Abstract
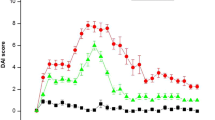
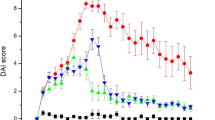
Acute inflammatory bowel disease (AIBD) is a wide clinical entity including severe gastrointestinal pathologies with common histopathological basis. Epidemiologically increasing diseases, such as necrotizing enterocolitis (NEC), gastrointestinal graft versus host disease (GVHD), and the primary acute phase of chronic inflammatory bowel disease (CIBD), exhibit a high necessity for new therapeutic strategies. Mesenchymal stem cell (MSC) cellular therapy represents a promising option for the treatment of these diseases. In our study, we comparatively assess the efficacy of human MSCs derived from bone marrow (BM), umbilical cord blood (UCB), human embryonic stem cells (ESCs), or human-induced pluripotent stem cells (iPSCs) in a mouse model of chemically induced acute enterocolitis. The laboratory animals were provided ad libitum potable dextrane sulfate sodium solution (DSS) in order to reproduce an AIBD model and then individually exposed intraperitoneally to MSCs derived from BM (BM-MSCs), UCB (UCB-MSCs), ESCs (ESC-MSCs), or iPSCs (iPSC-MSCs). The parameters used to evaluate the cellular treatment efficacy were the animal survival prolongation and the histopathological-macroscopic picture of bowel sections. Although all categories of mesenchymal stem cells led to statistically significant survival prolongation compared to the control group, significant clinical and histopathological improvement was observed only in mice receiving BM-MSCs and UCB-MSCs. Our results demonstrated that the in vivo anti-inflammatory effect of ESC-MSCs and iPSC-MSCs was inferior to that of UCB-MSCs and BM-MSCs. Further investigation will clarify the potential of ESCs and iPSC-derived MSCs in AIBD treatment.




Similar content being viewed by others
References
Duci, Miriam, Francesco Fascetti-Leon, Marta Erculiani, Elena Priante, Maria Elena Cavicchiolo, Giovanna Verlato, and Piergiorgio Gamba. 2018. Neonatal independent predictors of severe NEC. Pediatric Surgery International 34: 663–669. https://doi.org/10.1007/s00383-018-4261-1.
Boghossian, Nansi S., Marco Geraci, Erika M. Edwards, and Jeffrey D. Horbar. 2018. Neonatal and fetal growth charts to identify preterm infants <30 weeks gestation at risk of adverse outcomes. American Journal of Obstetrics and Gynecology 219: 195.e1–195.e14. https://doi.org/10.1016/j.ajog.2018.05.002.
Manjili, Masoud H., and Amir A. Toor. 2014. Etiology of GVHD: Alloreactivity or Impaired Cellular Adaptation? Immunological Investigations 43: 851–857. https://doi.org/10.3109/08820139.2014.953636.
Na, K.-S., Y.-S. Yoo, J.W. Mok, J.W. Lee, and C.-K. Joo. 2015. Incidence and risk factors for ocular GVHD after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplantation 50: 1459–1464.
Scarisbrick, J.J., F.L. Dignan, S. Tulpule, E.D. Gupta, S. Kolade, B. Shaw, F. Evison, et al. 2014. A multicentre UK study of GVHD following DLI: Rates of GVHD are high but mortality from GVHD is infrequent. Bone Marrow Transplantation 50: 62.
Ananthakrishnan, Ashwin N. 2015. Epidemiology and risk factors for IBD. Nature Reviews Gastroenterology & Hepatology 12: 205.
Kaplan, Gilaad G. 2014. Global variations in environmental risk factors for IBD. Nature Reviews Gastroenterology & Hepatology 11: 708.
Ng, Siew C., Charles N. Bernstein, Morten H. Vatn, Peter Laszlo Lakatos, Edward V. Loftus, Curt Tysk, Colm O’Morain, Bjorn Moum, and Jean-Frédéric Colombel. 2013. Geographical variability and environmental risk factors in inflammatory bowel disease. Gut 62: 630–649. https://doi.org/10.1136/gutjnl-2012-303661.
Aloi, Marina, Federica Nuti, Laura Stronati, and Salvatore Cucchiara. 2013. Advances in the medical management of paediatric IBD. Nature Reviews Gastroenterology & Hepatology 11: 99.
Crisan, Mihaela, Chien-Wen Chen, Mirko Corselli, Gabriella Andriolo, Lorenza Lazzari, and Bruno Péault. 2009. Perivascular multipotent progenitor cells in human organs. Annals of the New York Academy of Sciences 1176: 118–123. https://doi.org/10.1111/j.1749-6632.2009.04967.x.
Hass, Ralf, Cornelia Kasper, Stefanie Böhm, and Roland Jacobs. 2011. Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Communication and Signaling: CCS 9: 12. https://doi.org/10.1186/1478-811X-9-12.
Pittenger, M.F., A.M. Mackay, S.C. Beck, R.K. Jaiswal, R. Douglas, J.D. Mosca, M.A. Moorman, D.W. Simonetti, S. Craig, and D.R. Marshak. 1999. Multilineage potential of adult human mesenchymal stem cells. Science (New York, N.Y.) 284: 143–147.
Uccelli, Antonio, Lorenzo Moretta, and Vito Pistoia. 2006. Immunoregulatory function of mesenchymal stem cells. European Journal of Immunology 36: 2566–2573. https://doi.org/10.1002/eji.200636416.
Munneke, J. Marius, Melchior J.A. Spruit, Anne S. Cornelissen, Vera van Hoeven, Carlijn Voermans, and Mette D. Hazenberg. 2016. The Potential of Mesenchymal Stromal Cells as Treatment for Severe Steroid-Refractory Acute Graft-Versus-Host Disease: A Critical Review of the Literature. Transplantation 100: 2309–2314. https://doi.org/10.1097/TP.0000000000001029.
Sharma, Ratti Ram, Kathryn Pollock, Allison Hubel, and David McKenna. 2014. Mesenchymal stem or stromal cells: a review of clinical applications and manufacturing practices. Transfusion 54: 1418–1437. https://doi.org/10.1111/trf.12421.
Liu, Cai Xia, Ren Li Zhang, Jun Gao, Tao Li, Zi Ren, Can Quan Zhou, and An Min Wen. 2014. Derivation of human embryonic stem cell lines without any exogenous growth factors. Molecular Reproduction and Development 81: 470–479. https://doi.org/10.1002/mrd.22312.
Takahashi, Kazutoshi, and Shinya Yamanaka. 2006. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126: 663–676. https://doi.org/10.1016/j.cell.2006.07.024.
Chami, Belal, Amanda W.S. Yeung, Caryn van Vreden, Nicholas J.C. King, and Shisan Bao. 2014. The role of CXCR3 in DSS-induced colitis. PLoS One 9: e101622. https://doi.org/10.1371/journal.pone.0101622.
Li, B., R. Alli, P. Vogel, and T.L. Geiger. 2014. IL-10 modulates DSS-induced colitis through a macrophage-ROS-NO axis. Mucosal Immunology 7: 869–878. https://doi.org/10.1038/mi.2013.103.
Secunda, R., Vennila Rosy, A.M. Mohanashankar, M. Rajasundari, S. Jeswanth, and R. Surendran. 2015. Isolation, expansion and characterisation of mesenchymal stem cells from human bone marrow, adipose tissue, umbilical cord blood and matrix: a comparative study. Cytotechnology 67: 793–807. https://doi.org/10.1007/s10616-014-9718-z.
Luo, H.J., X.M. Xiao, J. Zhou, and W. Wei. 2015. Therapeutic influence of intraperitoneal injection of Wharton’s jelly-derived mesenchymal stem cells on oviduct function and fertility in rats with acute and chronic salpingitis. Genetics and molecular research: GMR 14: 3606–3617. https://doi.org/10.4238/2015.April.17.10.
Sun, Xue-cheng, Jian-sheng Wu, Jin-ming Wu, Zhi-ming Huang, and Yu. Zhen. 2013. Effects of intraperitoneal injection of marrow mesenchymal stem cells on intestinal barrier in acute pancreatitis. Zhonghua Yi Xue Za Zhi 93: 951–955.
Parys, M., N. Nelson, K. Koehl, R. Miller, J.B. Kaneene, J.M. Kruger, and V. Yuzbasiyan-Gurkan. 2016. Safety of Intraperitoneal Injection of Adipose Tissue-Derived Autologous Mesenchymal Stem Cells in Cats. Journal of Veterinary Internal Medicine 30: 157–163. https://doi.org/10.1111/jvim.13655.
Liu, Jingquan, Bin Shi, Kai Shi, and Hongze Zhang. 2015. Applications of induced pluripotent stem cells in the modeling of human inflammatory bowel diseases. Current Stem Cell Research & Therapy 10: 228–235.
Algeri, M., A. Conforti, A. Pitisci, N. Starc, L. Tomao, M.E. Bernardo, and F. Locatelli. 2015. Mesenchymal stromal cells and chronic inflammatory bowel disease. Immunology Letters 168: 191–200. https://doi.org/10.1016/j.imlet.2015.06.018.
Pérez-Merino, E.M., J.M. Usón-Casaús, C. Zaragoza-Bayle, J. Duque-Carrasco, L. Mariñas-Pardo, M. Hermida-Prieto, R. Barrera-Chacón, and M. Gualtieri. 2015. Safety and efficacy of allogeneic adipose tissue-derived mesenchymal stem cells for treatment of dogs with inflammatory bowel disease: Clinical and laboratory outcomes. Veterinary Journal (London, England: 1997) 206: 385–390. https://doi.org/10.1016/j.tvjl.2015.08.003.
Pérez-Merino, E.M., J.M. Usón-Casaús, J. Duque-Carrasco, C. Zaragoza-Bayle, L. Mariñas-Pardo, M. Hermida-Prieto, M. Vilafranca-Compte, R. Barrera-Chacón, and M. Gualtieri. 2015. Safety and efficacy of allogeneic adipose tissue-derived mesenchymal stem cells for treatment of dogs with inflammatory bowel disease: Endoscopic and histological outcomes. Veterinary Journal (London, England: 1997) 206: 391–397. https://doi.org/10.1016/j.tvjl.2015.07.023.
García-Arranz, Mariano, Maria Dolores Herreros, Carolina González-Gómez, Paloma de la Quintana, Héctor Guadalajara, Tihomir Georgiev-Hristov, Jacobo Trébol, and Damián Garcia-Olmo. 2016. Treatment of Crohn’s-Related Rectovaginal Fistula With Allogeneic Expanded-Adipose Derived Stem Cells: A Phase I-IIa Clinical Trial. Stem Cells Translational Medicine 5: 1441–1446. https://doi.org/10.5966/sctm.2015-0356.
Peng, Kaiyue, Xiaowen Qian, Zhiheng Huang, Junping Lu, Yuhuan Wang, Ying Zhou, Huijun Wang, Bingbing Wu, Ying Wang, Lingli Chen, Xiaowen Zhai, and Ying Huang. 2018. Umbilical Cord Blood Transplantation Corrects Very Early-Onset Inflammatory Bowel Disease in Chinese Patients With IL10RA-Associated Immune Deficiency. Inflammatory Bowel Diseases 24: 1416–1427. https://doi.org/10.1093/ibd/izy028.
Qu, Bo, Guo-Rong Xin, Li-Xia Zhao, Hui Xing, Li-Ying Lian, Hai-Yan Jiang, Jia-Zhao Tong, Bei-Bei Wang, and Shi-Zhu Jin. 2014. Testing stem cell therapy in a rat model of inflammatory bowel disease: role of bone marrow stem cells and stem cell factor in mucosal regeneration. PLoS One 9: e107891. https://doi.org/10.1371/journal.pone.0107891.
Park, Jin Seok, Tac-Ghee Yi, Jong-Min Park, Young Min Han, Jun-Hyung Kim, Dong-Hee Shin, Seon Ji Tak, Kyuheon Lee, Youn Sook Lee, Myung-Shin Jeon, Ki-Baik Hahm, Sun U. Song, and Seok Hee Park. 2015. Therapeutic effects of mouse bone marrow-derived clonal mesenchymal stem cells in a mouse model of inflammatory bowel disease. Journal of Clinical Biochemistry and Nutrition 57: 192–203. https://doi.org/10.3164/jcbn.15-56.
Fujino, Masayuki, Ping Zhu, Yusuke Kitazawa, Ji-Mei Chen, Jian Zhuang, and Xiao-Kang Li. 2014. Mesenchymal stem cells attenuate rat graft-versus-host disease. Methods in Molecular Biology (Clifton, N.J.) 1213: 341–353. https://doi.org/10.1007/978-1-4939-1453-1_28.
Eaton, Simon, Augusto Zani, Agostino Pierro, and Paolo De Coppi. 2013. Stem cells as a potential therapy for necrotizing enterocolitis. Expert Opinion on Biological Therapy 13: 1683–1689. https://doi.org/10.1517/14712598.2013.849690.
Drucker, Natalie A., Christopher J. McCulloh, Bo Li, Agostino Pierro, Gail E. Besner, and Troy A. Markel. 2018. Stem cell therapy in necrotizing enterocolitis: Current state and future directions. Seminars in Pediatric Surgery 27: 57–64. https://doi.org/10.1053/j.sempedsurg.2017.11.011.
Zani, Augusto, Mara Cananzi, Giuseppe Lauriti, Francesco Fascetti-Leon, Jack Wells, Bernard Siow, Mark F. Lythgoe, Agostino Pierro, Simon Eaton, and Paolo De Coppi. 2014. Amniotic fluid stem cells prevent development of ascites in a neonatal rat model of necrotizing enterocolitis. European Journal of Pediatric Surgery: Official Journal of Austrian Association of Pediatric Surgery ... [et Al] = Zeitschrift Fur Kinderchirurgie 24: 57–60. https://doi.org/10.1055/s-0033-1350059.
Hynes, K., R. Bright, V. Marino, J. Ng, P.J. Verma, S. Gronthos, and P.M. Bartold. 2018. Potential of iPSC-Derived Mesenchymal Stromal Cells for Treating Periodontal Disease. Stem Cells International 2018: 2601945–2601912. https://doi.org/10.1155/2018/2601945.
Soontararak, Sirikul, Lyndah Chow, Valerie Johnson, Jonathan Coy, William Wheat, Daniel Regan, and Steven Dow. 2018. Mesenchymal Stem Cells (MSC) Derived from Induced Pluripotent Stem Cells (iPSC) Equivalent to Adipose-Derived MSC in Promoting Intestinal Healing and Microbiome Normalization in Mouse Inflammatory Bowel Disease Model. Stem Cells Translational Medicine 7: 456–467. https://doi.org/10.1002/sctm.17-0305.
Acknowledgments
G.E., T.M., K.E.: conception and design, provision of study material manuscript writing, final approval of manuscript, scientific guidance K.A, V.I., S.I., M.A.: mesenchymal stem cell derivation P.E.: provision of experimental surgery structures and animal model design, D.E.: histopathological evaluation and over all special contribution to my father Kagias Dimitrios for his continuous support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The animal experiments were conducted under protocols reviewed and approved by Institutional Animal Care and Use Committee (Athens, Protocol Number K/5449). All procedures performed in studies involving animals were in accordance with the ethical standards of the institution at which the studies were conducted.
Conflict of Interest
The authors declare that they have no conflict of interest.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Kagia, A., Tzetis, M., Kanavakis, E. et al. Therapeutic Effects of Mesenchymal Stem Cells Derived From Bone Marrow, Umbilical Cord Blood, and Pluripotent Stem Cells in a Mouse Model of Chemically Induced Inflammatory Bowel Disease. Inflammation 42, 1730–1740 (2019). https://doi.org/10.1007/s10753-019-01033-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-019-01033-x