Abstract
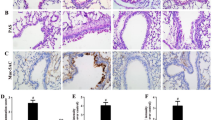
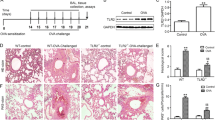
Asthma is a chronic disease closely related to airway inflammation. It has been proven that type 2 innate lymphoid cells (ILC2s) play an essential role in airway inflammation in asthma. Furthermore, there is growing evidence that Follistatin-like 1 (FSTL1) can participate in various inflammatory reactions mediated by the JAK/STAT signaling pathway, among others. Therefore, we put forward a new hypothesis: FSTL1 promotes asthmatic airway inflammation by activating ILC2. This study generated an ovalbumin-sensitized asthma model in C57BL/6 and Fstl1+/− mice. The results showed that the absolute number and the proportion of ILC2 in the ovalbumin-challenged Fstl1+/− group were lower than in the ovalbumin-challenged wild-type group. We also measured the levels of Th2-type cytokines in the serum and bronchoalveolar lavage fluid (BALF) of mice and found that the corresponding cytokines in the Fstl1+/− were lower than in the wild-type groups. Finally, we tested whether MEK-JAK-STAT-GATA3 is the specific pathway for FSTL1 to activate ILC2, and further tested our working hypothesis by adding various inhibitors of proteins from this pathway. Overall, these findings reveal that FSTL1 can activate ILC2 through MEK-JAK-STAT-GATA3 to promote airway inflammation and participate in the pathogenesis of asthma.







Similar content being viewed by others
Data Availability
All data and materials are available from the corresponding author upon request.
Abbreviations
- ILC2s:
-
Type 2 innate lymphoid cells
- FSTL1:
-
Follistatin-like 1
- JAK:
-
Janus kinase
- STAT:
-
Signal transducer and activator of transcription
- OVA:
-
Ovalbumin
- WT:
-
Wild-type
- BALF:
-
Bronchoalveolar lavage fluid
- MEK:
-
Mitogen-activated protein kinase kinase
- GATA3:
-
GATA binding protein 3
- DCs:
-
Dendritic cells
- IL-4:
-
Interleukin-4
- SCA1:
-
Spinocerebellar ataxia type 1
- KLRG1:
-
Killer cell lectin-like receptor G1
- TSC-36:
-
TGF-β1-stimulated clone 36
- SPARC:
-
Secreted protein acidic and rich in cysteine
- SADs:
-
Systemic autoimmune diseases
- IFN-γ:
-
Interferon-γ
- TNF-α:
-
Tumor necrosis factor-α
- NLRP3:
-
The nod-like receptor family, pyrin domain-containing 3
- TSLP:
-
Thymic stromal lymphopoietin
- PBS:
-
Phosphate-buffered saline
- TBST:
-
Tris-buffered saline containing 0.05% (v/v) Tween 20
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- ELISA:
-
Enzyme-linked immunosorbent assay
- MAPK:
-
Mitogen-activated protein kinase
- NF-κB:
-
Nuclear factor kappa B
- FO:
-
FSTL1+/− OVA
- WO:
-
WT OVA
- FC:
-
FSTL1+/− CONTROL
- WC:
-
WT CONTROL
References
Global Initiative for Asthma. 2020. Global strategy for asthma management and prevention. Available from:www.ginasthma.org.
Gandhi, N.A., B.L. Bennett, N.M. Graham, G. Pirozzi, N. Stahl, and G.D. Yancopoulos. 2016. Targeting key proximal drivers of type 2 inflammation in disease. Nature reviews. Drug discovery 15 (1): 35–50.
Kucuksezer, U.C., C. Ozdemir, M. Akdis, and C.A. Akdis. 2018. Precision/personalized medicine in allergic diseases and asthma. Archivum immunologiae et therapiae experimentalis 66 (6): 431–442.
Krabbendam, L., S.M. Bal, H. Spits, and K. Golebski. 2018. New insights into the function, development, and plasticity of type 2 innate lymphoid cells. Immunological reviews 286 (1): 74–85.
Vivier, E., D. Artis, M. Colonna, A. Diefenbach, J.P. Di Santo, G. Eberl, S. Koyasu, R.M. Locksley, A. McKenzie, R.E. Mebius, F. Powrie, and H. Spits. 2018. Innate lymphoid cells: 10 years on. Cell 174 (5): 1054–1066.
McKenzie, A.N. 2014. Type-2 innate lymphoid cells in asthma and allergy. Annals of the American Thoracic Society 11 (Suppl 5): S263–S270.
Mjösberg, J.M., S. Trifari, N.K. Crellin, C.P. Peters, C.M. van Drunen, B. Piet, W.J. Fokkens, T. Cupedo, and H. Spits. 2011. Human IL-25- and IL-33-responsive type 2 innate lymphoid cells are defined by expression of CRTH2 and CD161. Nature immunology 12 (11): 1055–1062.
Puttur, F., L. Denney, L.G. Gregory, J. Vuononvirta, R. Oliver, L.J. Entwistle, S.A. Walker, M.B. Headley, E.J. McGhee, J.E. Pease, M.F. Krummel, L.M. Carlin, and C.M. Lloyd. 2019. Pulmonary environmental cues drive group 2 innate lymphoid cell dynamics in mice and humans. Science immunology 4(36): eaav7638.
Ealey, K.N., and S. Koyasu. 2017. How many subsets of innate lymphoid cells do we need? Immunity 46 (1): 10–13.
Liu, T., J. Wu, J. Zhao, J. Wang, Y. Zhang, L. Liu, L. Cao, Y. Liu, and L. Dong. 2015. Type 2 innate lymphoid cells: A novel biomarker of eosinophilic airway inflammation in patients with mild to moderate asthma. Respiratory medicine 109 (11): 1391–1396.
Stadhouders, R., B. Li, M. de Bruijn, A. Gomez, T.N. Rao, H.J. Fehling, and van IJcken, W., Lim, A. I., Di Santo, J. P., Graf, T., and Hendriks, R. W. 2018. Epigenome analysis links gene regulatory elements in group 2 innate lymphocytes to asthma susceptibility. The Journal of allergy and clinical immunology 142 (6): 1793–1807.
Sundaram, G.M., J.E. Common, F.E. Gopal, S. Srikanta, K. Lakshman, D.P. Lunny, T.C. Lim, V. Tanavde, E.B. Lane, and P. Sampath. 2013. ‘See-saw’ expression of microRNA-198 and FSTL1 from a single transcript in wound healing. Nature 495 (7439): 103–106.
Mattiotti, A., S. Prakash, P. Barnett, and M. van den Hoff. 2018. Follistatin-like 1 in development and human diseases. Cellular and molecular life sciences : CMLS 75 (13): 2339–2354.
Clutter, S.D., D.C. Wilson, A.D. Marinov, and R. Hirsch. 2009. Follistatin-like protein 1 promotes arthritis by up-regulating IFN-gamma. Journal of immunology 182 (1): 234–239.
Li, W., M. Alahdal, Z. Deng, J. Liu, Z. Zhao, X. Cheng, X. Chen, J. Li, J. Yin, Y. Li, G. Wang, D. Wang, K. Tang, and J. Zhang. 2020. Molecular functions of FSTL1 in the osteoarthritis. International immunopharmacology 83: 106465.
Li, D., Y. Wang, N. Xu, Q. Wei, M. Wu, X. Li, P. Zheng, S. Sun, Y. Jin, G. Zhang, R. Liao, and P. Zhang. 2011. Follistatin-like protein 1 is elevated in systemic autoimmune diseases and correlated with disease activity in patients with rheumatoid arthritis. Arthritis research & therapy 13 (1): R17.
Chaly, Y., Y. Fu, A. Marinov, B. Hostager, W. Yan, B. Campfield, J.A. Kellum, D. Bushnell, Y. Wang, J. Vockley, and R. Hirsch. 2014. Follistatin-like protein 1 enhances NLRP3 inflammasome-mediated IL-1β secretion from monocytes and macrophages. European journal of immunology 44 (5): 1467–1479.
Liu, Y., J. Wei, Y. Zhao, Y. Zhang, Y. Han, B. Chen, K. Cheng, J. Jia, L. Nie, and L. Cheng. 2017. Follistatin-like protein 1 promotes inflammatory reactions in nucleus pulposus cells by interacting with the MAPK and NFκB signaling pathways. Oncotarget 8 (26): 43023–43034.
Liu, Y., T. Liu, J. Wu, T. Li, X. Jiao, H. Zhang, J. Zhao, J. Wang, L. Liu, L. Cao, S. Li, J. Xu, J. Xu, X. Ma, L. Yang, and L. Dong. 2017. The correlation between FSTL1 expression and airway remodeling in asthmatics. Mediators of inflammation 2017: 7918472.
Miller, M., A. Beppu, P. Rosenthal, A. Pham, S. Das, M. Karta, D.J. Song, C. Vuong, T. Doherty, M. Croft, B. Zuraw, X. Zhang, X. Gao, S. Aceves, F. Chouiali, Q. Hamid, and D.H. Broide. 2015. Fstl1 promotes asthmatic airway remodeling by inducing oncostatin M. Journal of immunology 195 (8): 3546–3556.
Miller, M., S. Esnault, R.C. Kurten, E.A. Kelly, A. Beppu, S. Das, P. Rosenthal, J. Ramsdell, M. Croft, B. Zuraw, N. Jarjour, Q. Hamid, and D.H. Broide. 2016. Segmental allergen challenge increases levels of airway follistatin-like 1 in patients with asthma. The Journal of allergy and clinical immunology 138 (2): 596-599.e4.
Liu, T., Y. Liu, M. Miller, L. Cao, J. Zhao, J. Wu, J. Wang, L. Liu, S. Li, M. Zou, J. Xu, D.H. Broide, and L. Dong. 2017. Autophagy plays a role in FSTL1-induced epithelial mesenchymal transition and airway remodeling in asthma. American journal of physiology. Lung cellular and molecular physiology 313(1): L27–L40.
Li, K.C., F.X. Zhang, C.L. Li, F. Wang, M.Y. Yu, Y.Q. Zhong, K.H. Zhang, Y.J. Lu, Q. Wang, X.L. Ma, J.R. Yao, J.Y. Wang, L.B. Lin, M. Han, Y.Q. Zhang, R. Kuner, H.S. Xiao, L. Bao, X. Gao, and X. Zhang. 2011. Follistatin-like 1 suppresses sensory afferent transmission by activating Na+, K+-ATPase. Neuron 69 (5): 974–987.
Deo, S.S., K.J. Mistry, A.M. Kakade, and P.V. Niphadkar. 2010. Role played by Th2 type cytokines in IgE mediated allergy and asthma. Lung India: Official organ of Indian Chest Society 27 (2): 66–71.
Kay, A.B. 1996. TH2-type cytokines in asthma. Annals of the New York Academy of Sciences 796: 1–8.
Yu, Q.N., Y.B. Guo, X. Li, C.L. Li, W.P. Tan, X.L. Fan, Z.L. Qin, D. Chen, W.P. Wen, S.G. Zheng, and Q.L. Fu. 2018. ILC2 frequency and activity are inhibited by glucocorticoid treatment via STAT pathway in patients with asthma. Allergy 73 (9): 1860–1870.
Ni, S., C. Li, N. Xu, X. Liu, W. Wang, W. Chen, Y. Wang, and A.J. van Wijnen. 2018. Follistatin-like protein 1 induction of matrix metalloproteinase 1, 3 and 13 gene expression in rheumatoid arthritis synoviocytes requires MAPK, JAK/STAT3 and NF-κB pathways. Journal of cellular physiology 234 (1): 454–463.
Huang, K., T. Yang, J. Xu, L. Yang, J. Zhao, X. Zhang, et al. 2019. Prevalence, risk factors, and management of asthma in China: A national cross-sectional study. Lancet 394 (10196): 407–418.
Busse, W.W., W.F. Calhoun, and J.D. Sedgwick. 1993. Mechanism of airway inflammation in asthma. The American review of respiratory disease 147 (6 Pt 2): S20–S24.
Ma, B., S.S. Athari, E. Mehrabi Nasab, and L. Zhao. 2021. PI3K/AKT/mTOR and TLR4/MyD88/NF-κB signaling inhibitors attenuate pathological mechanisms of allergic asthma. Inflammation. https://doi.org/10.1007/s10753-021-01466-3.
Skevaki, C., and H. Renz. 2018. Advances in mechanisms of allergic disease in 2017. The Journal of allergy and clinical immunology 142 (6): 1730–1739.
Lara-Pezzi, E., L.E. Felkin, E.J. Birks, P. Sarathchandra, K.D. Panse, R. George, J.L. Hall, M.H. Yacoub, N. Rosenthal, and P.J. Barton. 2008. Expression of follistatin-related genes is altered in heart failure. Endocrinology 149 (11): 5822–5827.
Widera, C., R. Horn-Wichmann, T. Kempf, K. Bethmann, B. Fiedler, S. Sharma, R. Lichtinghagen, H. Leitolf, B. Ivandic, H.A. Katus, E. Giannitsis, and K.C. Wollert. 2009. Circulating concentrations of follistatin-like 1 in healthy individuals and patients with acute coronary syndrome as assessed by an immunoluminometric sandwich assay. Clinical chemistry 55 (10): 1794–1800.
Chiou, J., C.Y. Su, Y.H. Jan, C.J. Yang, M.S. Huang, Y.L. Yu, and M. Hsiao. 2017. Decrease of FSTL1-BMP4-Smad signaling predicts poor prognosis in lung adenocarcinoma but not in squamous cell carcinoma. Scientific reports 7 (1): 9830.
Liu, Y., X. Tan, W. Liu, X. Chen, X. Hou, D. Shen, Y. Ding, J. Yin, L. Wang, H. Zhang, Y. Yu, J. Hou, T.C. Thompson, and G. Cao. 2018. Follistatin-like protein 1 plays a tumor suppressor role in clear-cell renal cell carcinoma. Chinese journal of cancer 37 (1): 2.
Lund, S., H.H. Walford, and T.A. Doherty. 2013. Type 2 innate lymphoid cells in allergic disease. Current immunology reviews 9 (4): 214–221.
Lambrecht, B.N., and H. Hammad. 2015. The immunology of asthma. Nature immunology 16 (1): 45–56.
Hu, P.F., C.Y. Ma, F.F. Sun, W.P. Chen, and L.D. Wu. 2019. Follistatin-like protein 1 (FSTL1) promotes chondrocyte expression of matrix metalloproteinase and inflammatory factors via the NF-κB pathway. Journal of cellular and molecular medicine 23 (3): 2230–2237.
Guo, J., W. Liang, J. Li, and J. Long. 2016. Knockdown of FSTL1 inhibits oxLDL-induced inflammation responses through the TLR4/MyD88/NF-κB and MAPK pathway. Biochemical and biophysical research communications 478 (4): 1528–1533.
Nakamura, Y., and M. Hoshino. 2005. TH2 cytokines and associated transcription factors as therapeutic targets in asthma. Current drug targets. Inflammation and allergy 4 (2): 267–270.
Mjösberg, J., J. Bernink, K. Golebski, J.J. Karrich, C.P. Peters, B. Blom, A.A. te Velde, W.J. Fokkens, C.M. van Drunen, and H. Spits. 2012. The transcription factor GATA3 is essential for the function of human type 2 innate lymphoid cells. Immunity 37 (4): 649–659.
Zhang, D.H., L. Yang, L. Cohn, L. Parkyn, R. Homer, P. Ray, and A. Ray. 1999. Inhibition of allergic inflammation in a murine model of asthma by expression of a dominant-negative mutant of GATA-3. Immunity 11 (4): 473–482.
Wang, J., L. Xiao, L. Zhu, M. Hu, Q. Wang, and T. Yan. 2015. The effect of synthetic salidroside on cytokines and airway inflammation of asthma induced by diisocyanate (TDI) in mice by regulating GATA3/T-bet. Inflammation 38 (2): 697–704.
Chong, L., W. Zhang, Y. Nie, G. Yu, L. Liu, L. Lin, S. Wen, L. Zhu, and C. Li. 2014. Protective effect of curcumin on acute airway inflammation of allergic asthma in mice through Notch1-GATA3 signaling pathway. Inflammation 37 (5): 1476–1485.
Opina, M.T., and W.C. Moore. 2017. Phenotype-driven therapeutics in severe asthma. Current allergy and asthma reports 17 (2): 10.
Acknowledgements
We would like to thank Prof. Xu Zhang and Xiang Gao for generous help with the FSTL1 knockout mice.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 81770029).
Author information
Authors and Affiliations
Contributions
Siyuan Huang designed the experiments, analyzed the results, designed the images, and wrote the manuscript. Siyuan Huang performed the experiments with the assistance of Rong Zeng and Xinrui Qiao. Shuo Li and Dong Zhang reviewed the results and the manuscript. Jing Wang and Rong Zeng assisted in solving the problem during the revision process. Liang Dong approved final version of manuscript.
Ethics declarations
Ethics Approval
All mouse experiments were approved by the Institutional Animal Care and Use Committee of Shandong University.
Consent to Participate
Not applicable.
Consent to Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, S., Zeng, R., Wang, J. et al. Follistatin-Like 1 Induces the Activation of Type 2 Innate Lymphoid Cells to Promote Airway Inflammation in Asthma. Inflammation 45, 904–918 (2022). https://doi.org/10.1007/s10753-021-01594-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-021-01594-w