1Abstract
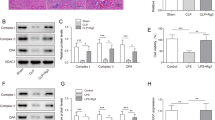
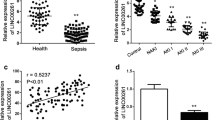
Sepsis is recognized as the acute systemic inflammatory response to severe infection. It is main cause of multiple organ system dysfunction and even organ failure. Long non-coding RNA X inactivate-specific transcript (XIST) is implicated in multiple inflammatory diseases. The aim of present work was to investigate the precise mechanism of XIST underlying sepsis-induced acute liver injury. Rats were underwent cecal ligation and puncture (CLP) to establish sepsis-induced the animal models of acute liver injury. Hematoxylin and eosin (H&E) staining was performed to observe pathological alterations. Corresponding commercial assay kits were employed to analyze the levels of inflammatory cytokines and oxidative stress. Western blot and reverse transcriptional quantitative PCR (RT-qPCR) were performed to determine the expression of proteins and target genes. Finally, TUNEL and CCK-8 assays were performed to test apoptosis rate and cell viability, respectively. In our study, XIST and BRD4 were highly expressed in serum of patients with sepsis-induced acute liver injury. XIST knockdown ameliorated sepsis-induced acute liver injury and inhibited inflammation, oxidative stress, and cell apoptosis in sepsis-induced acute liver injury rats. Interestingly, XIST knockdown downregulated the expression of BRD4, and BRD4 overexpression abolished the impacts of XIST knockdown on inflammation, oxidative stress, and apoptosis of that LPS-induced Kupffer cells. We conclude that lncRNA XIST silencing protects against sepsis-induced acute liver injury via inhibition of the BRD4 expression. Therefore, XIST may be a biomarker for sepsis diagnosis and treatment.







Similar content being viewed by others
Abbreviations
- XIST:
-
X inactivate-specific transcript
- CLP:
-
cecal ligation and puncture
- lncRNAs:
-
long non-coding RNAs
- LPS:
-
lipopolysaccharide
- AST:
-
aminotransferase
- ALT:
-
alanine aminotransferase
- MDA:
-
malondialdehyde
- MPO:
-
myeloperoxidase
- GSH:
-
glutathione peroxidase
References
Wang, Y.M., R. Ji, W.W. Chen, S.W. Huang, Y.J. Zheng, Z.T. Yang, H.P. Qu, H. Chen, E.Q. Mao, Y. Chen, and E.Z. Chen. 2019. Paclitaxel alleviated sepsis-induced acute lung injury by activating MUC1 and suppressing TLR-4/NF-kappaB pathway. Drug Design, Development and Therapy 13: 3391–3404.
Zhang, H., W. Wang, H. Fang, Y. Yang, X. Li, J. He, X. Jiang, et al. 2014. GSK-3ﺧ٢ inhibition attenuates CLP-induced liver injury by reducing inflammation and hepatic cell apoptosis. Mediators of Inflammation 2014: 629507.
Zhang, Z., N. Han, and Y. Shen. 2020. S100A12 promotes inflammation and cell apoptosis in sepsis-induced ARDS via activation of NLRP3 inﻡ،∙ammasome signaling. Molecular Immunology 122: 38–48.
Chen, Y.L., G. Xu, X. Liang, J. Wei, J. Luo, G.N. Chen, X.D. Yan, X.P. Wen, M. Zhong, and X. Lv. 2016. Inhibition of hepatic cells pyroptosis attenuates CLP-induced acute liver injury. American Journal of Translational Research 8: 5685–5695.
Savio, L.E.B., P. de Andrade Mello, V.R. Figliuolo, T.F. de Avelar Almeida, P.T. Santana, S.D.S. Oliveira, C.L.M. Silva, L. Feldbrügge, E. Csizmadia, R.D. Minshall, M.S. Longhi, Y. Wu, S.C. Robson, and R. Coutinho-Silva. 2017. CD39 limits P2X7 receptor inflammatory signaling and attenuates sepsis-induced liver injury. Journal of Hepatology 67: 716–726.
Recknagel, P., F.A. Gonnert, M. Westermann, S. Lambeck, A. Lupp, A. Rudiger, A. Dyson, J.E. Carré, A. Kortgen, C. Krafft, J. Popp, C. Sponholz, V. Fuhrmann, I. Hilger, R.A. Claus, N.C. Riedemann, R. Wetzker, M. Singer, M. Trauner, and M. Bauer. 2012. Liver dysfunction and phosphatidylinositol-3-kinase signalling in early sepsis: experimental studies in rodent models of peritonitis. PLoS Medicine 9: e1001338.
Yan, J., and S. Li. 2014. The role of the liver in sepsis. International Reviews of Immunology 33: 498–510.
Zhang, Y., Y. Zhu, G. Gao, and Z. Zhou. 2019. Knockdown XIST alleviates LPS-induced WI-38 cell apoptosis and inflammation injury via targeting miR-370-3p/TLR4 in acute pneumonia. Cell Biochemistry and Function 37: 348–358.
Ma, M., Y. Pei, X. Wang, J. Feng, Y. Zhang, and M.Q. Gao. 2019. LncRNA XIST mediates bovine mammary epithelial cell inflammatory response via NF-ﺧﻑB/NLRP3 inflammasome pathway. Cell Proliferation 52: e12525.
Sathishkumar, C., P. Prabu, V. Mohan, and M. Balasubramanyam. 2018. Linking a role of lncRNAs (long non-coding RNAs) with insulin resistance, accelerated senescence, and inflammation in patients with type 2 diabetes. Human Genomics 12: 41.
Xu, G., L. Mo, C. Wu, X. Shen, H. Dong, L. Yu, P. Pan, and K. Pan. 2019. The miR-15a-5p-XIST-CUL3 regulatory axis is important for sepsis-induced acute kidney injury. Renal Failure 41: 955–966.
Singer, M., C.S. Deutschman, C.W. Seymour, M. Shankar-Hari, D. Annane, M. Bauer, R. Bellomo, G.R. Bernard, J.D. Chiche, C.M. Coopersmith, R.S. Hotchkiss, M.M. Levy, J.C. Marshall, G.S. Martin, S.M. Opal, G.D. Rubenfeld, T. van der Poll, J.L. Vincent, and D.C. Angus. 2016. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 315: 801–810.
Bakker, J., R. Grover, A. McLuckie, L. Holzapfel, J. Andersson, R. Lodato, D. Watson, S. Grossman, J. Donaldson, and J. Takala. 2004. Administration of the nitric oxide synthase inhibitor NG-methyl-L-arginine hydrochloride (546C88) by intravenous infusion for up to 72 hours can promote the resolution of shock in patients with severe sepsis: results of a randomized, double-blind, placebo-controlled multicenter study (study no. 144-002). Critical Care Medicine 32: 1–12.
Zhu, Y., K. Wang, Z. Ma, D. Liu, Y. Yang, M. Sun, A. Wen, Y. Hao, S. Ma, F. Ren, Z. Xin, Y. Li, S. di, and J. Liu. 2019. SIRT1 activation by butein attenuates sepsis-induced brain injury in mice subjected to cecal ligation and puncture via alleviating inflammatory and oxidative stress. Toxicology and Applied Pharmacology 363: 34–46.
Liang, X., T. Li, Q. Zhou, S. Pi, Y. Li, X. Chen, Z. Weng, H. Li, Y. Zhao, H. Wang, and Y. Chen. 2019. Mesenchymal stem cells attenuate sepsis-induced liver injury via inhibiting M1 polarization of Kupffer cells. Molecular and Cellular Biochemistry 452: 187–197.
Liang, H., X. Ding, Y. Yu, H. Zhang, L. Wang, Q. Kan, S. Ma, F. Guan, and T. Sun. 2019. Adipose-derived mesenchymal stem cells ameliorate acute liver injury in rat model of CLP induced-sepsis via sTNFR1. Experimental Cell Research 383: 111465.
Wang, S., F. Cao, X. Gu, J. Chen, R. Xu, Y. Huang, and L. Ying. 2019. LncRNA XIST, as a ceRNA of miR-204, aggravates lipopolysaccharide-induced acute respiratory distress syndrome in mice by upregulating IRF2. International Journal of Clinical and Experimental Pathology 12: 2425–2434.
Huang, M., S. Cai, and J. Su. 2019. The pathogenesis of sepsis and potential therapeutic targets. International Journal of Molecular Sciences 20.
Stearns-Kurosawa, D.J., M.F. Osuchowski, C. Valentine, S. Kurosawa, and D.G. Remick. 2011. The pathogenesis of sepsis. Annual Review of Pathology 6: 19–48.
van der Poll, T., F.L. van de Veerdonk, B.P. Scicluna, and M.G. Netea. 2017. The immunopathology of sepsis and potential therapeutic targets. Nature Reviews. Immunology 17: 407–420.
Zhou, Y., Y. Gu, and J. Liu. 2019. BRD4 suppression alleviates cerebral ischemia-induced brain injury by blocking glial activation via the inhibition of inflammatory response and pyroptosis. Biochemical and Biophysical Research Communications 519: 481–488.
Bao, Y., X. Wu, J. Chen, X. Hu, F. Zeng, J. Cheng, H. Jin, X. Lin, and L.F. Chen. 2017. Brd4 modulates the innate immune response through Mnk2-eIF4E pathway-dependent translational control of IﺧﻑBﺧ١. Proceedings of the National Academy of Sciences of the United States of America 114: E3993–E4001.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shen, C., Li, J. LncRNA XIST silencing protects against sepsis-induced acute liver injury via inhibition of BRD4 expression. Inflammation 44, 194–205 (2021). https://doi.org/10.1007/s10753-020-01321-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-020-01321-x