Abstract
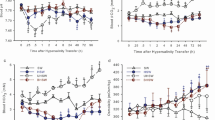
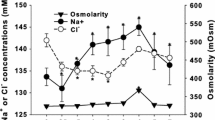
Metabolite changes in Nile tilapia in response to carbonate alkalinity stress were investigated by transferring the fish directly from freshwater into different carbonate alkaline water. Levels of plasma pH/HCO3− concentration, the mRNA and protein expression of two carbonic anhydrases (CAhz and CAIV), and two HCO3− transporters (Na+/HCO3− cotransporter and Cl−/HCO3– exchanger) in the gill, kidney, and intestine were determined using a pH meter, UV spectrophotometer, quantitative real-time PCR, and western blotting within 192 h of exposure. Plasma pH showed an “up-peak-down” variation, whereas HCO3− concentration decreased at first and then increased in all alkaline water groups. The overall mRNA expression was regulated in an alkalinity- and time-dependent manner. Western blot results showed that the Cl−/HCO3− exchanger protein was detected in all tissues examined, whereas the two carbonic anhydrases and Na+/HCO3− cotransporter proteins were only expressed in the gill and kidney. Therefore, the studied carbonic anhydrases and HCO3− transporters are involved in the HCO3− metabolism and transport to maintain acid–base balance in Nile tilapia under carbonate alkalinity stress.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Alper, S. L. & A. K. Sharma, 2013. The SLC26 gene family of anion transporters and channels. Molecular Aspects of Medicine 34: 494–515. https://doi.org/10.1016/j.mam.2012.07.009.
Boyle, D., A. M. Clifford, E. Orr, D. Chamot & G. G. Goss, 2015. Mechanisms of Cl− uptake in rainbow trout: Cloning and expression of slc26a6, a prospective Cl−/HCO3− exchanger. Comparative Biochemistry and Physiology Part A Molecular and Integrative Physiology 180: 43–50. https://doi.org/10.1016/j.cbpa.2014.11.001.
Brauner, C. J., R. B. Shartau, C. Damsgaard, A. J. Esbaugh, R. W. Wilson & M. Grosell, 2019. 3-Acid–base physiology and CO2 homeostasis: regulation and compensation in response to elevated environmental CO2. Fish Physiology 37: 69–132. https://doi.org/10.1016/bs.fp.2019.08.003.
Chang, Y. M., X. F. Zhao, H. J. Liew, B. Sun, S. Y. Wang, L. Luo, L. M. Zhang & L. Q. Liang, 2021. Effects of bicarbonate stress on serum ions and gill transporters in alkali and freshwater forms of amur ide (Leuciscus waleckii). Frontiers in Physiology 12: 676096. https://doi.org/10.3389/fphys.2021.676096.
Evans, D. H., P. M. Piermarini & K. P. Choe, 2005. The multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid–base regulation, and excretion of nitrogen waste. Physiological Reviews 85: 97–177. https://doi.org/10.1152/physrev.00050.2003.
Galat, D. L., G. Post, T. J. Keefe & G. R. Bouck, 1985. Histological changes in the gill, kidney and liver of Lahontan cutthroat trout, Salmo clarki henshawi, living in lakes of different salinity-alkalinity. Journal of Fish Biology 27: 533–552. https://doi.org/10.1111/j.1095-8649.1985.tb03200.x.
Georgalis, T., S. F. Perry & K. M. Gilmour, 2006. The role of branchial carbonic anhydrase in acid–base regulation in rainbow trout (Oncorhynchus mykiss). Journal of Experimental Biology 209: 518. https://doi.org/10.1242/jeb.02018.
Gilmour, K. M. & S. F. Perry, 2009. Carbonic anhydrase and acid–base regulation in fish. Journal of Experimental Biology 212: 1647–1661. https://doi.org/10.1242/jeb.029181.
Goss, G. G. & S. F. Perry, 1994. Different mechanisms of acid–base regulation in rainbow trout (Oncorhynchus mykiss) and american eel (Anguilla rostrata) during NaHCO3 infusion. Physiological Zoology 67: 381–406. https://doi.org/10.1086/physzool.67.2.30163854.
Goss, G. G., S. F. Perry, C. M. Wood & P. Laurent, 1992. Mechanisms of ion and acid–base regulation at the gills of freshwater fish. The Journal of Experimental Zoology 263: 143–159. https://doi.org/10.1002/jez.1402630205.
Grosell, M., 2006. Intestinal anion exchange in marine fish osmoregulation. The Journal of Experimental Biology 209: 2813–2827. https://doi.org/10.1242/jeb.02345.
Grosell, M., 2010. 4-The role of the gastrointestinal tract in salt and water balance. Fish Physiology 30: 135–164. https://doi.org/10.1016/S1546-5098(10)03004-9.
Grosell, M., 2011. Intestinal anion exchange in marine teleosts is involved in osmoregulation and contributes to the oceanic inorganic carbon cycle. Acta Physiologica 202: 421–434. https://doi.org/10.1111/j.1748-1716.2010.02241.x.
Grosell, M. & J. R. Taylor, 2007. Intestinal anion exchange in teleost water balance. Comparative Biochemistry and Physiology Part a, Molecular and Integrative Physiology 148: 14–22. https://doi.org/10.1016/j.cbpa.2006.10.017.
Kurita, Y., T. Nakada, A. Kato, H. Doi, A. C. Mistry, M. H. Chang & S. Hirose, 2008. Identification of intestinal bicarbonate transporters involved in formation of carbonate precipitates to stimulate water absorption in marine teleost fish. American Journal of Physiology Regulatory Integrative and Comparative Physiology 294: R1402. https://doi.org/10.1152/ajpregu.00759.2007.
Li, C. C. & J. C. Chen, 2008. The immune response of white shrimp Litopenaeus vannamei and its susceptibility to Vibrio alginolyticus under low and high pH stress. Fish and Shellfish Immunology 25: 701–709. https://doi.org/10.1016/j.fsi.2008.01.007.
Liu, Y., M. Yao, S. Li, X. Wei, L. Ding, S. Han, P. Wang, B. Lv, Z. Chen & Y. Sun, 2022. Integrated application of multi-omics approach and biochemical assays provides insights into physiological responses to saline-alkaline stress in the gills of crucian carp (Carassius auratus). Science of the Total Environment 822: 153622. https://doi.org/10.1016/j.scitotenv.2022.153622.
Michael, K., C. M. Kreiss, M. Y. Hu, N. Koschnick, U. Bickmeyer, S. Dupont, H. O. Pörtner & M. Lucassen, 2016. Adjustments of molecular key components of branchial ion and pH regulation in Atlantic cod (Gadus morhua) in response to ocean acidification and warming. Comparative Biochemistry and Physiology Part b, Biochemistry and Molecular Biology 193: 33–46. https://doi.org/10.1016/j.cbpb.2015.12.006.
Mount, D. B. & M. F. Romero, 2004. The SLC26 gene family of multifunctional anion exchangers. Pflügers Archiv European Journal of Physiology 447: 710–721. https://doi.org/10.1007/s00424-003-1090-3.
Parra, J. G. & B. Baldisserotto, 2007. Effect of Water pH and Hardness on Survival and Growth of Freshwater Teleosts. In Kapoor, B. (ed), Fish Osmoregulation Science Publishers, New York: 135–150.
Perry, S. F., M. Furimsky, M. Bayaa, T. Georgalis, A. Shahsavarani, J. G. Nickerson & T. W. Moon, 2003. Integrated responses of Na+/HCO3- cotransporters and V-type H+-ATPases in the fish gill and kidney during respiratory acidosis. Biochimica Et Biophysica Acta (BBA) Biomembranes 1618: 175–184. https://doi.org/10.1016/j.bbamem.2003.09.015.
Pierre, L., P. W. Michael, C. Claudine & M. W. Chris, 2000. The effect of highly alkaline water (pH 9.5) on the morphology and morphometry of chloride cells and pavement cells in the gills of the freshwater rainbow trout: Relationship to ionic transport and ammonia excretion. Canadian Journal of Zoology 78: 307–319. https://doi.org/10.1139/z99-207.
Romero, M. F. & W. F. Boron, 1999. Electrogenic Na+/HCO3− cotransporters: cloning and physiology. Annual Review of Physiology 61: 699–723. https://doi.org/10.1146/annurev.physiol.61.1.699.
Romero, M. F., C. M. Fulton & W. F. Boron, 2004. The SLC4 family of HCO3- transporters. Pflügers Archiv: European Journal of Physiology 447: 495–509. https://doi.org/10.1007/s00424-003-1180-2.
Ruiz-Jarabo, I., S. F. Gregório, P. Gaetano, F. Trischitta & J. Fuentes, 2017. High rates of intestinal bicarbonate secretion in seawater tilapia (Oreochromis mossambicus). Comparative Biochemistry and Physiology Part a, Molecular and Integrative Physiology 207: 57–64. https://doi.org/10.1016/j.cbpa.2017.02.022.
Sender, S., K. Böttcher, Y. Cetin & G. Gros, 1999. Carbonic anhydrase in the gills of seawater- and freshwater-acclimated flounders Platichthys flesus: purification, characterization, and immunohistochemical localization. Journal of Histochemistry and Cytochemistry 47: 43–50. https://doi.org/10.1177/002215549904700105.
Tang, C. H., L. Y. Hwang & T. H. Lee, 2010. Chloride channel ClC-3 in gills of the euryhaline teleost, Tetraodon nigroviridis: expression, localization and the possible role of chloride absorption. Journal of Experimental Biology 213: 683–693. https://doi.org/10.1242/jeb.040212.
Taylor, J. R., E. M. Mager & M. Grosell, 2010. Basolateral NBCe1 plays a rate-limiting role in transepithelial intestinal HCO3- secretion, contributing to marine fish osmoregulation. Journal of Experimental Biology 213: 459–468. https://doi.org/10.1242/jeb.029363.
Wang, P., Q. F. Lai, Z. L. Yao, K. Zhou, T. T. Lin & H. Wang, 2015. Differential expressions of genes related to HCO3- secretion in the intestine of Gymnocypris przewalskiii during saline-alkaline water transfer. Marine Fisheries 37: 341–348. https://doi.org/10.13233/j.cnki.mar.fish.2015.04.007 (in Chinese).
Wang, Y., J. L. Zhao, J. W. Wu, T. Jeerawat, M. M. Yue & Y. Zhao, 2016. Effects of alkalinity on morphology of gill ionocytes and HCO3- transporters in gill, kidney and intestine of nile tilapia (Oreochromis niloticus). Chinese Journal of Zoology 51: 1027–1037. https://doi.org/10.13859/j.cjz.201606011 (in Chinese).
Wang, S., Y. Kuang, L. Liang, B. Sun, X. Zhao, L. Zhang & Y. Chang, 2021. Resequencing and SNP discovery of Amur ide (Leuciscus waleckii) provides insights into local adaptations to extreme environments. Scientific Reports 11: 5064. https://doi.org/10.1038/s41598-021-84652-5.
Wilkie, M. P. & C. M. Wood, 1996. The adaptations of fish to extremely alkaline environments. Comparative Biochemistry and Physiology Part b: Biochemistry and Molecular Biology 113: 665–673. https://doi.org/10.1016/0305-0491(95)02092-6.
Wilkie, M. P., P. A. Wright, G. K. Iwama & C. M. Wood, 1994. The physiological adaptations of the lahontan cutthroat trout (Oncorhynchus clarki henshawi) following transfer from well water to the highly alkaline waters of Pyramid Lake, Nevada (pH 9.4). Physiological Zoology 67: 355–380. https://doi.org/10.1086/physzool.67.2.30163853.
Xu, Z. X., L. Gan, T. Y. Li, C. Xu, K. Chen, X. D. Wang, J. G. Qin, L. Q. Chen & E. Li, 2015. Transcriptome profiling and molecularpathway analysis of genes in association with salinity adaptation in Nile Tilapia Oreochromis niloticus. PLoS ONE 10: e0136506. https://doi.org/10.1371/journal.pone.0136506.
Zhao, L. H., J. L. Zhao, J. Thammaratsuntorn & Y. Zhao, 2014. Effects of salinity-alkalinity on serum osmolality, ion concentration and mRNA expression of ion transport enzymes of Oreochromis niloticus. Journal of Fisheries of China 38: 1696–1704 (in Chinese).
Zhao, Y., C. S. Zhang, H. T. Zhou, L. Y. Song, J. Wang & J. L. Zhao, 2020. Transcriptome changes for Nile tilapia (Oreochromis niloticus) in response to alkalinity stress. Comparative Biochemistry and Physiology Part d, Genomics and Proteomics 33: 100651. https://doi.org/10.1016/j.cbd.2019.100651.
Acknowledgements
This research was supported by the National Key R&D Program of China (Grant No. 2020YFD0900400), the China Agriculture Research System of MOF and MARA (Grant No. CARS-46), and the National Natural Science Foundation of China (Grant No. 31602128). We thank International Science Editing (http://www.internationalscienceediting.com) for editing this manuscript. I certify that this manuscript is original and has not been published elsewhere in any form or language and will not be submitted elsewhere for publication while being considered by Hydrobiologia. In addition, the study is not split into several parts to increase the number of submissions and submitted to various journals or one journal over time. No data have been fabricated or manipulated (including images) to support our conclusions. No data, text, or theories by others are presented as if they were our own. Proper acknowledgments to other works have been given.
Funding
This research was supported by the National Key R&D Program of China (Grant No. 2020YFD0900400), the China Agriculture Research System of MOF and MARA (Grant No. CARS-46), and the National Natural Science Foundation of China (Grant No. 31602128).
Author information
Authors and Affiliations
Contributions
The submission has been received explicitly from all co-authors. Authors whose names appear on the submission have contributed sufficiently to the scientific work and therefore share collective responsibility and accountability for the results.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that no competing financial interests are associated with the work submitted for publication. We declare that we do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted. The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the animal ethics committee of Shanghai Ocean University (approval no. 2018–085).
Research involving human rights
This article does not contain any studies with human participants performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guest editors: S. Koblmüller, R. C. Albertson, M. J. Genner, K. M. Sefc & T. Takahashi / Advances in Cichlid Research V: Behavior, Ecology and Evolutionary Biology
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, Y., Wang, Y., Zhang, C. et al. Variation in pH, HCO3−, carbonic anhydrases, and HCO3− transporters in Nile tilapia during carbonate alkalinity stress. Hydrobiologia 850, 2447–2459 (2023). https://doi.org/10.1007/s10750-022-05020-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-022-05020-6