nature reviews. Rheumatology 16 (7): 380–390.
Zhao, P., K. L. Chen, G. L. Zhang, G. R. Deng, and J. Li. 2017. Pharmacological Basis for Use of Selaginella moellendorffii in Gouty Arthritis: Antihyperuricemic, Anti-Inflammatory, and Xanthine Oxidase Inhibition. Evidence-based complementary and alternative medicine : eCAM 2017, 2103254.
McGeachy, M.J., D.J. Cua, and S.L. Gaffen. 2019. The IL-17 Family of cytokines in health and disease. Immunity 50 (4): 892–906.
Li, X., R. Bechara, J. Zhao, M.J. McGeachy, and S.L. Gaffen. 2019. IL-17 receptor-based signaling and implications for disease. Nature immunology 20 (12): 1594–1602.
Mansour, A.A., F. Raucci, A. Saviano, S. Tull, F. Maione, and A.J. Iqbal. 2021. Galectin-9 regulates monosodium urate crystal-induced gouty inflammation through the modulation of Treg/Th17 ratio. Frontiers in immunology 12: 762016.
Raucci, F., A.J. Iqbal, A. Saviano, P. Minosi, M. Piccolo, C. Irace, F. Caso, R. Scarpa, S. Pieretti, N. Mascolo, and F. Maione. 2019. IL-17A neutralizing antibody regulates monosodium urate crystal-induced gouty inflammation. Pharmacological research 147: 104351.
Gu, C., L. Wu, and X. Li. 2013. IL-17 family: Cytokines, receptors and signaling. Cytokine 64 (2): 477–485.
Yang, Q.B., Y.L. He, Q.B. Zhang, Q.S. Mi, and J.G. Zhou. 2019. Downregulation of transcription factor T-bet as a protective strategy in monosodium urate-induced gouty inflammation. Frontiers in immunology 10: 1199.
Ruiz de Morales, J. M. G., L. Puig, E. Daudén, J. D. Cañete, J. L. Pablos, A. O. MartÃn, C. G. Juanatey, A. Adán, X. Montalbán, N. Borruel, G. OrtÃ, E. Holgado-MartÃn, C. GarcÃa-Vidal, C. Vizcaya-Morales, V. MartÃn-Vázquez, and M. González-Gay. 2020. Critical role of interleukin (IL)-17 in inflammatory and immune disorders. an updated review of the evidence focusing in controversies 19 (1): 102429.
Ruiz de Morales, J. M. G., L. Puig, E. Daudén, J. D. Cañete, J.L. Pablos, A. O. Martín, C. G. Juanatey, A. Adán, X. Montalbán, N. Borruel, G. Ortí, E. Holgado-Martín, C. García-Vidal, C. Vizcaya-Morales, V. Martín-Vázquez, and M. González-Gay. 2020. Critical role of interleukin (IL)-17 in inflammatory and immune disorders:an updated review of the evidence focusing in controversies. Autoimmunity reviews 19 (1): 102429.
Beringer, A., and P. Miossec. 2019. Systemic effects of IL-17 in inflammatory arthritis. Nature reviews. Rheumatology 15 (8): 491–501.
Funding
This research was financially supported by National Natural Science Foundation of China (NSFC) (NO. 82274390); Guangzhou Science and Technology Plan Project (NO. 2023A04J1289); Natural Science Foundation of Guangdong Province (NO. 2019A1515011636, 2022A1515110789); Guangzhou Key Laboratory of Formula-Pattern of Traditional Chinese Medicine (NO. 202102010014).
Author information
Authors and Affiliations
Contributions
QX, SZ, and YG designed and supervised the experiments. NL, SC, and WD conducted the experiments. WD and ZG analyzed the data and prepared the manuscript. NL and QX reviewed and revised the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, N., Chen, S., Deng, W. et al. Kaempferol Attenuates Gouty Arthritis by Regulating the Balance of Th17/Treg Cells and Secretion of IL-17. Inflammation 46, 1901–1916 (2023). https://doi.org/10.1007/s10753-023-01849-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-023-01849-8