Abstract
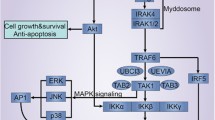
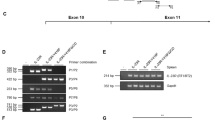
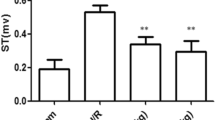
This study was designed to explore the role of toll like receptor 4 (TLR4) in myocardial ischemia reperfusion injury (MI/RI). Male Sprague–Dawley rats were randomly divided into sham and IR groups (36/group). The rats were sacrificed at various times following reperfusion (0, 1/2, 1, 2, 4 and 8 h). The histopathological and ultrastructual changes in the myocardium were examined under a light microscope and a transmission electron microscope. The TLR4 protein and mRNA expression were detected by immunohistochemistry and real-time reverse transcription polymerase chain reaction, respectively. The levels of tumor necrosis factor (TNF)-α and interleukin (IL)-6 in the myocardium were measured by enzyme-linked immunosorbent assays. The injury to the myocardium was severe in the IR group, and there were no significant improvements in histopathology and ultrastructure of the myocardium during the first 8 h following reperfusion. Positive TLR4 protein staining was observed in both sham and IR groups. The TLR4 protein levels were significantly increased in the IR group, peaking at 1 h post reperfusion. Additionally, the TLR4 mRNA levels were also up-regulated in the IR group. At all time points, IR rats had significantly higher TNF-α and IL-6 levels than the sham rats (P < 0.05). The TLR4 mRNA expression positively correlated with the levels of TNF-α and IL-6 (r = 0.728 and 0.676, P < 0.01). Myocardial TLR4 expression was elevated at the early stage of myocardial ischemia reperfusion. Activated TLR4 may play a role in MI/RI by increasing TNF-α and IL-6 expression.





Similar content being viewed by others
References
Ribichini, F., and W. Wijns. 2002. Acute myocardial infarction: reperfusion treatment. Heart 88(3):298–305 doi:10.1136/heart.88.3.298.
Braunwald, E.. 2002. Personal reflections on efforts to reduce ischemic myocardial damage. Cardiovasc. Res. 56(3):332–338 doi:10.1016/S0008-6363(02)00643-0.
Vakeva, A. P., A. Agah, S. A. Rollins, L. A. Matis, L. Li, and G. L. Stahl. 1998. Myocardial infarction and apoptosis after myocardial ischemia and reperfusion: role of the terminal complement components and inhibition by anti-C5 therapy. Circulation 97(22):2259–2267.
Ing, D. J., J. Zang, V. J. Dzau, K. A. Webster, and N. H. Bishopric. 1999. Modulation of cytokine-induced cardiac myocyte apoptosis by nitric oxide, Bak, and Bcl-x. Circ. Res. 84(1):21–33.
Frangogiannis, N. G., C. W. Smith, and M. L. Entman. 2002. The inflammatory response in myocardial infarction. Cardiovasc. Res. 53(1):31–47 doi:10.1016/S0008-6363(01)00434-5.
Ding, J. W., J. Yang, S. Li, L. Li, and Y. Chen. 2006. Effects of adenosine preconditioning on cardiomyocyte apoptosis induced by reperfusion. Chin. J. Exp. Surg. 23(12):1565 in Chinese.
Takeda, K., T. Kaisho, and S. Akira. 2003. Toll-like receptors. Annu. Rev. Immunol. 21:335–376 doi:10.1146/annurev.immunol.21.120601.141126.
Baumgarten, G., P. Knuefermann, N. Nozaki, N. Sivasubramanian, D. L. Mann, and J. G. Vallejo. 2001. In vivo expression of proinflammatory mediators in the adult heart after endotoxin administration: the role of toll-like receptor-4. J. Infect. Dis. 183(11):1617–1624 doi:10.1086/320712.
Oyama, J., C. Blais Jr, X. Liu, M. Pu, L. Kobzik, R. A. Kelly et al. 2004. Reduced myocardial ischemia-reperfusion injury in toll-like receptor 4-deficient mice. Circulation 109(6):784–789 doi:10.1161/01.CIR.0000112575.66565.84.
Chong, A. J., A. Shimamoto, C. R. Hampton, H. Takayama, D. J. Spring, C. L. Rothnie et al. 2004. Toll-like receptor 4 mediates ischemia/reperfusion injury of the heart. J. Thorac. Cardiovasc. Surg. 128(2):170–179 doi:10.1016/j.jtcvs.2003.11.036.
Maulik, N., R. M. Engelman, J. A. Rousou, J. E. Flack 3rd, D. Deaton, and D. K. Das. 1999. Ischemic preconditioning reduces apoptosis by upregulating anti-death gene Bcl-2. Circulation 100(Suppl19):II369–II375.
Arpornmaeklong, P., N. Suwatwirote, P. Pripatnanont, and K. Oungbho. 2007. Growth and differentiation of mouse osteoblasts on chitosan-collagen sponges. Int. J. Oral Maxillofac. Surg. 36(4):328–337 doi:10.1016/j.ijom.2006.09.023.
Marino, J. H., P. Cook, and K. S. Miller. 2003. Accurate and statistically verified quantification of relative mRNA abundances using SYBR Green I and real-time RT-PCR. J. Immunol. Methods 283(12):291–306 doi:10.1016/S0022-1759(03)00103-0.
Shibata, R., K. Sato, M. Kumada, Y. Izumiya, M. Sonoda, S. Kihara et al. 2007. Adiponectin accumulates in myocardial tissue that has been damaged by ischemia-reperfusion injury via leakage from the vascular compartment. Cardiovasc. Res. 74(3):471–479 doi:10.1016/j.cardiores.2007.02.010.
Liu, X., W. Xie, P. Liu, M. Duan, Z. Jia, W. Li et al. 2006. Mechanism of the cardioprotection of rhEPO pretreatment on suppressing the inflammatory response in ischemia-reperfusion. Life Sci. 78(19):2255–2264 doi:10.1016/j.lfs.2005.09.053.
Zhang, D., G. Zhang, M. S. Hayden, M. B. Greenblatt, C. Bussey, R. A. Flavell et al. 2004. A toll-like receptor that prevents infection by uropathogenic bacteria. Science 303(5663):1522–1526 doi:10.1126/science.1094351.
Poltorak, A., X. He, I. Smirnova, M. Y. Liu, C. Van Huffel, X. Du et al. 1998. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science 282(5396):2085–2088 doi:10.1126/science.282.5396.2085.
Chao, W., Y. Shen, X. Zhu, H. Zhao, M. Novikov, U. Schmidt et al. 2005. Lipopolysaccharide improves cardiomyocyte survival and function after serum deprivation. J. Biol. Chem. 280(23):21997–22005 doi:10.1074/jbc.M413676200.
Faxon, D. P., R. J. Gibbons, N. A. Chronos, P. A. Gurbel, and F. Sheehan. 2002. HALT-MI Investigators. The effect of blockade of the CD11/CD18 integrin receptor on infarct size in patients with acute myocardial infarction treated with direct angioplasty: the results of the HALT-MI study. J. Am. Coll. Cardiol. 40(7):1199–1204 doi:10.1016/S0735-1097(02)02136-8.
Frantz, S., L. Kobzik, Y. D. Kim, R. Fukazawa, R. Medzhitov, R. T. Lee et al. 1999. Toll4 (TLR4) expression in cardiacmyocytes in normal and failing myocardium. J. Clin. Invest. 104(3):271–280 doi:10.1172/JCI6709.
Otsui, K., N. Inoue, S. Kobayashi, R. Shiraki, T. Honjo, M. Takahashi et al. 2007. Enhanced expression of TLR4 in smooth muscle cells in human atherosclerotic coronary arteries. Heart Vessels 22(6):416–422 doi:10.1007/s00380-007-1001-1.
Zhai, Y., X. D. Shen, R. O’Connell, F. Gao, C. Lassman, R. W. Busuttil et al. 2004. Cutting edge: TLR4 activation mediates liver ischemia/reperfusion inflammatory response via IFN regulatory factor3-depend ent MyD88-independent pathway. J. Immunol. 173(12):7115–7119.
Boyd, J. H., S. Mathur, Y. Wang, R. M. Bateman, and K. R. Walley. 2006. Toll-like receptor stimulation in cardiomyoctes decreases contractility and initiates an NF-kappaB dependent inflammatory response. Cardiovasc. Res. 72(3):384–393 doi:10.1016/j.cardiores.2006.09.011.
D’Aiuto, F., M. Parkar, P. M. Brett, D. Ready, and M. S. Tonetti. 2004. Gene polymorphisms in pro-inflammatory cytokines are associated with systemic inflammation in patients with severe periodontal infections. Cytokine 28(1):29–34 doi:10.1016/j.cyto.2004.06.005.
Bhattacharyya, S., D. E. Brown, J. A. Brewer, S. K. Vogt, and L. J. Muglia. 2007. Macrophage glucocorticoid receptors regulate toll-like receptor 4-mediated inflammatory responses by selective inhibition of p38 MAP kinase. Blood 109(10):4313–4319 doi:10.1182/blood-2006-10-048215.
Sabroe, I., L. R. Prince, E. C. Jones, M. J. Horsburgh, S. J. Foster, S. N. Vogel et al. 2003. Selective roles for Toll-like receptor (TLR)2 and TLR4 in the regulation of neutrophil activation and life span. J. Immunol. 170(10):5268–5275.
Matsuguchi, T., A. Masuda, K. Sugimoto, Y. Nagai, and Y. Yoshikai. 2003. JNK-interacting protein 3 associates with Toll-like receptor 4 and is involved in LPS-mediated JNK activation. EMBO J. 22(17):4455–4464 doi:10.1093/emboj/cdg438.
Ohashi, K., V. Burkart, S. Flohé, and H. Kolb. 2000. Cutting edge: heat shock protein 60 is a putative endogenous ligand of the toll-like receptor-4 complex. J. Immunol. 164(2):558–561.
Kirchhoff, S. R., S. Gupta, and A. A. Knowlton. 2002. Cytosolic heat shock protein 60, apoptosis, and myocardial injury. Circulation 105(24):2899–2904 doi:10.1161/01.CIR.0000019403.35847.23.
Satoh, M., Y. Shimoda, T. Akatsu, Y. Ishikawa, Y. Minami, and M. Nakamura. 2006. Elevated circulating levels of heat shock protein 70 are related to systemic inflammatory reaction through monocyte Toll signal in patients with heart failure after acute myocardial infarction. Eur. J. Heart Fail. 8(8):810–815 doi:10.1016/j.ejheart.2006.03.004.
Stapel, H., S. C. Kim, S. Osterkamp, P. Knuefermann, A. Hoeft, R. Meyer et al. 2006. Toll-like receptor 4 modulates myocardial ischaemia-reperfusion injury: role of matrix metalloproteinases. Eur. J. Heart Fail. 8(7):665–672 doi:10.1016/j.ejheart.2006.03.005.
Medzhitov, R., P. Preston-Hurlburt, and C. A. Janeway Jr. 1997. A human homologue of the drosophila toll protein signals activation of adaptive immunity. Nature 388(6640):394–397 doi:10.1038/41131.
Magder, S., J. Neculcea, V. Neculcea, and R. Sladek. 2006. Lipopolysaccharide and TNF-alpha produce very similar changes in gene expression in human endothelial cells. J Vasc Res. 43(5):447–461 doi:10.1159/000095162.
Acknowledgements
We thank Professors C. Zhang and L. Han of the Faculty of the Microbiology Department, Professor Y. Hu and Dr. B. Gao of the Department of Pathology of the First College of Clinical Medical Sciences, and the China Three Gorges University for their helpful discussions and suggestions on this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, J., Yang, J., Ding, Jw. et al. Sequential Expression of TLR4 and its Effects on the Myocardium of Rats with Myocardial Ischemia-Reperfusion Injury. Inflammation 31, 304–312 (2008). https://doi.org/10.1007/s10753-008-9079-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-008-9079-x