Abstract
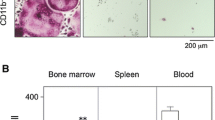
This study analyzes the effect of interleukin-15 (IL-15) on osteoclast formation using a coculture of mouse osteoblasts and bone marrow cells (BMCs) stimulated with prostaglandin E2 (PGE2), which both have important role in rheumatoid arthritis (RA) and periodontal disease (PD). BMCs isolate lacking T (BMT−) or NK (BMNK−) cells, BMCs with no cells removed (BMT+NK+), purified NK cells, and purified T cells were each cocultured with osteoblasts in the presence or absence of PGE2 and/or IL-15. The number of both osteoclasts and osteoblasts was decreased by IL-15 in a dose-dependent manner in BMT+NK+, BMT−. However, the reductions were improved in BMNK−. The expression of caspase3 in osteoblasts cocultured with NK cells was increased in a dose-dependent manner by IL-15. IL-15 stimulates apoptosis of osteoblasts via activation of NK cells. Since osteoblasts have an important role in bone formation, IL-15 may be an inflammatory bone destructive factor in RA and PD.








Similar content being viewed by others
References
Matei, I., and L. Matei. 2002. Cytokine patterns and pathogenicity in autoimmune diseases. Romanian Journal of Internal Medicine 40: 27–41.
Reinhardt, R.A., M.P. Masada, W.B. Kaldahl, L.M. DuBois, K.S. Kornman, J.I. Choi, et al. 1993. Gingival fluid IL-1 and IL-6 levels in refractory periodontitis. Journal of Clinical Periodontology 20: 225–231.
Rossomando, E.F., J.E. Kennedy, and J. Hadjimichael. 1990. Tumour necrosis factor alpha in gingival crevicular fluid as a possible indicator of periodontal disease in humans. Archives of Oral Biology 35: 431–434.
Katagiri, T., and N. Takahashi. 2002. Regulatory mechanisms of osteoblast and osteoclast differentiation. Oral Diseases 8: 147–159.
McInnes, I.B., J. al-Mughales, M. Field, B.P. Leung, F.P. Huang, R. Dixon, et al. 1996. The role of interleukin-15 in T-cell migration and activation in rheumatoid arthritis. Nature Medicine 2: 175–182.
Tymkiw, K.D., D.H. Thunell, G.K. Johnson, S. Joly, K.K. Burnell, J.E. Cavanaugh, et al. 2011. Influence of smoking on gingival crevicular fluid cytokines in severe chronic periodontitis. Journal of Clinical Periodontology 38: 219–228.
Thurkow, E.W., I.M. van der Heijden, F.C. Breedveld, T.J. Smeets, M.R. Daha, P.M. Kluin, et al. 1997. Increased expression of IL-15 in the synovium of patients with rheumatoid arthritis compared with patients with Yersinia-induced arthritis and osteoarthritis. The Journal of Pathology 181: 444–450.
Burton, J.D., R.N. Bamford, C. Peters, A.J. Grant, G. Kurys, C.K. Goldman, et al. 1994. A lymphokine, provisionally designated interleukin T and produced by a human adult T-cell leukemia line, stimulates T-cell proliferation and the induction of lymphokine-activated killer cells. Proceedings of the National Academy of Sciences of the United States of America 91: 4935–4939.
Grabstein, K.H., J. Eisenman, K. Shanebeck, C. Rauch, S. Srinivasan, V. Fung, et al. 1994. Cloning of a T cell growth factor that interacts with the beta chain of the interleukin-2 receptor. Science 264: 965–968.
Carson, W.E., J.G. Giri, M.J. Lindemann, M.L. Linett, M. Ahdieh, R. Paxton, et al. 1994. Interleukin (IL) 15 is a novel cytokine that activates human natural killer cells via components of the IL-2 receptor. The Journal of Experimental Medicine 180: 1395–1403.
Armitage, R.J., B.M. Macduff, J. Eisenman, R. Paxton, and K.H. Grabstein. 1995. IL-15 has stimulatory activity for the induction of B cell proliferation and differentiation. The Journal of Immunology 154: 483–490.
Mitani, A., H. Nishimura, K. Hirose, J. Washizu, Y. Kimura, S. Tanaka, et al. 1999. Interleukin-15 production at the early stage after oral infection with Listeria monocytogenes in mice. Immunology 97: 92–99.
Buduneli, E., F. Genel, G. Atilla, and N. Kutukculer. 2003. Evaluation of p53, bcl-2, and interleukin-15 levels in gingival crevicular fluid of cyclosporin A-treated patients. The Journal of Periodontology 74: 506–511.
Andersson, A.K., M. Feldmann, and F.M. Brennan. 2008. Neutralizing IL-21 and IL-15 inhibits pro-inflammatory cytokine production in rheumatoid arthritis. Scandinavian Journal of Immunology 68: 103–111.
Ogata, Y., A. Kukita, T. Kukita, M. Komine, A. Miyahara, S. Miyazaki, et al. 1999. A novel role of IL-15 in the development of osteoclasts: inability to replace its activity with IL-2. The Journal of Immunology 162: 2754–2760.
Ferrari-Lacraz, S., E. Zanelli, M. Neuberg, E. Donskoy, Y.S. Kim, X.X. Zheng, et al. 2004. Targeting IL-15 receptor-bearing cells with an antagonist mutant IL-15/Fc protein prevents disease development and progression in murine collagen-induced arthritis. The Journal of Immunology 173: 5818–5826.
Miranda-Carus, M.E., M. Benito-Miguel, A. Balsa, T. Cobo-Ibanez, C. Perez de Ayala, D. Pascual-Salcedo, et al. 2006. Peripheral blood T lymphocytes from patients with early rheumatoid arthritis express RANKL and interleukin-15 on the cell surface and promote osteoclastogenesis in autologous monocytes. Arthritis and Rheumatism 54: 1151–1164.
Djaafar, S., D.D. Pierroz, R. Chicheportiche, X.X. Zheng, S.L. Ferrari, and S. Ferrari-Lacraz. 2010. Inhibition of T cell-dependent and RANKL-dependent osteoclastogenic processes associated with high levels of bone mass in interleukin-15 receptor-deficient mice. Arthritis and Rheumatism 62: 3300–3310.
Suda, T., E. Jimi, I. Nakamura, and N. Takahashi. 1997. Role of 1 alpha,25-dihydroxyvitamin D3 in osteoclast differentiation and function. Methods in Enzymology 282: 223–235.
Noguchi, K., M. Yanai, M. Shitashige, T. Nishihara, and I. Ishikawa. 2000. Cyclooxygenase-2-dependent prostaglandin production by peripheral blood monocytes stimulated with lipopolysaccharides isolated from periodontopathogenic bacteria. The Journal of Periodontology 71: 1575–1582.
Yucel-Lindberg, T., H. Ahola, J. Carlstedt-Duke, and T. Modeer. 1999. Involvement of tyrosine kinases on cyclooxygenase expression and prostaglandin E2 production in human gingival fibroblasts stimulated with interleukin-1beta and epidermal growth factor. Biochemical and Biophysical Research Communications 257: 528–532.
Hughes, F.J., L.D. Buttery, M.V. Hukkanen, A. O'Donnell, J. Maclouf, and J.M. Polak. 1999. Cytokine-induced prostaglandin E2 synthesis and cyclooxygenase-2 activity are regulated both by a nitric oxide-dependent and -independent mechanism in rat osteoblasts in vitro. The Journal of Biological Chemistry 274: 1776–1782.
Yasuda, H., N. Shima, N. Nakagawa, K. Yamaguchi, M. Kinosaki, S. Mochizuki, et al. 1998. Osteoclast differentiation factor is a ligand for osteoprotegerin/osteoclastogenesis-inhibitory factor and is identical to TRANCE/RANKL. Proceedings of the National Academy of Sciences of the United States of America 95: 3597–3602.
Nagata, T., K. Kaho, S. Nishikawa, H. Shinohara, Y. Wakano, and H. Ishida. 1994. Effect of prostaglandin E2 on mineralization of bone nodules formed by fetal rat calvarial cells. Calcified Tissue International 55: 451–457.
Jimi, E., T. Ikebe, N. Takahashi, M. Hirata, T. Suda, and T. Koga. 1996. Interleukin-1 alpha activates an NF-kappaB-like factor in osteoclast-like cells. The Journal of Biological Chemistry 271: 4605–4608.
Fuster, G., V. Almendro, C.C. Fontes-Oliveira, M. Toledo, P. Costelli, S. Busquets, et al. 2011. Interleukin-15 affects differentiation and apoptosis in adipocytes: implications in obesity. Lipids 46: 1033–1042.
Tsuboi, M., A. Kawakami, T. Nakashima, N. Matsuoka, S. Urayama, Y. Kawabe, et al. 1999. Tumor necrosis factor-alpha and interleukin-1beta increase the Fas-mediated apoptosis of human osteoblasts. The Journal of Laboratory and Clinical Medicine 134: 222–231.
Yamamoto, S., M. Mogi, K. Kinpara, Y. Ishihara, N. Ueda, K. Amano, et al. 1999. Anti-proliferative capsular-like polysaccharide antigen from Actinobacillus actinomycetemcomitans induces apoptotic cell death in mouse osteoblastic MC3T3-E1 cells. Journal of Dental Research 78: 1230–1237.
McInnes, I.B., B.P. Leung, R.D. Sturrock, M. Field, and F.Y. Liew. 1997. Interleukin-15 mediates T cell-dependent regulation of tumor necrosis factor-alpha production in rheumatoid arthritis. Nature Medicine 3: 189–195.
Carson, W.E., M.E. Ross, R.A. Baiocchi, M.J. Marien, N. Boiani, K. Grabstein, et al. 1995. Endogenous production of interleukin 15 by activated human monocytes is critical for optimal production of interferon-gamma by natural killer cells in vitro. The Journal of Clinical Investigation 96: 2578–2582.
Schulthess, J., B. Meresse, E. Ramiro-Puig, N. Montcuquet, S. Darche, B. Begue, et al. 2012. Interleukin-15-dependent NKp46+ innate lymphoid cells control intestinal inflammation by recruiting inflammatory monocytes. Immunity 37: 108–121.
Kennedy, M.K., M. Glaccum, S.N. Brown, E.A. Butz, J.L. Viney, M. Embers, et al. 2000. Reversible defects in natural killer and memory CD8 T cell lineages in interleukin 15-deficient mice. The Journal of Experimental Medicine 191: 771–780.
Lodolce, J.P., D.L. Boone, S. Chai, R.E. Swain, T. Dassopoulos, S. Trettin, et al. 1998. IL-15 receptor maintains lymphoid homeostasis by supporting lymphocyte homing and proliferation. Immunity 9: 669–676.
Fehniger, T.A., K. Suzuki, A. Ponnappan, J.B. VanDeusen, M.A. Cooper, S.M. Florea, et al. 2001. Fatal leukemia in interleukin 15 transgenic mice follows early expansions in natural killer and memory phenotype CD8+ T cells. The Journal of Experimental Medicine 193: 219–231.
Ohta, N., T. Hiroi, M.N. Kweon, N. Kinoshita, M.H. Jang, T. Mashimo, et al. 2002. IL-15-dependent activation-induced cell death-resistant Th1 type CD8 alpha beta + NK1.1+ T cells for the development of small intestinal inflammation. The Journal of Immunology 169: 460–468.
Russell, J.H., and T.J. Ley. 2002. Lymphocyte-mediated cytotoxicity. Annual Review of Immunology 20: 323–370.
Hoogduijn, M.J., M. Roemeling-van Rhijn, S.S. Korevaar, A.U. Engela, W. Weimar, and C.C. Baan. 2011. Immunological aspects of allogeneic and autologous mesenchymal stem cell therapies. Human Gene Therapy 22: 1587–1591.
Carayol, G., C. Robin, J.H. Bourhis, A. Bennaceur-Griscelli, S. Chouaib, L. Coulombel, et al. 1998. NK cells differentiated from bone marrow, cord blood and peripheral blood stem cells exhibit similar phenotype and functions. European Journal of Immunology 28: 1991–2002.
Shikama, Y., M. Yamada, and T. Miyashita. 2003. Caspase-8 and caspase-10 activate NF-kappaB through RIP, NIK and IKKalpha kinases. European Journal of Immunology 33: 1998–2006.
Mouratidis, P.X., K.W. Colston, and A.G. Dalgleish. 2007. Doxycycline induces caspase-dependent apoptosis in human pancreatic cancer cells. International Journal of Cancer 120: 743–752.
Wang, T., R. Allie, K. Conant, N. Haughey, J. Turchan-Chelowo, K. Hahn, et al. 2006. Granzyme B mediates neurotoxicity through a G-protein-coupled receptor. The FASEB Journal 20: 1209–1211.
Acknowledgments
This work was supported in part by a Grant-in-Aid for Scientific Research (20791628) from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT), Tokyo, Japan, and a Aichi Gakuin University High-Tech Research Center (Aichi, Japan) Project for Private Universities, a matching fund subsidy from MEXT.
Conflict of Interest
All authors declare that they have no conflicts of interest to disclose in relation to the publication of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takeda, H., Kikuchi, T., Soboku, K. et al. Effect of IL-15 and Natural Killer Cells on Osteoclasts and Osteoblasts in a Mouse Coculture. Inflammation 37, 657–669 (2014). https://doi.org/10.1007/s10753-013-9782-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-013-9782-0