Abstract
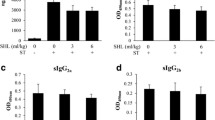
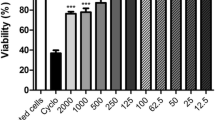
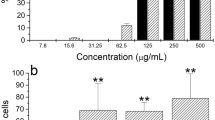
Although several studies have shown that scorpion venoms cause a systemic inflammatory response syndrome, little is known about the contribution of the hematopoietic organs. The aim of this study was to investigate the effect of Androctonus australis hector venom on the bone marrow and on local inflammatory mediators, in concordance with the systemic inflammatory reaction elicited in mice. The consequences of a direct interaction of venom with murine bone marrow cells were also assessed by in vitro study. Obtained results showed that the early systemic neutrophilia correlated with a rapid granulocyte mobilization. This response was followed by an accelerated granulopoiesis that was supported by TNF-α and IL-6 signals. In vitro data revealed that the venom exerted a proliferative effect on murine hematopoietic cells and stimulated their differentiation towards granulocyte lineage mainly through cytokine secretion. In conclusion, this study indicated that the bone marrow rapidly exerts its activity in response to the experimental envenomation via the granulopoiesis process and inflammatory mediators in concert with the development of a systemic response. The ability of venom to directly switch steady-state granulopoiesis to an accelerated state in vitro could aggravate the disturbance caused by the venom. Better understanding of the mechanisms involved may lead to the emergence of new targets to avoid cell spreading and accumulation by acting on the very early stage of the systemic inflammatory response.






Similar content being viewed by others
References
Furze, R.C., and S.M. Rankin. 2008. Neutrophil mobilization and clearance in the bone marrow. Immunology 125(3): 281–288.
Jiang, D., and H. Schwarz. 2010. Regulation of granulocyte and macrophage populations of murine bone marrow cells by G-CSF and CD137 protein. PLoS One 5(12): e15565.
Pfeifer, R., T. Tschernig, P. Lichte, D. Dombroski, P. Kobbe, and H.C. Pape. 2013. MALP-2 pre-treatment modulates systemic inflammation in hemorrhagic shock. Journal of Inflammation 10(1): 1.
Min, S.H., M.H. Kang, J.H. Sur, and H.M. Park. 2014. Staphylococcus pseudintermedius infection associated with nodular skin lesions and systemic inflammatory response syndrome in a dog. The Canadian Veterinary Journal 55(5): 480.
Meki, A.R., and Z.M. Mohey El-Dean. 1998. Serum interleukin-1β, interleukin-6, nitric oxide and α1-antitrypsin in scorpion envenomed children. Toxicon 36(12): 1851–9.
D’Suze, G., S. Moncada, C. González, C. Sevcik, V. Aguilar, and A. Alagón. 2003. Relationship between plasmatic levels of various Lung immunoreactivity and airway inflammation cytokines, tumour necrosis factor, enzymes, glucose and venom concentration following Tityus scorpion sting. Toxicon 41(3): 367–75.
Ismail, M. 1995. The scorpion envenoming syndrome. Toxicon 33(7): 825–858.
Adi-Bessalem, S., D. Hammoudi-Triki, and F. Laraba-Djebari. 2013. Scorpion venom interactions with the immune system. In Toxinology, ed. P. Gopalakrishnakone, 1–18. Dordrecht: Springer Netherlands.
Magalhães, M.M., M.E. Pereira, C.F. Amaral, N.A. Rezende, D. Campolina, F. Bucaretchi, R.T. Gazzinelli, and J.R Cunha-Melo. Serum levels of cytokines in patients envenomed by Tityus serrulatus scorpion sting. Toxicon 37(8): 1155–1164.
Fukuhara, Y.D., M.L. Reis, R. Dellalibera-Joviliano, F.Q. Cunha, and E.A. Donadi. 2003. Increased plasma levels of IL-1beta, IL-6, IL-8, IL-10 and TNF-alpha in patients moderately or severely envenomed by Tityus serrulatus scorpion sting. Toxicon 41(1): 49–55.
Hammoudi-Triki, D., E. Ferquel, A. Robbe-Vincent, C. Bon, V. Choumet, and F. Laraba-Djebari. 2004. Epidemiological data, clinical admission gradation and biological quantification by ELISA of scorpion envenomations in Algeria: effect of immunotherapy. Transactions of the Royal Society of Tropical Medicine and Hygiene 98(4): 240–250.
Adi-Bessalem, S., D. Hammoudi-Triki, and F. Laraba-Djebari. 2008. Pathophysiological effects of Androctonus australis hector scorpion venom: tissue damages and inflammatory response. Experimental and Toxicologic Pathology 60(4): 373–380.
Raouraoua-Boukari, R., S. Sami-Merah, D. Hammoudi-Triki, M.F. Martin-Eauclaire, and F. Laraba-Djebari. 2012. Immunomodulation of the inflammatory response induced by Androctonus australis hector neurotoxins: biomarker interactions. Neuroimmunomodulation 19(2): 103–110.
D’Suze, G., V. Salazar, P. Díaz, C. Sevcik, H. Azpurua, and N. Bracho. 2004. Histopathological changes and inflammatory response induced by Tityus discrepans scorpion venom in rams. Toxicon 44(8): 851–860.
Sami-Merah, S., D. Hammoudi-Triki, M.F. Martin-Eauclaire, and F. Laraba-Djebari. 2008. Combination of two antibody fragments F(ab’)2/Fab: an alternative for scorpion envenoming treatment. International Immunopharmacology 8(10): 1386–1394.
Taibi-Djennah, Z., and F. Laraba-Djebari. 2015. Effect of cytokine antibodies in the immunomodulation of inflammatory response and metabolic disorders induced by scorpion venom. International Immunopharmacology 27(1): 122–129.
Takizawa, H., S. Boettcher, and M.G. Manz. 2012. Demand-adapted regulation of early hematopoiesis in infection and inflammation. Blood 119(13): 2991–3002.
Yáñez, A., H.S. Goodridge, D. Gozalbo, and M.L. Gil. 2013. TLRs control hematopoiesis during infection: highlights. European Journal of Immunology 43(10): 2526–2533.
Laraba-Djebari, F., and D. Hammoudi. 1998. Use of toxic fraction isolated from Algerian Androctonus australis hector scorpion venom for the assessment of anti-venom serum. Archives de l’ Institut Pasteur d’Algerie 62: 254–266.
Bradford, M.M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Analytical Biochemistry 72(1-2): 248–254.
Bradley, P.P., D.A. Priebat, R.D. Christensen, and G. Fricchione. 1982. Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. Journal of Investigative Dermatology 78(3): 206–209.
Van Oosterhout, A.J., I. Van Ark, G. Hofman, H.J. Van Der Linde, D. Fattah, and F.P. Nijkamp. 1996. Role of interleukin-5 and substance P in development of airway hyperreactivity to histamine in guinea-pigs. The European Respiratory Journal 9(3): 493–499.
Dobson, K.R., L. Reading, M. Haberey, X. Marine, and A. Scutt. 1999. Centrifugal isolation of bone marrow from bone: an improved method for the recovery and quantitation of bone marrow osteoprogenitor cells from rat tibiae and femurae. Calcified Tissue International 65(5): 411–413.
Eash, K.J., A.M. Greenbaum, P.K. Gopalan, and D.C. Link. 2010. CXCR2 and CXCR4 antagonistically regulate neutrophil trafficking from murine bone marrow. Journal of Clinical Investigation 120(7): 2423–2431.
Griess, P. 1879. Griess reagent: a solution of sulphanilic acid and A-Naphthylamine in acetic acid which gives a pink colour on reaction with the solution obtained after decomposition of nitrosyl complexes. Chemische Berichte 12: 427.
Borghesi, L. 2014. Hematopoiesis in steady-state versus stress: self-renewal, lineage fate choice, and the conversion of danger signals into cytokine signals in hematopoietic stem cells. The Journal of Immunology 193(5): 2053–2058.
Ogata, K., C. Satoh, M. Tachibana, H. Hyodo, H. Tamura, K. Dan, and T. Tsuji. 2005. Identification and hematopoietic potential of CD45– clonal cells with very immature phenotype (CD45–CD34–CD38–Lin–) in patients with myelodysplastic Syndromes. Stem Cells 23(5): 619–630.
Pirih, F.Q., M.N. Michalski, S.W. Cho, A.J. Koh, J.E. Berry, E. Ghaname, and L.K. McCauley. 2010. Parathyroid hormone mediates hematopoietic cell expansion through Interleukin-6. PLoS ONE 5(10): e13657.
Kondo, M., A.J. Wagers, M.G. Manz, S.S. Prohaska, D.C. Scherer, G.F. Beilhack, J.A. Shizuru, and I.L. Weissman. 2003. Biology of hematopoietic stem cells and progenitors: implications for clinical application. Annual Review of Immunology 21(1): 759–806.
Keyhani, E., M.A. Zarei, and T. Lashgarblooki-Livani. 1999. Kinetics of peroxidases in guinea pig bone marrow under immunostimulation. FEBS Letters 452(3): 233–236.
Rodríguez, A.E., Y. Rodríguez, F. Yusef, E. Trejo, and M. Rossini. 2015. Hematological alterations induced by Tityus discrepans scorpion venom in mice. Revista de la Facultad de Ciencias Veterinarias 56(1): 9–16.
Coelho, M.F., A.C. Pessini, A.M. Coelho, V.S. Pinho, D.G. Souza, E.C. Arantes, M.M. Teixeira, and A.L. Teixeira. 2007. Platelet activating factor receptors drive CXC production, neutrophil influx and edema formation in lungs of mice injected with Tityus serrulatus venom. Toxicon 50(3): 420–427.
Bekkari, N., M.F. Martin-Eauclaire, and F. Laraba-Djebari. 2015. Complement system and immunological mediators: their involvements in the induced inflammatory process by Androctonus australis hector venom and its toxic components. Experimental and Toxicologic Pathology 67(7): 389–397.
Chaïr-Yousfi, I., F. Laraba-Djebari, and D. Hammoudi-Triki. 2015. Androctonus australis hector venom contributes to the interaction between neuropeptides and mast cells in pulmonary hyperresponsiveness. International Immunopharmacology 25(1): 19–29.
Lamraoui, A., S. Adi-Bessalem, and F. Laraba-Djebari. 2014. Modulation of tissue inflammatory response by histamine receptors in scorpion envenomation pathogenesis: involvement of H4 Receptor. Inflammation 37(5): 1689–1704.
Manz, M.G., and S. Boettcher. 2014. Emergency granulopoiesis. Nature Reviews Immunology 14(5): 302–314.
Scumpia, P.O., K.M. Kelly-Scumpia, M.J. Delano, J.S. Weinstein, A.G. Cuenca, S. Al-Quran, and L.L. Moldawer. 2010. Cutting edge: bacterial infection induces hematopoietic stem and progenitor cell expansion in the absence of TLR signaling. The Journal of Immunology 184(5): 2247–2251.
Boettcher, S., P. Ziegler, M.A. Schmid, H. Takizawa, N. Van Rooijen, M. Kopf, M. Heikenwalder, and M.G. Manz. 2012. Cutting edge: LPS-induced emergency myelopoiesis depends on TLR4-expressing nonhematopoietic cells. The Journal of Immunology 188(12): 5824–5828.
Borges, C.M., M.R. Silveira, M. Aparecida, C.L. Beker, L. Freire-Maia, and M.M. Teixeira. 2000. Scorpion venom-induced neutrophilia is inhibited by a PAF receptor antagonist in the rat. Journal of Leukocyte Biology 67(4): 515–519.
Fialho, E.M.S., M.C.G. Maciel, A.C.B. Silva, A.S. Reis, A.K.M. Assunção, T.S. Fortes, L.A. Silva, R.N.M. Guerra, F.H. Kwasniewski, and F.R.F. Nascimento. 2011. Immune cells recruitment and activation by Tityus serrulatus scorpion venom. Toxicon 58(6): 480–485.
Cain, D.W., P.B. Snowden, G.D. Sempowski, and G. Kelsoe. 2011. Inflammation triggers emergency granulopoiesis through a density-dependent feedback mechanism. PLoS One 6(5): e19957.
King, K.Y., and M.A. Goodell. 2011. Inflammatory modulation of HSCs: viewing the HSC as a foundation for the immune response. Nature Reviews Immunology 11(10): 685–692.
Kwak, H.J., P. Liu, B. Bajrami, Y. Xu, S.Y. Park, C. Nombela-Arrieta, S. Mondal, Y. Sun, H. Zhu, L. Chai, L.E. Silberstein, T. Cheng, and H.R. Luo. 2015. Myeloid cell-derived reactive oxygen species externally regulate the proliferation of myeloid progenitors in emergency granulopoiesis. Immunity 42(1): 159–171.
Baldridge, M.T., K.Y. King, and M.A. Goodell. 2011. Inflammatory signals regulate hematopoietic stem cells. Trends in Immunology 32(2): 57–65.
Saidi, H., S. Adi-Bessalem, D. Hammoudi-Triki, and F. Laraba-Djebari. 2013. Effects of atropine and propranolol on lung inflammation in experimental envenomation: comparison of two buthidae venoms. Journal of Venomous Animals and Toxins Including Tropical Diseases 19(1): 1.
Iversen, P.O. 1997. Blood flow to the hematopoietic bone marrow. Acta Physiologica Scandinavica 159(4): 269–276.
Petricevich, V.L., and C.F. Peña. 2002. The dynamics of cytokine and nitric oxide secretion in mice injected with Tityus serrulatus scorpion venom. Mediators of Inflammation 11(3): 173–180.
Wang, C., M. Zhou, T. Li, Y. Wang, B. Xing, T. Kong, and W. Dong. 2015. Effects of scorpion venom peptide B5 on hematopoietic recovery in irradiated mice and the primary mechanisms. Scientific Reports 5: 15363.
Qiu, Y., L. Jiang, C. Wang, Y. Wang, T. Li, B. Xing, M. Zhou, T. Kong, and W. Dong. 2013. Scorpion venom peptide SPVII promotes irradiated cells proliferation and increases the expression of the IL-3 receptor. Cell & Bioscience 3(1): 1.
Acknowledgements
The authors want to acknowledge Dr. Manuchehr Abedi-Valugerdi from the Department of Laboratory Medicine (Karolinska Institutet, Stockholm, Sweden) for his help with flow cytometry analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
All animal experiments were planned and performed in accordance with guidelines for the care of laboratory animals and approved by the European Community Council Directive (86/609/EEC).
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kaddache, A., Hassan, M., Laraba-Djebari, F. et al. Switch of Steady-State to an Accelerated Granulopoiesis in Response to Androctonus australis hector Venom. Inflammation 40, 871–883 (2017). https://doi.org/10.1007/s10753-017-0532-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-017-0532-6