Abstract
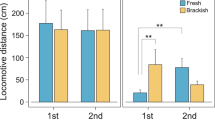
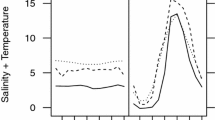
The widely distributed snail species Theodoxus fluviatilis (Linneaus, 1758) is rather diverse in shell size and patterning. It has formed regional subgroups in northern Germany that appear separate in either fresh- or brackish water, yet, according to literature, are indistinguishable by morphology, anatomy or mtRNA markers. Tolerance towards challenging salinities, however, is distinct. The question as to whether the observed difference in salinity tolerance is a result of local adaptation or of phenotypic plasticity remains. In this study, physiological limits to salinity stress and tolerance shift abilities were compared between individuals from both subgroups in a salinity transfer experiment and acclimation regime followed by a survival analysis. Brackish water animals struggled in freshwater but performed much better than freshwater individuals when directly transferred into higher salinities. With acclimation to their challenging salinities, individuals of both subgroups achieved shifts in the slopes of their reaction norms and were able to survive salinity levels otherwise lethal to them. Even with this shift freshwater animals were not able reach the tolerance levels of brackish water individuals. The results indicate that phenotypic plasticity as well as genetic adaptation may determine osmotolerance in regional subgroups of T. fluviatilis.



Similar content being viewed by others
References
Bandel, K., 2001. The history of Theodoxus and Neritina connected with description and systematic evaluation of related Neritimorpha (Gastropoda). Mitteilungen aus dem Geologisch-Palaeontologischen Institut der Universitaet Hamburg 85: 65–164.
Becker, K., 1949. Untersuchungen über das Farbmuster und das Wachstum der Molluskenschale. Biologisches Zentralblatt 68(7/8): 263–288.
Bondesen, P., 1940. Preliminary investigations into the development of Neritina fluviatilis L. in brackish and fresh water. Videnskabelige Meddelelser Dansk Naturhistorisk Forening 104: 283–318.
Bowler, K., 2005. Acclimation, heatshock and hardening. Journal of Thermal Biology 30: 125–130.
Bunje, P. M., 2005. Pan-European phylogeography of the aquatic snail Theodoxus fluviatilis (Gastropoda: Neritidae). Molecular Ecology 14(14): 4323–4340.
Constantini, D., N. B. Metcalfe & P. Monaghan, 2010. Ecological processes in a hormetic framework. Ecology Letters 13(11): 1435–1447.
Cypser, J. R. & T. E. Johnson, 2002. Multiple stressors in Caenorhabditis elegans induce stress hormesis and extended longevity. Journal of Gerontology 57A(3): B109–B114.
Gilles, R. & C. Jeuniaux, 1979. Osmoregulation and ecology in media of fluctuating salinity. R. Gilles (Ed.), Mechanisms of osmoregulation in animals. John Wiley, Chichester: 581–608.
Glöer, P. & V. Pešić, 2015. The morphological plasticity of Theodoxus fluviatilis (Linnaeus, 1758) (Mollusca: Gastropoda: Neritidae). Ecologica. Montenegrina 2(2): 88–92.
Harrington, D. P. & T. R. Fleming, 1982. A class of rank test procedures for censored survival data. Biometrika 69: 553–566. https://doi.org/10.1093/biomet/69.3.553.
Hubendick, B., 1947. Die Verbreitungsverhältnisse der Limnischen Gastropoden in Südschweden. Zoologiska Bidrag fran Uppsala 24: 423–559.
Johansen, A. C., H. Ditlevsen, F. Heerfordt, J. P. Jacobsen, A. Jessen, K. Jessen & H. Ussing, 1918. Randers fjords naturhistorie. Kopenhagen.
Kangas, P. & G. Skoog, 1978. Salinity tolerance of Theodoxus fluviatilis (Mollusca, Gastropoda) from freshwater and from different salinity regimes in the Baltic Sea. Estuarine and Coastal Marine Science 6: 409–416.
le Bourg, E., P. Valenti, P. Lucchetta & F. Payre, 2001. Effects of mild heat shocks at young age on aging and longevity in Drosophila melanogaster. Biogerontology 2: 155–164.
Loeschcke, V. & J. G. Sørensen, 2005. Acclimation, heat shock and hardening. Journal of Thermal Biology 30: 255–257.
Mattson, M. P., 2008. Homesis defined. Ageing Research Reviews 7: 1–7.
McMahon, R. F. & W. D. Russell-Hunter, 1981. The effects of physical variables and acclimation on survival and oxygen consumption in the high littoral salt-marsh snail, Melampus bidentatus say. The Biological Bulletin 161(2): 246–269.
Neumann, D., 1960. Osmotische Resistenz und Osmoregulation der Flußdeckelschnecke Theodoxus fluviatilis L. Biologisches Zentralblatt 79(5): 585–605.
Pierce, S. K., 1982. Invertebrate cell volume control mechanism: a coordinated use of intracellular amino acids and inorganic ions as osmotic solute. Biological Bulletin 163: 405–419.
Pohlert, T., 2014. The pairwise multiple comparison of mean ranks package (PMCMR). R package. http://CRAN.R-project.org/package=PMCMR.
R Development Core Team, 2008. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. URL http://www.R-project.org.
Skoog, G., 1976. Effects of acclimatization and physiological state on the tolerance to high temperatures and reactions to dessication of Theodoxus fluviatilis and Lymnea peregra. OIKOS 27: 50–56.
Skoog, G., 1978. Aspects on the biology and ecology of Theodoxus fluviatilis (L.) and Lymnea peregra (O.F. Müller) (Gastropoda) in the northern Baltic. Dissertation, University of Stockholm.
Symanowski, F. & J.-P. Hildebrandt, 2010. Differences in osmotolerance in freshwater and brackish water populations of Theodoxus fluviatilis (Gastropoda: Neritidae) are associated with differential protein expression. Journal of Comparative Physiology B 180(3): 337–346.
Therneau, T., 2015. A Package for Survival Analysis in S. version 2.38 (survival). R package. https://CRAN.R-project.org/package=survival.
Todd, M. & P. Dehnel, 1960. Effects of temperature and salinity on heat tolerance in two grapsoid crabs, Hemigrapsus nudus and Hemigrapsus oregonensis. The Biological Bulletin 118: 150–172.
Ulrich, H. & D. Neumann, 1956. Zur Biologie einer Salzwasserpopulation der Flußdeckelschnecke (Theodoxus fluviatilis L.). Natur und Jagd in Niedersachsen 219–222.
Yellon, D. M. & J. M. Downey, 2003. Preconditioning the myocardium: from cellular physiology to clinical cardiology. Physiological Reviews 83: 1113–1151.
Zettler, M. L., J. Frankowski, R. Bochert & M. Röhner, 2004. Morphological and ecological features of Theodoxus fluviatilis (LINNAEUS, 1758) from Baltic brackish water and German freshwater populations. Journal of Conchology 38: 305–316.
Acknowledgements
We would like to thank all members of the working group of ‘Animal Physiology and Biochemistry’ at the University of Greifswald for their help with the snail collections. A special thanks goes to Alexander Kolb for helping with the snail husbandry and Dana Kleimeier for critical reviewing the R script of the survival analysis. We also gratefully thank PD Dr. Blindow (Biological Station Hiddensee) for obtaining permissions to collect snails on the island. This research was funded by the DFG Research Training Group RESPONSE (DFG GRK2010). Permission for the snail collections was given by Nationalparkamt Vorpommersche Boddenlandschaft, Born, Germany (24-5303.3).
Author information
Authors and Affiliations
Contributions
AAW and J-PH conceived the ideas and designed methodology; AAW collected and analysed the data; AAW drafted the manuscript. All authors contributed to the preparation of the final manuscript and gave approval for publication.
Corresponding author
Additional information
Handling editor: Jonne Kotta
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wiesenthal, A.A., Müller, C. & Hildebrandt, JP. Potential modes of range shifts in euryhaline snails from the Baltic Sea and fresh water lakes in northern Germany. Hydrobiologia 811, 339–350 (2018). https://doi.org/10.1007/s10750-017-3501-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-017-3501-z