Abstract
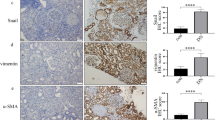
SUV39H1, the histone methyltransferase (HMTase) of histone H3 lysine 9 trimethylation (H3K9me3), is a known transcriptional repressor of inflammatory genes. The effect of SUV39H1 on inflammatory gene promoters under high-glucose stimulation in vascular smooth muscle cells (VSMCs), macrophages, and cardiomyocytes has been studied, but how SUV39H1 functions in renal tubules under diabetic conditions is unclear. Renal biopsy specimens of ten diabetic nephropathy (DN) subjects and seven non-DN minimal change diseases (MCD) subjects were collected. SUV39H1, IL-6, and MCP-1 expression in renal tissues were measured using immunohistochemical, while SUV39H1, H3K9me3, IL-6, and MCP-1 in human proximal tubular epithelial cells (HK-2) under varying glucose conditions were assayed by Western blot and ELISA. SUV39H1 was overexpressed in HK-2 cells; the regulation of SUV39H1 and H3K9me3 on NF-κB, IL-6, MCP-1, caspase 3, and apoptosis was measured. SUV39H1 was expressed more in diabetic human renal tubules. HK-2 cells with high glucose up-regulated IL-6 and MCP-1 in a dose- and time-dependent manner, and SUV39H1 expression was reduced with greater glucose and prolonged stimulation. Expression of H3K9me3 was synchronized with SUV39H1. Moreover, overexpression of SUV39H1 in high glucose environment was accompanied with increased H3K9me3 and decreased inflammation and apoptosis. SUV39H1 dysregulation may be involved in DN progression. Overexpression of SUV39H1 may reduce renal inflammation and apoptosis via epigenetic modulation, thus plays a protective role in DN.





Similar content being viewed by others
References
Guariguata, L., D.R. Whiting, I. Hambleton, J. Beagley, U. Linnenkamp, and J.E. Shaw. 2014. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Research and Clinical Practice 103: 137–149.
Cooper, M.E. 2012. Diabetes: treating diabetic nephropathy-still an unresolved issue. Nature Reviews. Endocrinology 8: 515–516.
Kanwar, Y.S., L. Sun, P. Xie, F.Y. Liu, and S. Chen. 2011. A glimpse of various pathogenetic mechanisms of diabetic nephropathy. Annual Review of Pathology 6: 395–423.
Hasegawa, G., K. Nakano, M. Sawada, K. Uno, Y. Shibayama, K. Ienaga, and M. Kondo. 1991. Possible role of tumor necrosis factor and interleukin-1 in the development of diabetic nephropathy. Kidney International 40: 1007–1012.
Suzuki, D., M. Miyazaki, R. Naka, T. Koji, M. Yagame, K. Jinde, M. Endoh, Y. Nomoto, and H. Sakai. 1995. In situ hybridization of interleukin 6 in diabetic nephropathy. Diabetes 44: 1233–1238.
Navarro, J.F., C. Mora, M. Maca, and J. Garca. 2003. Inflammatory parameters are independently associated with urinary albumin in type 2 diabetes mellitus. American Journal of Kidney Diseases 42: 53–61.
Moriwaki, Y., T. Yamamoto, Y. Shibutani, E. Aoki, Z. Tsutsumi, S. Takahashi, H. Okamura, M. Koga, M. Fukuchi, and T. Hada. 2003. Elevated levels of interleukin-18 and tumor necrosis factor-alpha in serum of patients with type 2 diabetes mellitus: relationship with diabetic nephropathy. Metabolism 52: 605–608.
Wei, Q., G. Dong, J.K. Chen, G. Ramesh, and Z. Dong. 2013. Bax and Bak have critical roles in ischemic acute kidney injury in global and proximal tubule-specific knockout mouse models. Kidney International 84: 138–148.
Johnson, A., and L.A. DiPietro. 2013. Apoptosis and angiogenesis: an evolving mechanism for fibrosis. The FASEB Journal 27: 3893–3901.
Kato, M., and R. Natarajan. 2014. Diabetic nephropathy—emerging epigenetic mechanisms. Nature Reviews. Nephrology 10: 517–530.
Villeneuve, L.M., M.A. Reddy, L.L. Lanting, M. Wang, L. Meng, and R. Natarajan. 2008. Epigenetic histone H3 lysine 9 methylation in metabolic memory and inflammatory phenotype of vascular smooth muscle cells in diabetes. Proceedings of the National Academy of Sciences of the United States of America 105: 9047–9052.
Keating, S.T., and A. El-Osta. 2013. Glycemic memories and the epigenetic component of diabetic nephropathy. Current Diabetes Reports 13: 574–581.
Li, M.F., R. Zhang, T.T. Li, M.Y. Chen, L.X. Li, J.X. Lu, and W.P. Jia. 2016. High glucose increases the expression of inflammatory cytokine genes in macrophages through H3K9 methyltransferase mechanism. Journal of Interferon & Cytokine Research 36: 48–61.
Yang, B., J. Yang, J. Bai, P. Pu, J. Liu, F. Wang, and B. Ruan. 2014. Suv39h1 protects from myocardial ischemia-reperfusion injury in diabetic rats. Cellular Physiology and Biochemistry 33: 1176–1185.
Zhu, X., X. Xiong, S. Yuan, L. Xiao, X. Fu, Y. Yang, C. Tang, L. He, F. Liu, and L. Sun. 2016. Validation of the interstitial fibrosis and tubular atrophy on the new pathological classification in patients with diabetic nephropathy: a single-center study in China. Journal of Diabetes and its Complications 30: 537–541.
Schotta, G., A. Ebert, and G. Reuter. 2003. SU(VAR)3-9 is a conserved key function in heterochromatic gene silencing. Genetica 117: 149–158.
Lachner, M., D. O'Carroll, S. Rea, K. Mechtler, and T. Jenuwein. 2001. Methylation of histone H3 lysine 9 creates a binding site for HP1 proteins. Nature 410: 116–120.
Vaute, O., E. Nicolas, L. Vandel, and D. Trouche. 2002. Functional and physical interaction between the histone methyl transferase Suv39H1 and histone deacetylases. Nucleic Acids Research 30: 475–481.
Grgic, I., G. Campanholle, V. Bijol, C. Wang, V.S. Sabbisetti, T. Ichimura, B.D. Humphreys, and J.V. Bonventre. 2012. Targeted proximal tubule injury triggers interstitial fibrosis and glomerulosclerosis. Kidney International 82: 172–183.
Sanz, A.B., M.D. Sanchez-Nino, A.M. Ramos, J.A. Moreno, B. Santamaria, M. Ruiz-Ortega, J. Egido, and A. Ortiz. 2010. NF-kappaB in renal inflammation. Journal of the American Society of Nephrology 21: 1254–1262.
Mezzano, S., C. Aros, A. Droguett, M.E. Burgos, L. Ardiles, C. Flores, H. Schneider, M. Ruiz-Ortega, and J. Egido. 2004. NF-kappaB activation and overexpression of regulated genes in human diabetic nephropathy. Nephrology, Dialysis, Transplantation 19: 2505–2512.
Zhou, L., D.Y. Xu, W.G. Sha, L. Shen, G.Y. Lu, X. Yin, and M.J. Wang. 2015. High glucose induces renal tubular epithelial injury via Sirt1/NF-kappaB/microR-29/Keap1 signal pathway. Journal of Translational Medicine 13: 352.
Funding
This work was supported by research grants (81373227, 81400721, and 81470947) from the National Natural Science Foundation of China. It was also supported by a research fund from Natural Science Foundation of Hunan Province (Grant No. 2016JJ3173).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of Interest
The authors declare that they have no conflict of interest.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Wang, J., Yan, W., Peng, X. et al. Functional Role of SUV39H1 in Human Renal Tubular Epithelial Cells Under High-glucose Ambiance. Inflammation 41, 1–10 (2018). https://doi.org/10.1007/s10753-017-0657-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-017-0657-7