Abstract
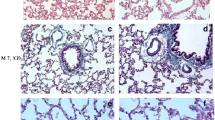
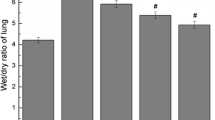
We aimed to investigate the preventive effect of Infliximab (IFX), a tumor necrosis factor (TNF)-α inhibitor, on bleomycin (BLC)-induced lung fibrosis in rats. Rats were assigned into four groups as follows: I—BLC group, a single intra-tracheal BLC (2.5 mg/kg) was installed; II—control group, a single intra-tracheal saline was installed; III—IFX + BLC group, a single-dose IFX (7 mg/kg) was administered intraperitoneally (i.p.), 72 h before the intra-tracheal BLC installation; IV—IFX group, IFX (7 mg/kg) was administered alone i.p. on the same day with IFX + BLC group. All animals were sacrificed on the 14th day of BLC installation. Levels of tumor necrosis factor (TNF)-α, transforming growth factor (TGF)-β, interleukin (IL)-6, periostin, YKL-40, nitric oxide (NO) in rat serum were measured, as well as, myeloperoxidase (MPO), superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx) activity, and reduced glutathione (GSH), hydroxyproline, malondialdehyde (MDA) content in lung homogenates. Lung tissues were stained with hematoxylin and eosin (H&E) for quantitative histological evaluation. The inducible nitric oxide synthase (iNOS) expression and cell apoptosis in the lung tissues were determined quantitatively by immunohistochemical staining (INOS) and by TUNNEL staining, respectively. BLC installation worsened antioxidant status (such as SOD, CAT, GPx, GSH, MPO), while it increased the serum TNF-α, TGF-β, IL-6, periostin, YKL-40, and lipid peroxidation, and collagen deposition, measured by MDA and hydroxyproline, respectively. IFX pretreatment improved antioxidant status as well as BLC-induced lung pathological changes, while it decreased the TNF-α, TGF-β, IL-6, periostin, YKL-40, lipid peroxidation and collagen deposition. Finally, histological, immunohistochemical, and TUNNEL evidence also supported the ability of IFX to prevent BLC-induced lung fibrosis. The results of the present study indicate that IFX pretreatment can attenuate BLC-induced pulmonary fibrosis.






Similar content being viewed by others
References
Mercer, P.F., K. Abbott-Banner, I.M. Adcock, and R.G. Knowles. 2015. Translational models of lung disease. Clinical science (London, England : 1979) 128: 235–256.
Daba, M.H., K.E. El-Tahir, M.N. Al-Arifi, and O.A. Gubara. 2004. Drug-induced pulmonary fibrosis. Saudi Medical Journal 25: 700–706.
Sabry, M.M., S.A.-E. Elkalawy, R.K.E.-D. Abo-Elnour, and D.F. Abd-El-Maksod. 2014. Histolgical and immunohistochemical study on the effect of stem cell therapy on bleomycin induced pulmonary fibrosis in albino rat. International Journal of Stem Cells 7: 33–42.
Bargagli, E., C. Olivieri, D. Bennett, A. Prasse, J. Muller-Quernheim, and P. Rottoli. 2009. Oxidative stress in the pathogenesis of diffuse lung diseases: a review. Respiratory Medicine 103: 1245–1256.
Ahluwalia, N., B.S. Shea, and A.M. Tager. 2014. New therapeutic targets in idiopathic pulmonary fibrosis. Aiming to rein in runaway Wound-healing responses. American Journal of Respiratory and Critical Care Medicine 190: 867–878.
Black, R.A., C.T. Rauch, C.J. Kozlosky, J.J. Peschon, J.L. Slack, M.F. Wolfson, B.J. Castner, et al. 1997. A metalloproteinase disintegrin that releases tumour-necrosis factor-alpha from cells. Nature 385: 729–733.
Piguet, P.F., C. Ribaux, V. Karpuz, G.E. Grau, and Y. Kapanci. 1993. Expression and localization of tumor necrosis factor-alpha and its mRNA in idiopathic pulmonary fibrosis. The American Journal of Pathology 143: 651–655.
Piguet, P.F., M.A. Collart, G.E. Grau, A.P. Sappino, and P. Vassalli. 1990. Requirement of tumour necrosis factor for development of silica-induced pulmonary fibrosis. Nature 344: 245–247.
Chizzolini, C., Y. Parel, C. De Luca, A. Tyndall, A. Akesson, A. Scheja, and J.-M. Dayer. 2003. Systemic sclerosis Th2 cells inhibit collagen production by dermal fibroblasts via membrane-associated tumor necrosis factor alpha. Arthritis and Rheumatism 48: 2593–2604.
Redente, E.F., R.C. Keith, W. Janssen, P.M. Henson, L.A. Ortiz, G.P. Downey, D.L. Bratton, and D.W.H. Riches. 2014. Tumor necrosis factor-α accelerates the resolution of established pulmonary fibrosis in mice by targeting profibrotic lung macrophages. American Journal of Respiratory Cell and Molecular Biology 50: 825–837.
Tracey, D., L. Klareskog, E.H. Sasso, J.G. Salfeld, and P.P. Tak. 2008. Tumor necrosis factor antagonist mechanisms of action: a comprehensive review. Pharmacology & Therapeutics 117: 244–279.
Di Sabatino, A., R. Ciccocioppo, L. Benazzato, G.C. Sturniolo, and G.R. Corazza. 2004. Infliximab downregulates basic fibroblast growth factor and vascular endothelial growth factor in Crohn’s disease patients. Alimentary Pharmacology & Therapeutics 19: 1019–1024.
Bargagli, E., M. Galeazzi, and P. Rottoli. 2004. Infliximab treatment in a patient with rheumatoid arthritis and pulmonary fibrosis. The European Respiratory Journal: Official Journal of the European Society for Clinical Respiratory Physiology 24: 708.
Antoniou, K.M., M. Mamoulaki, K. Malagari, H.D. Kritikos, D. Bouros, N.M. Siafakas, and D.T. Boumpas. 2007. Infliximab therapy in pulmonary fibrosis associated with collagen vascular disease. Clinical and Experimental Rheumatology 25: 23–28.
Ostor, A.J.K., A.J. Crisp, M.F. Somerville, and D.G.I. Scott. 2004. Fatal exacerbation of rheumatoid arthritis associated fibrosing alveolitis in patients given infliximab. BMJ (Clinical Research ed.) 329: 1266.
Chatterjee, S. 2004. Severe interstitial pneumonitis associated with infliximab therapy. Scandinavian Journal of Rheumatology 33: 276–277.
Guide for the Care and Use of Laboratory Animals. 1996. Guide for the care and use of laboratory animals. Washington (DC): National Academies Press (US).
Teixeira, K.C., F.S. Soares, L.G.C. Rocha, P.C.L. Silveira, L.A. Silva, S.S. Valença, F. Dal Pizzol, E.L. Streck, and R.A. Pinho. 2008. Attenuation of bleomycin-induced lung injury and oxidative stress by N-acetylcysteine plus deferoxamine. Pulmonary Pharmacology & Therapeutics 21: 309–316.
Akgedik, R., Ş. Akgedik, H. Karamanlı, S. Uysal, B. Bozkurt, D. Ozol, F. Armutçu, and Z. Yıldırım. 2012. Effect of resveratrol on treatment of bleomycin-induced pulmonary fibrosis in rats. Inflammation 35: 1732–1741.
Gonçalves, D.C., R.C. Evangelista, R.R. da Silva, M.J.S. Santos, F.S. Silva, K.S. Aragão, G.A.C. Brito, H.B.M. Lucena, R.C. Leitão, and R.B. Oriá. 2014. Infliximab attenuates inflammatory osteolysis in a model of periodontitis in Wistar rats. Experimental Biology and Medicine (Maywood, N.J.) 239: 442–453.
Chen, C.-Y., W.-H. Peng, L.-C. Wu, C.-C. Wu, and S.-L. Hsu. 2010. Luteolin ameliorates experimental lung fibrosis both in vivo and in vitro: implications for therapy of lung fibrosis. Journal of Agricultural and Food Chemistry 58: 11653–11661.
Hübner, R.-H., W. Gitter, N. Eddine El Mokhtari, M. Mathiak, M. Both, H. Bolte, S. Freitag-Wolf, and B. Bewig. 2008. Standardized quantification of pulmonary fibrosis in histological samples. BioTechniques 44: 507–517.
Hsu, S.M., L. Raine, and H. Fanger. 1981. Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. The Journal of Histochemistry and Cytochemistry: Official Journal of the Histochemistry Society 29: 577–580.
Kanter, M., S.H. Sahin, U.N. Basaran, S. Ayvaz, B. Aksu, M. Erboga, and A. Colak. 2015. The effect of methylene blue treatment on aspiration pneumonia. The Journal of Surgical Research 193: 909–919.
Sun, Y., L.W. Oberley, and Y. Li. 1988. A simple method for clinical assay of superoxide dismutase. Clinical Chemistry 34: 497–500.
Luck, H. 1963. A spectrophotometric method for the estimation of catalase. In Methods of enzymatic analysis, ed. H.U. Bergmeyer, 886–887. New York: Academic.
Paglia, D.E., and W.N. Valentine. 1967. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. The Journal of Laboratory and Clinical Medicine 70: 158–169.
Ellman, G.L. 1959. Tissue sulfhydryl groups. Archives of Biochemistry and Biophysics 82: 70–77.
Wei, H., and K. Frenkel. 1993. Relationship of oxidative events and DNA oxidation in SENCAR mice to in vivo promoting activity of phorbol ester-type tumor promoters. Carcinogenesis 14: 1195–1201.
Draper, H.H., and M. Hadley. 1990. Malondialdehyde determination as index of lipid peroxidation. Methods in Enzymology 186: 421–431.
Cortas, N.K., and N.W. Wakid. 1990. Determination of inorganic nitrate in serum and urine by a kinetic cadmium-reduction method. Clinical Chemistry 36: 1440–1443.
Brown, S., M. Worsfold, and C. Sharp. 2001. Microplate assay for the measurement of hydroxyproline in acid-hydrolyzed tissue samples. BioTechniques 30: 38–40.
Mouratis, M.A., and V. Aidinis. 2011. Modeling pulmonary fibrosis with bleomycin. Current Opinion in Pulmonary Medicine 17: 355–361.
Rossi, R.E., I. Parisi, E.J. Despott, A.K. Burroughs, J. O’Beirne, D. Conte, M.I. Hamilton, and C.D. Murray. 2014. Anti-tumour necrosis factor agent and liver injury: literature review, recommendations for management. World Journal of Gastroenterology: WJG 20: 17352–17359.
Khasnis, A.A., and L.H. Calabrese. 2010. Tumor necrosis factor inhibitors and lung disease: a paradox of efficacy and risk. Seminars in Arthritis and Rheumatism 40: 147–163.
Jenkins, G., and A. Goodwin. 2014. Novel approaches to pulmonary fibrosis. Clinical Medicine (London, England) 14(Suppl 6): s45–49.
Yara, S., K. Kawakami, N. Kudeken, M. Tohyama, K. Teruya, T. Chinen, A. Awaya, and A. Saito. 2001. FTS reduces bleomycin-induced cytokine and chemokine production and inhibits pulmonary fibrosis in mice. Clinical and Experimental Immunology 124: 77–85.
Zhu, B., A.-Q. Ma, L. Yang, and X.-M. Dang. 2013. Atorvastatin attenuates bleomycin-induced pulmonary fibrosis via suppressing iNOS expression and the CTGF (CCN2)/ERK signaling pathway. International Journal of Molecular Sciences 14: 24476–24491.
Williamson, J.D., L.R. Sadofsky, and S.P. Hart. 2015. The pathogenesis of bleomycin-induced lung injury in animals and its applicability to human idiopathic pulmonary fibrosis. Experimental Lung Research 41: 57–73.
Goldblum, S.E., and W.L. Sun. 1990. Tumor necrosis factor-alpha augments pulmonary arterial transendothelial albumin flux in vitro. The American Journal of Physiology 258: L57–67.
Ortiz, L.A., J. Lasky, R.F. Hamilton, A. Holian, G.W. Hoyle, W. Banks, J.J. Peschon, A.R. Brody, G. Lungarella, and M. Friedman. 1998. Expression of TNF and the necessity of TNF receptors in bleomycin-induced lung injury in mice. Experimental Lung Research 24: 721–743.
Carré, P., and P. Léophonte. 1993. Cytokines and pulmonary fibroses. Revue des Maladies Respiratoires 10: 193–207.
Underwood, D.C., R.R. Osborn, S. Bochnowicz, E.F. Webb, D.J. Rieman, J.C. Lee, A.M. Romanic, J.L. Adams, D.W. Hay, and D.E. Griswold. 2000. SB 239063, a p38 MAPK inhibitor, reduces neutrophilia, inflammatory cytokines, MMP-9, and fibrosis in lung. American Journal of Physiology. Lung Cellular and Molecular Physiology 279: L895–902.
Uchida, M., H. Shiraishi, S. Ohta, K. Arima, K. Taniguchi, S. Suzuki, M. Okamoto, et al. 2012. Periostin, a matricellular protein, plays a role in the induction of chemokines in pulmonary fibrosis. American Journal of Respiratory Cell and Molecular Biology 46: 677–686.
Korthagen, N.M., C.H.M. van Moorsel, N.P. Barlo, H.J.T. Ruven, A. Kruit, M. Heron, J.M.M. van den Bosch, and J.C. Grutters. 2011. Serum and BALF YKL-40 levels are predictors of survival in idiopathic pulmonary fibrosis. Respiratory Medicine 105: 106–113.
Furuhashi, K., T. Suda, Y. Nakamura, N. Inui, D. Hashimoto, S. Miwa, H. Hayakawa, et al. 2010. Increased expression of YKL-40, a chitinase-like protein, in serum and lung of patients with idiopathic pulmonary fibrosis. Respiratory Medicine 104: 1204–1210.
Oikonomou, N., V. Harokopos, J. Zalevsky, C. Valavanis, A. Kotanidou, D.E. Szymkowski, G. Kollias, and V. Aidinis. 2006. Soluble TNF mediates the transition from pulmonary inflammation to fibrosis. PLoS One 1: e108.
Theiss, A.L., J.G. Simmons, C. Jobin, and P.K. Lund. 2005. Tumor necrosis factor (TNF) alpha increases collagen accumulation and proliferation in intestinal myofibroblasts via TNF receptor 2. The Journal of Biological Chemistry 280: 36099–36109.
Piguet, P.F., M.A. Collart, G.E. Grau, Y. Kapanci, and P. Vassalli. 1989. Tumor necrosis factor/cachectin plays a key role in bleomycin-induced pneumopathy and fibrosis. The Journal of Experimental Medicine 170: 655–663.
Mauviel, A., M. Daireaux, F. Rédini, P. Galera, G. Loyau, and J.P. Pujol. 1988. Tumor necrosis factor inhibits collagen and fibronectin synthesis in human dermal fibroblasts. FEBS Letters 236: 47–52.
Fujita, M., J.M. Shannon, O. Morikawa, J. Gauldie, N. Hara, and R.J. Mason. 2003. Overexpression of tumor necrosis factor-alpha diminishes pulmonary fibrosis induced by bleomycin or transforming growth factor-beta. American Journal of Respiratory and Critical Care Medicine 29: 669–676.
Piguet, P.F., and C. Vesin. 1994. Treatment by human recombinant soluble TNF receptor of pulmonary fibrosis induced by bleomycin or silica in mice. The European Respiratory Journal: Official Journal of the European Society for Clinical Respiratory Physiology 7: 515–518.
Bargagli, E., M. Galeazzi, F. Bellisai, L. Volterrani, and P. Rottoli. 2008. Infliximab treatment in a patient with systemic sclerosis associated with lung fibrosis and pulmonary hypertension. Respiration 75: 346–349.
Cairns, A.P., and A.J. Taggart. 2002. Anti-tumour necrosis factor therapy for severe inflammatory arthritis: two years of experience in Northern Ireland. The Ulster Medical Journal 71: 101–105.
Ledingham, J., C. Deighton, British Society for Rheumatology Standards, Guidelines and Audit Working Group. 2005. Update on the British Society for Rheumatology guidelines for prescribing TNFalpha blockers in adults with rheumatoid arthritis (update of previous guidelines of April 2001). Rheumatology (Oxford, England). Oxford University Press.
Saag, K.G., G.G. Teng, N.M. Patkar, J. Anuntiyo, C. Finney, J.R. Curtis, H.E. Paulus, et al. 2008. American College of Rheumatology 2008 recommendations for the use of nonbiologic and biologic disease-modifying antirheumatic drugs in rheumatoid arthritis. Arthritis and Rheumatism 59: 762–784. Wiley Subscription Services, Inc., A Wiley Company.
Conflict of Interest
The authors have no conflict of interest to declare. Funding for this study was received from “Namik Kemal University, Scientific Research Projects Unit”, Project No: NKUBAP.00.20.AR.1417
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Altintas, N., Erboga, M., Aktas, C. et al. Protective Effect of Infliximab, a Tumor Necrosis Factor-Alfa Inhibitor, on Bleomycin-Induced Lung Fibrosis in Rats. Inflammation 39, 65–78 (2016). https://doi.org/10.1007/s10753-015-0224-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-015-0224-z