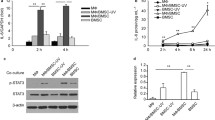
Abstract
In our study, we explored the bidirectional communication via soluble factors between bone cells and endotoxin-stimulated splenic lymphocytes in an in vitro coculture model that mimics the inflammatory environment. Both the ability of lymphocytes to affect differentiation and immune properties of bone cells, osteoblasts (OBL) and osteoclasts (OCL), and of bone cells to modulate cytokine and activation profile of endotoxin-stimulated lymphocytes were tested. LPS-pulsed lymphocytes enhanced OCL but inhibited OBL differentiation and increased the RANKL/OPG ratio, and, at the same time, upregulated chemotactic properties of bone cells, specifically CCL2, CCL5, and CXCL10 in OCL and CCL5 and CXCL13 in OBL. In parallel, bone cells had immunosuppressive effects by downregulating the lymphocyte expression of interleukin (IL)-1, IL-6, TNF-α and co-stimulatory molecules. OCL stimulated the production of osteoclastogenic cytokine RANKL in T lymphocytes. The anti-inflammatory effect, especially of OBL, suggests a possible compensatory mechanism to limit the inflammatory reaction during infection.





Similar content being viewed by others
References
Lorenzo, J., M. Horowitz, and Y. Choi. 2008. Osteoimmunology: interactions of the bone and immune system. Endocrine Reviews 29: 403–440.
Pittenger, M.F., A.M. Mackay, S.C. Beck, R.K. Jaiswal, R. Douglas, J.D. Mosca, M.A. Moorman, D.W. Simonetti, S. Craig, and D.R. Marshak. 1999. Multilineage potential of adult human mesenchymal stem cells. Science 284: 143–147.
Takayanagi, H. 2010. New immune connections in osteoclast formation. Annals of the New York Academy of Sciences 1192: 117–123.
Mundy, G.R. 2007. Osteoporosis and inflammation. Nutrition Reviews 65: S147–151.
Romas, E., and M.T. Gillespie. 2006. Inflammation-induced bone loss: can it be prevented? Rheumatic Diseases Clinics of North America 32: 759–773.
Theill, L.E., W.J. Boyle, and J.M. Penninger. 2002. RANK-L and RANK: T cells, bone loss, and mammalian evolution. Annual Review of Immunology 20: 795–823.
Kaisho, T., and S. Akira. 2000. Critical roles of Toll-like receptors in host defense. Critical Reviews in Immunology 20: 393–405.
Makó, V., J. Czúcz, Z. Weiszhár, E. Herczenik, J. Matkó, Z. Prohászka, and L. Cervenak. 2010. Proinflammatory activation pattern of human umbilical vein endothelial cells induced by IL-1β, TNF-α, and LPS. Cytometry A 77: 962–970.
Gao Y, Grassi F, Ryan MR, Terauchi M, Page K, Yang X, Weitzmann MN, Pacifici R. 207. IFN-gamma stimulates osteoclast formation and bone loss in vivo via antigen-driven T cell activation. Journal of Clinical Investigation 117: 122-132.
Hayashi, S., T. Yamada, M. Tsuneto, T. Yamane, M. Takahashi, L.D. Shultz, and H. Yamazaki. 2003. Distinct osteoclast precursors in the bone marrow and extramedullary organs characterized by responsiveness to Toll-like receptor ligands and TNF-alpha. Journal of Immunology 171: 5130–5139.
Kikuchi, T., T. Matsuguchi, N. Tsuboi, A. Mitani, S. Tanaka, M. Matsuoka, G. Yamamoto, T. Hishikawa, T. Noguchi, and Y. Yoshikai. 2001. Gene expression of osteoclast differentiation factor is induced by lipopolysaccharide in mouse osteoblasts via Toll-like receptors. Journal of Immunology 166: 3574–3579.
Mormann, M., M. Thederan, I. Nackchbandi, T. Giese, C. Wagner, and G.M. Hansch. 2008. Lipopolysaccharides (LPS) induce the differentiation of human monocytes to osteoclasts in a tumour necrosis factor (TNF) alpha-dependent manner: a link between infection and pathological bone resorption. Molecular Immunology 45: 3330–3337.
Kozuka, Y., Y. Ozaki, T. Ukai, T. Kaneko, and Y. Hara. 2006. B cells play an important role in lipopolysaccharide-induced bone resorption. Calcified Tissue International 78: 125–132.
Rodo, J., L.A. Goncalves, J. Demengeot, A. Coutinho, and C. Penha-Goncalves. 2006. MHC class II molecules control murine B cell responsiveness to lipopolysaccharide stimulation. Journal of Immunology 177: 4620–4626.
Ozaki, Y., T. Ukai, M. Yamaguchi, M. Yokoyama, E.R. Haro, M. Yoshimoto, T. Kaneko, M. Yoshinaga, H. Nakamura, C. Shiraishi, and Y. Hara. 2009. Locally administered T cells from mice immunized with lipopolysaccharide (LPS) accelerate LPS-induced bone resorption. Bone 44: 1169–1176.
Teng, Y.T., H. Nguyen, X. Gao, Y.Y. Kong, R.M. Gorczynski, B. Singh, R.P. Ellen, and J.M. Penninger. 2000. Functional human T-cell immunity and osteoprotegerin ligand control alveolar bone destruction in periodontal infection. Journal of Clinical Investigation 106: R59–67.
Garcia-Martinez, O., C. Reyes-Botella, O. Aguilera-Castillo, M.F. Vallecillo-Capilla, and C. Ruiz. 2006. Antigenic profile of osteoblasts present in human bone tissue sections. Bioscience Reports 26: 39–43.
Sato, K., A. Suematsu, K. Okamoto, A. Yamaguchi, Y. Morishita, Y. Kadono, S. Tanaka, T. Kodama, S. Akira, Y. Iwakura, D.J. Cua, and H. Takayanagi. 2006. Th17 functions as an osteoclastogenic helper T cell subset that links T cell activation and bone destruction. Journal of Experimental Medicine 203: 2673–2682.
Stanley, K.T., C. VanDort, C. Motyl, J. Endres, and D.A. Fox. 2006. Immunocompetent properties of human osteoblasts: interactions with T lymphocytes. Journal of Bone and Mineral Research 21: 29–36.
Hegyi, B., B. Sági, J. Kovács, J. Kiss, V.S. Urbán, G. Mészáros, E. Monostori, and F. Uher. 2010. Identical, similar or different? Learning about immunomodulatory function of mesenchymal stem cells isolated from various mouse tissues: bone marrow, spleen, thymus and aorta wall. International Immunology 22: 551–559.
Liu, H., D.M. Kemeny, B.C. Heng, H.W. Ouyang, A.J. Melendez, and T. Cao. 2006. The immunogenicity and immunomodulatory function of osteogenic cells differentiated from mesenchymal stem cells. Journal of Immunology 176: 2864–2871.
Uccelli, A., L. Moretta, and V. Pistoia. 2006. Immunoregulatory function of mesenchymal stem cells. Eururopean Journal of Immunology 36: 2566–2573.
Waterman, R.S., S.L. Tomchuck, S.L. Henkle, and A.M. Betancourt. 2010. A new mesenchymal stem cell (MSC) paradigm: polarization into a pro-inflammatory MSC1 or an Immunosuppressive MSC2 phenotype. PLoS One 5: e10088.
Li, H., S. Hong, J. Qian, Y. Zheng, J. Yang, and Q. Yi. 2010. Cross talk between the bone and immune systems: osteoclasts function as antigen-presenting cells and activate CD4+ and CD8+ T cells. Blood 116: 210–217.
Lee, S.H., T.S. Kim, Y. Choi, and J. Lorenzo. 2008. Osteoimmunology: cytokines and the skeletal system. BMB Reports 41: 495–510.
Ohno, T., N. Okahashi, I. Morisaki, and A. Amano. 2008. Signaling pathways in osteoblast proinflammatory responses to infection by Porphyromonas gingivalis. Oral Microbiology and Immunology 23: 96–104.
Silva, T.A., G.P. Garlet, S.Y. Fukada, J.S. Silva, and F.Q. Cunha. 2007. Chemokines in oral inflammatory diseases: apical periodontitis and periodontal disease. Journal of Dental Research 86: 306–319.
Kovacic, N., I.K. Lukic, D. Grcevic, V. Katavic, P. Croucher, and A. Marusic. 2007. The Fas/Fas ligand system inhibits differentiation of murine osteoblasts but has a limited role in osteoblast and osteoclast apoptosis. Journal of Immunology 178: 3379–3389.
Grcevic, D., I.K. Lukic, N. Kovacic, S. Ivcevic, V. Katavic, and A. Marusic. 2006. Activated T lymphocytes suppress osteoclastogenesis by diverting early monocyte/macrophage progenitor lineage commitment towards dendritic cell differentiation through down-regulation of receptor activator of nuclear factor-kappaB and c-Fos. Clinical and Experimental Immunology 146: 146–158.
Kovacic, N., D. Grcevic, V. Katavic, I.K. Lukic, V. Grubisic, K. Mihovilovic, H. Cvija, P.I. Croucher, and A. Marusic. 2010. Fas receptor is required for estrogen deficiency-induced bone loss in mice. Laboratory Investigation 90: 402–413.
Mattern, T., H.D. Flad, L. Brade, E.T. Rietschel, and A.J. Ulmer. 1998. Stimulation of human T lymphocytes by LPS is MHC unrestricted, but strongly dependent on B7 interactions. Journal of Immunology 160: 3412–3418.
Janeway Jr., C.A., and R. Medzhitov. 2002. Innate immune recognition. Annual Review of Immunology 20: 197–216.
Dumont, N., E. Aubin, D.P. Proulx, R. Lemieux, and R. Bazin. 2009. Increased secretion of hyperimmune antibodies following lipopolysaccharide stimulation of CD40-activated human B cells in vitro. Immunology 126: 588–595.
Dye, J.R., A. Palvanov, B. Guo, and T.L. Rothstein. 2007. B cell receptor cross-talk: exposure to lipopolysaccharide induces an alternate pathway for B cell receptor-induced ERK phosphorylation and NF-kappa B activation. Journal of Immunology 179: 229–235.
Tough, D.F., S. Sun, and J. Sprent. 1997. T cell stimulation in vivo by lipopolysaccharide (LPS). Journal of Experimental Medicine 185: 2089–2094.
Matsuguchi, T., K. Takagi, T. Musikacharoen, and Y. Yoshikai. 2000. Gene expressions of lipopolysaccharide receptors, toll-like receptors 2 and 4, are differently regulated in mouse T lymphocytes. Blood 95: 1378–1385.
Cazalis, J., S. Tanabe, G. Gagnon, T. Sorsa, and D. Grenier. 2009. Tetracyclines and chemically modified tetracycline-3 (CMT-3) modulate cytokine secretion by lipopolysaccharide-stimulated whole blood. Inflammation 32: 130–137.
Grcevic, D., S.K. Lee, A. Marusic, and J.A. Lorenzo. 2000. Depletion of CD4 and CD8 T lymphocytes in mice in vivo enhances 1,25-dihydroxyvitamin D3-stimulated osteoclast-like cell formation in vitro by a mechanism that is dependent on prostaglandin synthesis. Journal of Immunology 165: 4231–4238.
Craig, M.J., and R.D. Loberg. 2006. CCL2 (Monocyte chemoattractant protein-1) in cancer bone metastases. Cancer and Metastasis Reviews 25: 611–619.
Grassi, F., S. Cristino, S. Toneguzzi, A. Piacentini, A. Facchini, and G. Lisignoli. 2004. CXCL12 chemokine up-regulates bone resorption and MMP-9 release by human osteoclasts: CXCL12 levels are increased in synovial and bone tissue of rheumatoid arthritis patients. Journal of Cellular Physiology 199: 244–251.
Ryu, O.H., S.J. Choi, A.M. Linares, I.S. Song, Y.J. Kim, K.T. Jang, and T.C. Hart. 2007. Gingival epithelial cell expression of macrophage inflammatory protein-1alpha induced by interleukin-1beta and lipopolysaccharide. Journal of Periodontology 78: 1627–1634.
Wright, K.M., and J.S. Friedland. 2004. Regulation of monocyte chemokine and MMP-9 secretion by proinflammatory cytokines in tuberculous osteomyelitis. Journal of Leukocyte Biology 75: 1086–1092.
Kim, M.S., C.L. Magno, C.J. Day, and N.A. Morrison. 2006. Induction of chemokines and chemokine receptors CCR2b and CCR4 in authentic human osteoclasts differentiated with RANKL and osteoclast like cells differentiated by MCP-1 and RANTES. Journal of Cellular Biochemistry 97: 512–518.
Kwak, H.B., H. Ha, H.N. Kim, J.H. Lee, H.S. Kim, S. Lee, H.M. Kim, J.Y. Kim, H.H. Kim, Y.W. Song, and Z.H. Lee. 2008. Reciprocal cross-talk between RANKL and interferon-gamma-inducible protein 10 is responsible for bone-erosive experimental arthritis. Arthritis & Rheumatism 58: 1332–1342.
Lisignoli, G., S. Cristino, S. Toneguzzi, F. Grassi, A. Piacentini, C. Cavallo, A. Facchini, and E. Mariani. 2004. IL1beta and TNFalpha differently modulate CXCL13 chemokine in stromal cells and osteoblasts isolated from osteoarthritis patients: evidence of changes associated to cell maturation. Experimental Gerontology 39: 659–65.
Mansour, A., A. Anginot, S.J. Mancini, C. Schiff, G.F. Carle, A. Wakkach, and C. Blin-Wakkach. 2011. Osteoclast activity modulates B-cell development in the bone marrow. Cell Research 21: 1102–1115.
Liu, J., S. Wang, P. Zhang, N. Said-Al-Naief, S.M. Michalek, and X. Feng. 2009. Molecular mechanism of the bifunctional role of lipopolysaccharide in osteoclastogenesis. Journal of Biological Chemistry 284: 12512–1223.
Acknowledgments
Special thanks to Department of Immunology, Clinical Institute of Laboratory Diagnosis, University Clinical Hospital Centre Zagreb, Croatia. We thank Ms. K Zrinski-Petrović for her technical assistance. This work was supported by Croatian Ministry of Science, Education and Sports research grants (grant numbers 108–1080229–0140, 108–1080229–0142, 108–1080229–0341).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cvija, H., Kovacic, N., Katavic, V. et al. Chemotactic and Immunoregulatory Properties of Bone Cells are Modulated by Endotoxin-Stimulated Lymphocytes. Inflammation 35, 1618–1631 (2012). https://doi.org/10.1007/s10753-012-9477-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-012-9477-y