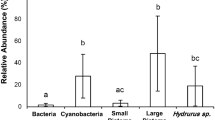
Abstract
Food webs in ecosystems are mainly considered on a large spatial scale, but in forest streams, canopy cover in riparian forests might shape the food web structures on a reach scale. We hypothesized that the food resources for invertebrate consumers (e.g., grazers and filter-feeding caddisflies) shift along the river channel from open to closed canopy-covered reaches and vice versa on the riffle scale. In other words, the distance from a canopy-covered reach determines the food resources available to those consumers. To test the hypothesis, we analyzed the riffle scale variability in invertebrate consumers food resources in a canopy-covered stream using carbon and nitrogen stable isotopes (δ13C and δ15N, ‰) We measured the isotopes of grazer: Glossosoma spp. (ranged δ13C: − 13.7 to − 33.3, δ15N: 0.3 to 11.0) and filter feeder: Stenopsyche marmorata Navas, 1920 (δ13C: − 16.8 to − 30.7, δ15N: 0.7 to 11.0). The δ13C and δ15N of resources are varied: periphyton (δ13C: − 11.3 to − 32.5, δ15N: 0.1–6.6), SPOM (δ13C: − 13.8 to − 33.0, δ15N: 0.1 to 8.4), and BPOM (δ13C: − 14.0 to − 32.4, δ15N: 0.1 to 7.0). The results from the stable isotope-mixing models show that the periphytic contribution to the filter feeders was lower within and downstream of the canopy-covered riffle than in the open riffle. We found that canopy cover changed the basal food resources available to invertebrate consumers from periphyton to terrestrial matter on a riffle scale, particularly for the filter feeders. Thus, even within a reach, a loss of riparian forest could change the basal resource availability to the dominant consumers. Canopy cover affects the resources of the stream food web and can be significant even on a riffle scale. Thus, small-spatial level management of riparian forest can induce the resource heterogeneity to stream food webs.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Behmer, D. J. & C. P. Hawkins, 1986. Effects of overhead canopy on macroinvertebrate production in a Utah USA stream. Freshwater Biology 16: 287–300. https://doi.org/10.1111/j.1365-2427.1986.tb01072.x.
Bootsma, H. A., R. E. Hecky, R. H. Hesslein & G. F. Turner, 1996. Food partitioning among lake Malawi nearshore fishes as revealed by stable isotope analysis. Ecology 77: 1286–1290. https://doi.org/10.2307/2265598.
Doi, H., 2009. Spatial patterns of autochthonous and allochthonous resources to aquatic food webs. Population Ecology 51: 57–64. https://doi.org/10.1007/s10144-008-0127-z.
Doi, H., I. Katano & E. Kikuchi, 2006. The use of algal-mat habitat by aquatic insect grazers: effects of microalgal cue. Basic and Applied Ecology 7: 153–158. https://doi.org/10.1016/j.baae.2005.04.009.
Doi, H., Y. Takemon, T. Ohta, Y. Ishida & E. Kikuchi, 2007. Effects of reach scale canopy cover on trophic pathways of caddisfly larvae in a mountain stream, Japan. Marine and Freshwater Research 58: 811–817. https://doi.org/10.1071/MF07067.
Doi, H., K. H. Chang, T. Ando, H. Imai, S. Nakano, A. Kajimoto, et al., 2008. Drifting plankton from a reservoir subsidize downstream food webs and alter community structure. Oecologia 156: 363–371. https://doi.org/10.1007/s00442-008-0988-z.
Erdozain, M., K. Kidd, D. Kreutzweiser & P. Sibley, 2019. Increased reliance of stream macroinvertebrates on terrestrial food sources linked to forest management intensity. Ecological Applications 29: 1–14. https://doi.org/10.1002/eap.1889.
Feminella, J. W., M. E. Power & V. H. Resh, 1989. Periphyton responses to invertebrate grazing and riparian canopy in three northern California streams. Freshwater Biology 22: 445–457. https://doi.org/10.1111/j.1365-2427.1989.tb01117.x.
Finlay, J. C., M. E. Power & G. Cabana, 1999. Effects of water velocity on algal carbon isotope ratios: implications of river food web studies. Limnology and Oceanography 44: 1198–1203. https://doi.org/10.4319/lo.1999.44.5.1198.
Finlay, J. C., S. Khandwala & M. E. Power, 2002. Spatial scales of carbon flow in a river food web. Ecology Letters 5: 197–207. https://doi.org/10.1890/0012-9658(2002)083[1845:SSOCFI]2.0.CO;2.
France, R. L., 1995. Critical examination of stable isotope analysis as a means for tracing carbon pathways in stream ecosystems. Canadian Journal of Fisheries and Aquatic Sciences 52: 651–656. https://doi.org/10.1139/f95-065.
Fry, B., 1996. 13C/12C fractionation by marine diatoms. Marine Ecology Progress Series 134: 283–294. https://doi.org/10.3354/meps134283.
Giling, D. P., P. Reich & R. M. Thompson, 2009. Loss of riparian vegetation alters the ecosystem role of a freshwater crayfish (Cherax destructor) in an Australian intermittent lowland stream. Journal of the North American Benthological Society 28: 626–637. https://doi.org/10.1899/08-130.1.
Gouhier, T. C., F. Guichard & A. Gonzalez, 2010. Synchrony and stability of food webs in metacommunities. American Naturalist 175: E16–E34. https://doi.org/10.1086/649579.
Hill, W. R., M. G. Ryon & E. M. Schilling, 1995. Light limitation in a stream ecosystem: responses by primary producers and consumers. Ecology 76: 1297–1309. https://doi.org/10.2307/1940936.
Hill, W. R., P. J. Mulholland & E. R. Marzolf, 2001. Stream ecosystem responses to forest leaf emergence in spring. Ecology 82: 2306–2319. https://doi.org/10.1890/0012-9658(2001)082[2306:SERTFL]2.0.CO;2.
Ishikawa, N. F., H. Doi & J. C. Finlay, 2012. Global meta-analysis for controlling factors on carbon stable isotope ratio of lotic periphyton. Oecologia 170: 541–549. https://doi.org/10.1007/s00442-012-2308-x.
Katano, I., H. Doi, A. Houki, Y. Isobe & T. Oishi, 2007. Changes in periphyton abundance and community structure with the dispersal of a caddisfly grazer, Micrasema quadriloba. Limnology 8: 219–226. https://doi.org/10.1007/s10201-007-0211-7.
Katano, I., J. N. Negishi, T. Minagawa, H. Doi, Y. Kawaguchi & Y. Kayaba, 2009. Longitudinal macroinvertebrate organization over contrasting discontinuities: effects of a dam and a tributary. Journal of the North American Benthological Society 28: 331–351. https://doi.org/10.1899/08-010.1.
Kato, Y., M. Hori, N. Okuda, I. Tayasu & Y. Takemon, 2009. Spatial heterogeneity of trophic pathways in the invertebrate community of a temperate bog. Freshwater Biology 55: 450–462. https://doi.org/10.1111/j.1365-2427.2009.02295.x.
Lamberti, G. A. & A. D. Steinman, 1997. A comparison of primary production in stream ecosystems. Journal of the North American Benthological Society 16: 95–103. https://doi.org/10.2307/1468241.
Lamberti, G. A., D. T. Chaloner & A. E. Hershey, 2010. Linkages among aquatic ecosystems. Journal of the North American Benthological Society 29: 245–263. https://doi.org/10.1899/08-166.1.
Lau, D. C. P., K. M. Y. Leung & D. David, 2009. What does stable isotope analysis reveal about trophic relationships and the relative importance of allochthonous and autochthonous resources in tropical streams? A synthetic study from Hong Kong. Freshwater Biology 54: 127–141. https://doi.org/10.1111/j.1365-2427.2008.02099.x.
McCutchan, J. H., Jr., W. M. Lewis Jr., C. Kendall & C. C. McGrath, 2003. Variation in trophic shift for stable isotope ratios of carbon, nitrogen, and sulfur. Oikos 102: 378–390. https://doi.org/10.1034/j.1600-0706.2003.12098.x.
Nakano, S. & M. Murakami, 2001. Reciprocal subsidies: dynamic interdependence between terrestrial and aquatic food webs. Proceedings of the National Academy of Sciences of the United States of America 98: 166–170. https://doi.org/10.1073/pnas.98.1.166.
Nishimura, N., 1966. Ecological studies on the net-spinning caddisfly, Stenopsyche griseipennis MCLACHLAN (Trichoptera, Stenopsychidae) 1. Life history and habit. Mushi 39: 103–114.
Ock, G., & Takemon, Y. (2009). Downstream changes in source composition of particulate organic matter in dam tailwaters in relation to channel geomorphology and benthos communities. In The 33rd IAHR Congress: Water Engineering for a Sustainable Environment: 3672–3679.
Polis, G. A., W. B. Anderson & R. D. Holt, 1997. Towards an integration of landscape and food web ecology: the dynamics of spatially subsidized food webs. Annual Review of Ecology and Systematics 28: 289–316. https://doi.org/10.1146/annurev.ecolsys.28.1.289.
Post, D. M., 2002. Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83: 703–718. https://doi.org/10.1890/0012-9658(2002)083[0703:USITET]2.0.CO;2.
R Core Team, 2021. R: A Language and Environment for Statistical Computing, R Foundation for Statistical Computing, Vienna:
Schindler, D. E. & M. D. Scheuerell, 2002. Habitat coupling in lake ecosystems. Oikos 98: 177–189. https://doi.org/10.1034/j.1600-0706.2002.980201.x.
SCOR-UNESCO, 1966. Determination of photosynthetic pigments. In Monographs on Oceanographic Methodology, Vol. 1. UNESCO, Paris: 11–18.
Takemon, Y., 2005. Life type concept and functional feeding groups of benthos communities as indicators of lotic ecosystem conditions. Japanese Journal of Ecology 55: 189–197.
Takemon, Y., Y. Imai, A. Kohzu, T. Nagata & S. Ikebuchi, 2008. Spatial distribution patterns of allochtonous and autochtonous benthic particulate organic matter on the riverbed of a mountain stream in Kyoto, Japan. In Water Down Under: 2393–2403.
Tanida, K., H. Mitsuhashi & T. Fujitani, 1999. A simple acrylate fiber sampler for stream periphyton. Japanese Journal of Limnology 60: 619–624.
Thompson, R. M. & C. R. Townsend, 2005. Food-web topology varies with spatial scale in a patchy environment. Ecology 86: 1916–1925. https://doi.org/10.1890/04-1352.
Thompson, R. M., U. Brose, J. A. Dunne, R. O. Hall Jr., S. Hladyz, R. L. Kitching, et al., 2012. Food webs: reconciling the structure and function of biodiversity. Trends in Ecology & Evolution 27: 689–697. https://doi.org/10.1016/j.tree.2012.08.005.
Vannote, R. L., G. W. Minshall, K. W. Cummins, J. R. Sedell & C. E. Cushing, 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences 37: 130–137. https://doi.org/10.1139/f80-017.
Venables, W. N. & B. D. Ripley, 2002. Modern Applied Statistics with S, 4th ed. Springer, Cham: https://doi.org/10.1007/978-0-387-21706-2.
Wallace, J. B., S. L. Eggert, J. L. Meyer & J. R. Webster, 1997. Multiple trophic levels of a forest stream linked to terrestrial litter inputs. Science 277: 102–104. https://doi.org/10.1126/science.277.5322.102.
Winemiller, K. O., A. S. Flecker & D. J. Hoeinghaus, 2010. Patch dynamics and environmental heterogeneity in lotic ecosystems. Journal of the North American Benthological Society 29: 84–99. https://doi.org/10.1899/08-048.1.
Acknowledgements
We thank T. Tanaka, K. Hatano, K. Yamamoto, and T. Ohta of the Graduate School of Engineering at Kyoto University for their help with the field work. This research was supported by JSPS KAKENHI Grant Numbers 15206058 and 19360224, and by CREST, JST.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no competing interests exist.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guest editors: Luiz Ubiratan Hepp, Frank Onderi Masese & Franco Teixeira de Mello / Stream Ecology and Environmental Gradients
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Doi, H., Ishida, Y. & Takemon, Y. Canopy cover influences food resources for stream grazers and filter feeders on a reach scale. Hydrobiologia 851, 299–311 (2024). https://doi.org/10.1007/s10750-023-05213-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-023-05213-7