Abstract
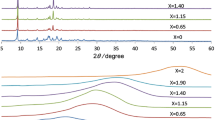
Linkage isomerism is the coexistence of iso-compositional molecules or solids differing by connectivity of the metal to a ligand. In a crystalline solid state, the rotation is possible for asymmetric ligands, e.g., for cyanide ligand. Here we report on our observation of a phase transition in anhydrous RbMn[Fe(CN)6] (nearly stoichiometric) and on the effect of linkage isomerism ensuing our interpretation of the results of Mössbauer study in which we observe the iron spin state crossover among two phases involved into this transition. The anhydrous RbMn[Fe(CN)6] can be prepared via prolonged thermal treatment (1 week at at 80 °C) of the as-synthesized hydrated RbMn[Fe(CN)6]·H2O. The latter compound famous for its charge-transfer phase transition is a precursor in our case. As the temperature is raising above 80 °C (remaining below 100 °C) we observe RbMn[Fe(CN)6] that inherited its F-43 m symmetry from RbMn[Fe(CN)6]·H2O transforming to a phase of the Fm-3 m symmetry. In the latter, more than half of Fe3 + ions are in high-spin state. We suggest a plausible way to explain the spin-crossover that is to allow the linkage isomerism by rotation of the cyanide ligands.
Similar content being viewed by others
References
Buschmann, W.E., Ensling, J., Gütlich, Ph., Miller, J.S.: Chem. Eur. J. 5, 3019–3028 (1999)
Salmon, L., Vertelman, E.J.M., Murgui, C.B., Cobo, S., Molnár, G., van Koningsbruggen, P.J., Bousseksou, A.: Eur. J. Inorg. Chem. 6, 760–768 (2009)
Moritomo, Y., Kato, K., Kuriki, A., Takata, M., Sakata, M., Tokoro, H., Ohkoshi, S.-I., Hashimoto, K.: J. Phys. Soc. Jpn. 71, 2078 (2002)
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Rykov, A.I., Wang, J., Zhang, T. et al. A structural phase transition coupled to the Fe3+ spin-state crossover in anhydrous RbMn[Fe(CN)6]. Hyperfine Interact 218, 139–143 (2013). https://doi.org/10.1007/s10751-012-0692-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10751-012-0692-6