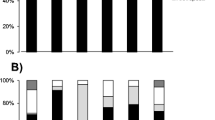
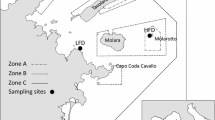
Abstract
Seaweeds play a key role in the rocky-shore fauna by providing shelter and food for many invertebrate species, which in turn compose the diet of benthic and pelagic predators. This study investigated the effect of the macroalgal host species on an assemblage of associated amphipods, and tested if the host mediates the vulnerability of amphipods to predation. The association of amphipods with the macroalgal hosts Dichotomaria marginata, Padina gymnospora, and Sargassum filipendula was evaluated seasonally for 1 year on a subtropical rocky shore on the southeastern coast of Brazil. The density of amphipods did not vary with season or host. However, the composition of amphipod species was sensitive to the host macroalgal species and varied temporally. A field experiment showed no clear effect of predation on the amphipod assemblage. Also, predation did not explain the spatial or temporal variation of the amphipod assemblage. Our results indicate that the identity of the macroalgal host drove the composition of the associated amphipods, while predation may have a minor role. Therefore, the loss of habitat-forming organisms such as seaweeds is expected to have drastic consequences for the composition of associated fauna, and consequently on the flow of matter and energy in coastal ecosystems.








Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Anderson, M. J., 2001. A new method for non-parametric multivariate analysis of variance. Austral Ecology 26: 32–46.
Angelini, C., A. H. Altieri, B. R. Silliman & M. D. Bertness, 2011. Interactions among foundation species and their consequences for community organization, biodiversity, and conservation. BioScience 61: 782–789.
Armitage, C. S. & K. Sjøtun, 2016. Epiphytic macroalgae mediate the impact of a non-native alga on associated fauna. Hydrobiologia 776: 35–49.
Benedetti-Cecchi, L., F. Pannacciulli, F. Bulleri, P. S. Moschella, L. Airoldi, G. Relini & F. Cinelli, 2001. Predicting the consequences of anthropogenic disturbance: large-scale effects of loss of canopy algae on rocky shores. Marine Ecology Progress Series 214: 137–150.
Bertness, M. D., G. H. Leonard, J. M. Levine, P. R. Schmidt & A. O. Ingraham, 1999. Testing the relative contribution of positive and negative interactions in rocky intertidal communities. Ecology 80: 2711–2726.
Best, R. J. & J. J. Stachowicz, 2014. Phenotypic and phylogenetic evidence for the role of food and habitat in the assembly of communities of marine amphipods. Ecology 95: 775–786.
Bueno, M., G. M. Dias & F. P. P. Leite, 2017. The importance of shore height and host identity for amphipod assemblages. Marine Biology Research 13: 870–877.
Chemello, R. & M. Milazzo, 2002. Effect of algal architecture on associated fauna: some evidence from phytal molluscs. Marine Biology 140: 981–990.
Choat, J. H. & P. D. Kingett, 1982. The influence of fish predation on the abundance cycles of an algal turf invertebrate fauna. Oecologia 54: 88–95.
Christie, H., K. M. Norderhaug & S. Fredriksen, 2009. Macrophytes as habitat for fauna. Marine Ecology Progress Series 396: 221–233.
Coull, B. C. & J. B. J. Wells, 1983. Refuges from fish predation: experiments with phytal meiofauna from the New Zealand rocky intertidal. Ecology 64: 1599–1609.
De Oliveira, D. A. S., S. Derycke, C. M. C. Da Rocha, D. F. Barbosa, W. Decraemer & G. A. P. Dos Santos, 2016. Spatiotemporal variation and sediment retention effects on nematode communities associated with Halimeda opuntia (Linnaeus) Lamouroux (1816) and Sargassum polyceratium Montagne (1837) seaweeds in a tropical phytal ecosystem. Marine Biology 163: 102–114.
Dubiaski-Silva, J. & S. Masunari, 2008. Natural diet of fish and crabs associated with the phytal community of Sargassum cymosum C. Agardh, 1820 (Phaeophyta, Fucales) at Ponta das Garoupas, Bombinhas, Santa Catarina State, Brazil. Journal of Natural History 42: 1907–1922.
Duffy, J. E., 1990. Amphipods on seaweeds: partners or pests? Oecologia 83: 267–276.
Duffy, J. E. & M. E. Hay, 1991. Food and shelter as determinants of food choice by an herbivorous marine amphipod. Ecology 72: 1286–1298.
Duffy, J. E. & M. E. Hay, 1994. Herbivory resistance to seaweed chemical defense: the roles of mobility and predation risk. Ecology 75: 1304–1319.
Duffy, J. E. & M. E. Hay, 2000. Strong impacts of grazing amphipods on the organization of a benthic community. Ecological Monographs 70: 237–263.
Duggins, D. O. & J. E. Eckman, 1997. Is kelp detritus a good food for suspension feeders? Effects of kelp species, age and secondary metabolites. Marine Biology 128: 489–495.
Eckman, J. E., D. O. Duggins & A. T. Sewell, 1989. Ecology of understory kelp environments. I. Effects of kelps on flow and particle transport near the bottom. Journal of Experimental Marine Biology and Ecology 129: 173–187.
Engqvist, L., 2005. The mistreatment of covariate interaction terms in linear model analyses of behavioural and evolutionary ecology studies. Animal Behaviour 70: 967–971.
Gibbons, M. J., 1988. The impact of sediment accumulations, relative habitat complexity and elevation on rocky shore meiofauna. Journal of Experimental Marine Biology and Ecology 122: 225–241.
Guerra-García, J. M., J. M. Tierno de Figueroa, C. Navarro-Barranco, M. Ros, J. E. Sánchez-Moyano & J. Moreira, 2014. Dietary analysis of the marine Amphipoda (Crustacea: Peracarida) from the Iberian Peninsula. Journal of Sea Research 85: 508–517.
Hacker, S. D. & R. S. Steneck, 1990. Habitat architecture and abundance and body-size-dependent habitat selection of a phytal amphipod. Ecology 71: 2269–2285.
Hammerschlag-Peyer, C. M., J. E. Allgeier & C. A. Layman, 2013. Predator effects on faunal community composition in shallow seagrass beds of The Bahamas. Journal of Experimental Marine Biology and Ecology 446: 282–290.
Heck, K. L. & G. S. Wetstone, 1977. Habitat complexity and invertebrate species richness and abundance in tropical seagrass meadows. Journal of Biogeography 4: 135–142.
Herkül, K., V. Lauringson & J. Kotta, 2016. Specialization among amphipods: the invasive Gammarus tigrinus has narrower niche space compared to native gammarids. Ecosphere 7: e01306.
Holmlund, M. B., C. H. Peterson & M. E. Hay, 1990. Does algal morphology affect amphipod susceptibility to fish predation? Journal of Experimental Marine Biology and Ecology 139: 65–83.
Huang, Y. M., J. B. McClintock, C. D. Amsler, K. J. Peters & B. J. Baker, 2006. Feeding rates of common Antarctic gammarid amphipods on ecologically important sympatric macroalgae. Journal of Experimental Marine Biology and Ecology 329: 55–65.
Jacobucci, G. B., M. O. Tanaka & F. P. P. Leite, 2009. Temporal variation of amphipod assemblages associated with Sargassum filipendula (Phaeophyta) and its epiphytes in a subtropical shore. Aquatic Ecology 43: 1031–1040.
Kelaher, B. P., 2003. Changes in habitat complexity negatively affect diverse gastropod assemblages in coralline algal turf. Oecologia 135: 431–441.
Koivisto, M. E. & M. Westerbom, 2010. Habitat structure and complexity as determinants of biodiversity in blue mussel on sublittoral rocky shores. Marine Biology 157: 1463–1474.
Kolding, S. & T. M. Fenchel, 1979. Coexistence and life cycle characteristics of five species of the amphipod genus Gammarus. Oikos 33: 323–327.
Kraufvelin, P. & S. Salovius, 2004. Animal diversity in Baltic rocky shore macroalgae: can Cladophora glomerate compensate for lost Fucus vesiculosus? Estuarine, Coastal and Shelf Science 61: 369–378.
Kraufvelin, P., S. Salovius, H. Christie, F. E. Moy, R. Karez & M. F. Pedersen, 2006. Eutrophication-induced changes in benthic algae affect the behaviour and fitness of the marine amphipod Gammarus locusta. Aquatic Botany 84: 199–209.
Lippert, H., K. Iken, E. Rachor & C. Wiencke, 2001. Macrofauna associated with macroalgae in the Kongsfjord (Spitsbergen). Polar Biology 24: 512–522.
Littler, D. S., M. M. Littler, K. E. Bucher & J. N. Norris, 1989. Marine Plants of the Caribbean. A Field Guide from Florida to Brazil. Smithsonian Institution Press, Washington, DC.
Lürig, M. D., R. J. Best & J. J. Stachowicz, 2016. Microhabitat partitioning in seagrass mesograzers is driven by consistent species choices across multiple predator and competitor contexts. Oikos 125: 1324–1333.
Machado, G. B. O., A. B. Neufeld, S. A. Dena, S. G. L. Siqueira & F. P. P. Leite, 2015. Variation of amphipod assemblage along the Sargassum stenophyllum (Phaeophyta, Fucales) thallus. Nauplius 23: 73–78.
Machado, G. B. O., S. G. L. Siqueira & F. P. P. Leite, 2017. Abundance, performance, and feeding preference of herbivorous amphipods associated with a host alga-epiphyte system. Journal of Experimental Marine Biology and Ecology 486: 328–335.
Martin-Smith, K. M., 1993. Abundance of mobile fauna: the role of habitat complexity and predation by fishes. Journal of Experimental Marine Biology and Ecology 174: 243–260.
McDonald, P. S. & B. L. Bingham, 2010. Comparing macroalgal food and habitat choice in sympatric tube-building amphipods, Ampithoe lacertosa and Peramphithoe humeralis. Marine Biology 157: 1513–1524.
Moksnes, P., M. Gullström, K. Tryman & S. Baden, 2008. Trophic cascades in a temperate seagrass community. Oikos 117: 763–777.
Östman, Ö., J. Eklöf, B. K. Eriksson, J. Olsson, P. Moksnes & U. Bergström, 2016. Top-down control as important as nutrient enrichment for eutrophication effects in North Atlantic coastal ecosystems. Journal of Applied Ecology 53: 1138–1147.
Pereira, P. H. C. & G. B. Jacobucci, 2008. Dieta e comportamento alimentar de Malacoctenus delalandii. Biota Neotropica 8: 141–149.
Poore, A. G. B. & P. D. Steinberg, 1999. Preference-performance relationships and effects of host plant choice in an herbivorous marine amphipod. Ecological Monographs 69: 443–464.
Poore, A. G. B., M. J. Watson, R. Nys, J. K. Lowry & P. D. Steinberg, 2000. Patterns of host use among alga- and sponge-associated amphipods. Marine Ecology Progress Series 208: 183–196.
Prathep, A., R. H. Marrs & T. A. Norton, 2003. Spatial and temporal variations in sediment accumulation in an algal turf and their impact on associated fauna. Marine Biology 142: 381–390.
Quinn, G. & M. Keough, 2002. Experimental Design and Data Analysis for Biologists. Cambridge University Press, New York.
Råberg, S. & L. Kautsky, 2007. A comparative biodiversity study of the associated fauna of perennial fucoids and filamentous algae. Estuarine, Coastal and Shelf Science 73: 249–258.
R Core Team, 2015. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna.
Saarinen, A., S. Salovius-Laurén & J. Mattila, 2017. Epifaunal community composition in five macroalgal species—What are the consequences if some algal species are lost? Estuarine, Coastal and Shelf Science 207: 402–413.
Sazima, I., 1986. Similarities in feeding behavior between some marine and freshwater fishes in two tropical communities. Journal of Fish Biology 29: 53–65.
Simons, E. B., 1906. A morphological study of Sargassum filipendula. Botanical Gazette 41: 161–182.
Sotka, E. E., 2007. Restricted host use by the herbivorous amphipod Peramphithoe tea is motivated by food quality and abiotic refuge. Marine Biology 151: 1831–1838.
Steele, L. & J. F. Valentine, 2015. Seagrass deterrence to mesograzer herbivory: evidence from mesocosm experiments and feeding preference trials. Marine Ecology Progress Series 524: 83–94.
Széchy, M. T. M. & E. J. Paula, 2000. Padrões estruturais quantitativos de bancos de Sargassum (Phaeophyta, Fucales) do litoral dos estados do Rio de Janeiro e São Paulo, Brasil. Revista Brasileira de Botânica 23: 121–132.
Tanaka, M. O. & F. P. P. Leite, 2003. Spatial scaling in the distribution of macrofauna associated with Sargassum stenophyllum (Mertens) Martius: analyses of faunal groups, gammarid life habits, and assemblage structure. Journal of Experimental Marine Biology and Ecology 293: 1–22.
Tanaka, M. O. & F. P. P. Leite, 2004. Distance effects on short-term recolonization of Sargassum stenophyllum by mobile fauna, with an analysis of gammarid life habits. Journal of the Marine Biological Association of the United Kingdom 84: 901–910.
Taylor, R. B. & R. G. Cole, 1994. Mobile epifauna on subtidal brown seaweeds in northeastern New Zealand. Marine Ecology Progress Series 115: 271–282.
Thomsen, M. S., T. Wernberg, A. Altieri, F. Tuya, D. Gulbransen, K. J. McGlathery, M. Holmer & B. R. Silliman, 2010. Habitat cascades: the conceptual context and global relevance of facilitation cascades via habitat formation and modification. Integrative and Comparative Biology 50: 158–175.
Ulrich, I., K. Anger & U. Schöttler, 1995. Tube-building in two epifaunal amphipod species, Corophium insidiosum and Jassa falcata. Helgoländer Meeresuntersuchungen 49: 393–398.
Vázquez-Luis, M., P. Sanchez-Jerez & J. T. Bayle-Sempere, 2010. Effects of Caulerpa recemosa var. cylindracea on prey availability: an experimental approach to predation of amphipods by Thalassoma pavo (Labridae). Hydrobiologia 654: 147–154.
Vérges, A., P. D. Steinberg, M. E. Hay, A. G. B. Poore, A. H. Campbell, et al., 2014. The tropicalization of temperate marine ecosystems: climate-mediated changes in herbivory and community phase shifts. Proceedings of the Royal Society B 281: 20140846.
Webb, C. O., D. D. Ackerly, M. A. McPeek & M. J. Donoghue, 2002. Phylogenies and community ecology. Annual Review of Ecology and Systematics 33: 475–505.
Wernberg, T., D. A. Smale, F. Tuya, M. S. Thomsen, T. J. Langlois, et al., 2013. An extreme climatic event alters marine ecosystem structure in a global biodiversity hotspot. Nature Climate Change 3: 78–82.
Zamzow, J. P., C. D. Amsler, J. B. McClintock & B. J. Baker, 2010. Habitat choice and predator avoidance by Antarctic amphipods: the roles of algal chemistry and morphology. Marine Ecology Progress Series 400: 155–163.
Acknowledgements
We thank Aline Neufeld for the assistance with field and laboratory work, and Pedro Bergamo for valuable comments. We are also grateful to anonymous reviewers for their comments and suggestions. This study was financially supported by a doctoral scholarship from the São Paulo Research Foundation (FAPESP) to GBO Machado (Grant No. 2013/17629-9).
Funding
This study was financially supported by the São Paulo Research Foundation (FAPESP) Grant to GBO Machado (Grant No. 2013/17629-9).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for sampling, care, and experimental use of organisms for the study have been followed and all necessary approvals have been obtained.
Additional information
Handling editor: Jonne Kotta
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Machado, G.B.O., Ferreira, A.P., Bueno, M. et al. Effects of macroalgal host identity and predation on an amphipod assemblage from a subtropical rocky shore. Hydrobiologia 836, 65–81 (2019). https://doi.org/10.1007/s10750-019-3941-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-019-3941-8