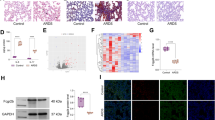
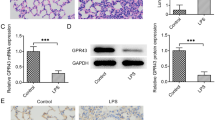
Abstract
Forkhead box protein A1 (FoxA1) is an evolutionarily conserved winged helix transcription factor with diverse regulatory functions. However, little is known about the role of FoxA1 in acute lung injury (ALI) and pulmonary cell injury. In this study, an in vivo model was employed whereby rats were administered an intravenous injection of oleic acid (OA, 0.1 ml/kg), and alveolar type II epithelial cells (AT-2 cells) injury was induced by hydrogen peroxide (H2O2) in vitro. OA injection resulted in lung injury and AT-2 cells apoptosis in vivo. OA injection and H2O2 upregulated FoxA1 mRNA and protein in lung tissue of the in vivo ALI model and in H2O2 challenged AT-2 cells. Overexpression of FoxA1 promoted apoptosis, whereas FoxA1 deficiency, induced by antisense oligonucleotides, decreased AT-2 cells apoptosis induced by H2O2, as shown by flow cytometry. These results suggest that FoxA1 may play an important role in ALI by promoting apoptosis of pulmonary epithelial cells.






Similar content being viewed by others
References
Goss, C. H., R. G. Brower, L. D. Hudson, and G. D. Rubenfeld. 2003. Incidence of acute lung injury in the United States. Crit. Care Med. 31:1607–1611. doi:10.1097/01.CCM.0000063475.65751.1D.
Mendez, J. L., and R. D. Hubmayr. 2005. New insights into the pathology of acute respiratory failure. Curr. Opin. Crit. Care. 11:29–36. doi:10.1097/00075198-200502000-00005.
Rubenfeld, G. D., E. Caldwell, E. Peabody, J. Weaver, D. P. Martin, M. Neff, et al. 2005. Incidence and outcomes of acute lung injury. N. Engl. J. Med. 353:1685–1693. doi:10.1056/NEJMoa050333.
Ware, L. B., and M. A. Matthay. 2000. The acute respiratory distress syndrome. N. Engl. J. Med. 342:1334–1349. doi:10.1056/NEJM200005043421806.
Crimi, E., and A. S. Slutsky. 2004. Inflammation and the acute respiratory distress syndrome. Best Pract. Res. Clin. Anaesthesiol. 18:477–492. doi:10.1016/j.bpa.2003.12.007.
Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. The Acute Respiratory Distress Syndrome Network. N. Engl. J. Med. 342:1301–1308. doi:10.1056/NEJM200005043421801
Matthay, M. A., G. A. Zimmerman, C. Esmon, J. Bhattacharya, B. Coller, C. M. Doerschuk, et al. 2003. Future research directions in acute lung injury: summary of a National Heart, Lung, and Blood Institute working group. Am. J. Respir. Crit. Care Med. 167:1027–1035. doi:10.1164/rccm.200208-966WS.
Mehta, D., J. Bhattacharya, M. A. Matthay, and A. B. Malik. 2004. Integrated control of lung fluid balance. Am. J. Physiol. Lung Cell. Mol. Physiol. 287:L1081–L1090. doi:10.1152/ajplung.00268.2004.
Slutsky, A. S., and L. D. Hudson. 2006. PEEP or no PEEP—lung recruitment may be the solution. N. Engl. J. Med. 354:1839–1841. doi:10.1056/NEJMe068045.
Tampo, Y., M. Tsukamoto, and M. Yonaha. 1999. Superoxide production from paraquat evoked by exogenous NADPH in pulmonary endothelial cells. Free Radic. Biol. Med. 27:588–595. doi:10.1016/S0891-5849(99)00110-0.
Zhang, H., A. S. Slutsky, and J. L. Vincent. 2000. Oxygen free radicals in ARDS, septic shock and organ dysfunction. Intensive Care Med. 26:474–476. doi:10.1007/s001340051185.
Yang, C., H. Moriuchi, J. Takase, Y. Ishitsuka, M. Irikura, and T. Irie. 2003. Oxidative stress in early stage of acute lung injury induced with oleic acid in guinea pigs. Biol. Pharm. Bull. 26:424–428. doi:10.1248/bpb.26.424.
Albertine, K. H., M. F. Soulier, Z. Wang, A. Ishizaka, S. Hashimoto, G. A. Zimmerman, et al. 2002. Fas and fas ligand are up-regulated in pulmonary edema fluid and lung tissue of patients with acute lung injury and the acute respiratory distress syndrome. Am. J. Pathol. 161:1783–1796.
Kitamura, Y., S. Hashimoto, N. Mizuta, A. Kobayashi, K. Kooguchi, I. Fujiwara, et al. 2001. Fas/FasL-dependent apoptosis of alveolar cells after lipopolysaccharide-induced lung injury in mice. Am. J. Respir. Crit. Care Med. 163:762–769.
Monaghan, A. P., K. H. Kaestner, E. Grau, and G. Schutz. 1993. Postimplantation expression patterns indicate a role for the mouse forkhead/HNF-3 alpha, beta and gamma genes in determination of the definitive endoderm, chordamesoderm and neuroectoderm. Development 119:567–578.
Ang, S. L., A. Wierda, D. Wong, K. A. Stevens, S. Cascio, J. Rossant, et al. 1993. The formation and maintenance of the definitive endoderm lineage in the mouse: involvement of HNF3/forkhead proteins. Development 119:1301–1315.
Sasaki, H., and B. L. Hogan. 1993. Differential expression of multiple fork head related genes during gastrulation and axial pattern formation in the mouse embryo. Development 118:47–59.
Ruiz i Altaba, A., C. Cox, T. M. Jessell, and A. Klar. 1993. Ectopic neural expression of a floor plate marker in frog embryos injected with the midline transcription factor Pintallavis. Proc. Natl. Acad. Sci. U. S. A. 90:8268–8272. doi:10.1073/pnas.90.17.8268.
Besnard, V., S. E. Wert, W. M. Hull, and J. A. Whitsett. 2004. Immunohistochemical localization of Foxa1 and Foxa2 in mouse embryos and adult tissues. Gene Expr. Patterns 5:193–208. doi:10.1016/j.modgep.2004.08.006.
Kaestner, K. H., H. Hiemisch, B. Luckow, and G. Schutz. 1994. The HNF-3 gene family of transcription factors in mice: gene structure, cDNA sequence, and mRNA distribution. Genomics 20:377–385. doi:10.1006/geno.1994.1191.
Lai, E., V. R. Prezioso, E. Smith, O. Litvin, R. H. Costa, and J. E. Darnell Jr. 1990. HNF-3A, a hepatocyte-enriched transcription factor of novel structure is regulated transcriptionally. Genes Dev. 4:1427–1436. doi:10.1101/gad.4.8.1427.
Besnard, V., S. E. Wert, K. H. Kaestner, and J. A. Whitsett. 2005. Stage-specific regulation of respiratory epithelial cell differentiation by Foxa1. Am. J. Physiol. Lung Cell. Mol. Physiol. 289:L750–L759. doi:10.1152/ajplung.00151.2005.
Vazquez de Lara, L., C. Becerril, M. Montano, C. Ramos, V. Maldonado, J. Melendez, et al. 2000. Surfactant components modulate fibroblast apoptosis and type I collagen and collagenase-1 expression. Am. J. Physiol. Lung Cell. Mol. Physiol. 279:L950–L957.
White, M. K., V. Baireddy, and D. S. Strayer. 2001. Natural protection from apoptosis by surfactant protein A in type II pneumocytes. Exp. Cell. Res. 263:183–192. doi:10.1006/excr.2000.5120.
Minoo, P., L. Hu, Y. Xing, N. L. Zhu, H. Chen, M. Li, et al. 2007. Physical and functional interactions between homeodomain NKX2.1 and winged helix/forkhead FOXA1 in lung epithelial cells. Mol. Cell. Biol. 27:2155–65. doi:10.1128/MCB.01133-06.
Nishina, K., K. Mikawa, Y. Takao, N. Maekawa, M. Shiga, and H. Obara. 1997. ONO-5046, an elastase inhibitor, attenuates endotoxin-induced acute lung injury in rabbits. Anesth. Analg. 84:1097–1103. doi:10.1097/00000539-199705000-00026.
Schittny, J. C., V. Djonov, A. Fine, and P. H. Burri. 1998. Programmed cell death contributes to postnatal lung development. Am. J. Respir. Cell. Mol. Biol. 18:786–793.
Mulier, B., I. Rahman, T. Watchorn, K. Donaldson, W. MacNee, and P. K. Jeffery. 1998. Hydrogen peroxide-induced epithelial injury: the protective role of intracellular nonprotein thiols (NPSH). Eur. Respir. J. 11:384–91. doi:10.1183/09031936.98.11020384.
Roberts, J. R., G. D. Perkins, T. Fujisawa, K. A. Pettigrew, F. Gao, A. Ahmed, et al. 2007. Vascular endothelial growth factor promotes physical wound repair and is anti-apoptotic in primary distal lung epithelial and A549 cells. Crit. Care Med. 35:2164–2170. doi:10.1097/01.CCM.0000281451.73202.F6.
Lockshin, R. A. 2005. Programmed cell death: history and future of a concept. J. Soc. Biol. 199:169–173. doi:10.1051/jbio:2005017.
Iuchi, T., M. Akaike, T. Mitsui, Y. Ohshima, Y. Shintani, H. Azuma, et al. 2003. Glucocorticoid excess induces superoxide production in vascular endothelial cells and elicits vascular endothelial dysfunction. Circ. Res. 92:81–87. doi:10.1161/01.RES.0000050588.35034.3C.
Cox, G., J. Crossley, and Z. Xing. 1995. Macrophage engulfment of apoptotic neutrophils contributes to the resolution of acute pulmonary inflammation in vivo. Am. J. Respir. Cell. Mol. Biol. 12:232–237.
Haslett, C., J. S. Savill, M. K. Whyte, M. Stern, I. Dransfield, and L. C. Meagher. 1994. Granulocyte apoptosis and the control of inflammation. Philos. Trans. R. Soc. Lond. B Biol. Sci. 345:327–333. doi:10.1098/rstb.1994.0113.
Matute-Bello, G., W. C. Liles, K. P. Steinberg, P. A. Kiener, S. Mongovin, E. Y. Chi, et al. 1999. Soluble Fas ligand induces epithelial cell apoptosis in humans with acute lung injury (ARDS). J. Immunol. 163:2217–2225.
Hashimoto, S., A. Kobayashi, K. Kooguchi, Y. Kitamura, H. Onodera, and H. Nakajima. 2000. Upregulation of two death pathways of perforin/granzyme and FasL/Fas in septic acute respiratory distress syndrome. Am. J. Respir. Crit. Care Med. 161:237–243.
Wan, H., S. Dingle, Y. Xu, V. Besnard, K. H. Kaestner, S. L. Ang, et al. 2005. Compensatory roles of Foxa1 and Foxa2 during lung morphogenesis. J. Biol. Chem. 280:13809–13816. doi:10.1074/jbc.M414122200.
Whitsett, J. A., and Y. Matsuzaki. 2006. Transcriptional regulation of perinatal lung maturation. Pediatr. Clin. North Am. 53:873–887. viii. doi:10.1016/j.pcl.2006.08.009.
Park, K. S., J. M. Wells, A. M. Zorn, S. E. Wert, V. E. Laubach, L. G. Fernandez, et al. 2006. Transdifferentiation of ciliated cells during repair of the respiratory epithelium. Am. J. Respir. Cell. Mol. Biol. 34:151–157 doi:10.1165/rcmb.2005-0332OC.
Bardales, R. H., S. S. Xie, R. F. Schaefer, and S. M. Hsu. 1996. Apoptosis is a major pathway responsible for the resolution of type II pneumocytes in acute lung injury. Am. J. Pathol. 149:845–852.
Wang, H. C., C. T. Shun, S. M. Hsu, S. H. Kuo, K. T. Luh, and P. C. Yang. 2002. Fas/Fas ligand pathway is involved in the resolution of type II pneumocyte hyperplasia after acute lung injury: evidence from a rat model. Crit. Care Med. 30:1528–34. doi:10.1097/00003246-200207000-00022.
Tewari, M., L. T. Quan, K. O’Rourke, S. Desnoyers, Z. Zeng, D. R. Beidler, et al. 1995. Yama/CPP32 beta, a mammalian homolog of CED-3, is a CrmA-inhibitable protease that cleaves the death substrate poly(ADP-ribose) polymerase. Cell 81:801–809. doi:10.1016/0092-8674(95)90541-3.
Matute-Bello, G., W. C. Liles, C. W. Frevert, M. Nakamura, K. Ballman, C. Vathanaprida, et al. 2001. Recombinant human Fas ligand induces alveolar epithelial cell apoptosis and lung injury in rabbits. Am. J. Physiol. Lung Cell. Mol. Physiol. 281:L328–L335.
Matute-Bello, G., R. K. Winn, M. Jonas, E. Y. Chi, T. R. Martin, and W. C. Liles. 2001. Fas (CD95) induces alveolar epithelial cell apoptosis in vivo: implications for acute pulmonary inflammation. Am. J. Pathol. 158:153–161.
Dobbs, L. G., R. Gonzalez, and M. C. Williams. 1986. An improved method for isolating type II cells in high yield and purity. Am. Rev. Respir. Dis. 134:141–145.
Dobbs, L. G. 1990. Isolation and culture of alveolar type II cells. Am. J. Physiol. 258:L134–L147.
Acknowledgments
This work was supported by the National Nature Science Foundation of China (grant no. 30330280), the National Nature Science Foundation of Hunan, China (grant no. 08JJ3030) and the Science Foundation Health Department of Hunan, China (grant no. 2007B090).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, L., Zhang, B., Feng, Y. et al. A Role for Forkhead Box A1 in Acute Lung Injury. Inflammation 32, 322–332 (2009). https://doi.org/10.1007/s10753-009-9139-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-009-9139-x